You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Dental implant-supported fixed restorations have long been established as a preferred treatment option for full-arch rehabilitation. Placement of implants based on the planned prosthetic position should coincide with the triangle of bone for predictable long-term function, with the base of the triangle positioned to the basal bone and the point of the triangle to the crestal portion of bone.1 Techniques using computed tomographic data combined with prosthetically driven scan appliances have been described.2 More recently 3-dimensional (3D) planning using cone-beam computed tomography (CBCT) scan data has become essential in achieving these restorative goals. Guided surgery using 3D-printed surgical guides has increased the efficiency and accuracy of implant placement by transferring the virtual plan from the computer to the actual, clinical patient situtation.3,4 Technological advancements and the availability of desktop 3D printers now allow practitioners to plan and print surgical guides and provisional restorations for full-arch cases in-house.5
In-House 3D Printing Process
The concept of "all-on-X" treatment for the rehabilitation of the terminal dentition has grown in popularity due to the predictability of the treatment, efficiency of the surgical procedure, and, in most cases, the ability to provide graftless solutions.6 Being able to treat the entire arch or mouth at one time also has inherent cost advantages for both the clinician and patient. In the past, full-arch procedures typically involved freehand placement of implants with conversion of an existing denture (in the case of edentulous patients) or prefabricated denture (in the case of dentate patients) into an immediate provisional fixed detachable appliance on four or more implants. This method relied heavily on the experience of the surgeon and necessitated long appointments and typically a laboratory technician to process the denture.
Surgical guides and prefabricated provisionals for all-on-X treatment changed the landscape by making these procedures more efficient and predictable.7 This is especially true for bone-supported reduction/stacked drilling guides. Laboratory costs and turnaround times still remain a challenge. Now, however, in-house 3D printing may be used to overcome these challenges.8,9 Designing and printing surgical guides, dentures,10 and other dental appliances in-house with turnaround times of hours or days instead of weeks is now possible. Typical planning of a case takes several hours and varies based on the complexity of the case, while the printing of the appliances requires about an hour per unit even on low-cost printers.11 Outsourcing design to a laboratory and then 3D printing in-house is also an appealing cost- and time-saving option for clinicians who may not be interested in learning new software, as surgical guides printed on desktop 3D printers have been shown to have similar accuracy to laboratory-produced guides.12,13
There are seven steps involved in the in-house 3D printing process: (1) data acquisition, comprising CBCT scan, intraoral scan, and dual denture or scan appliance scan; (2) merging and alignment of data sets; (3) bone segmentation of the CBCT image; (4) planning of implant positions and design of the bone reduction guide; (5) design of the drilling guide as a separate guide or stacked onto the bone reduction guide; (6) merging of the prosthetic model to create the temporary appliance; and (7) 3D printing of the bone reduction guide, drilling guide, and provisional prosthetic.
As with any procedure the process starts with proper data acquisition. Two different clinical presentations are possible: the fully edentulous patient or the partially edentulous/dentate patient. Each requires a different protocol.
The process when treating a fully edentulous patient requires a dual-scan protocol. This begins by taking a CBCT scan of the patient wearing a properly seated denture that is used as a scan appliance in occlusion with radiopaque fiducial or impression materials attached to the denture.14 A CBCT scan of the denture itself (with fiducial markers still attached) is then taken. The denture used for these two scans should not contain any metal and should have the proper contours, esthetics, and function desired in the final restoration.15 A CBCT image of the denture (scan appliance) provides a very clear image of the denture because the CBCT software averages the densities of each particular scan to improve image quality. This attribute of CBCT imaging makes it an ideal modality for dental imaging of hard tissues such as teeth and bone. However, this occurs at the expense of low-dentisty structures such as soft tissue. Novel techniques utilizing intraoral scanners combined with CBCT imaging for fully edentulous cases have also been presented in the literature.16
For a partially edentulous or dentate patient a CBCT with the maxillary and mandibular teeth apart and an intraoral scan of the arch are needed. Intraoral scans of both arches will be necessary if the vertical dimension or smile design needs to be altered.
In the fully edentulous patient scenario the data sets, which, to reiterate, consist of a CBCT scan of a scan appliance (with fiducial markers attached) in the mouth and a CBCT scan of the same scan appliance by itself, will be merged manually by matching the fiducial markers. In the partially edentulous or dentate patient scenario, the data sets, comprising a CBCT of the jaw and an intraoral scan of the teeth and soft tissues, will be merged either automatically by the software or by matching anatomical points from the CBCT 3D rendering with the same points on the intraoral scan. If the partially edentulous or dentate patient has metal or zirconia crowns a scan appliance with fiducial markers may be needed, and the merging process becomes similar to that of the fully edentulous patient scenario with the same data sets required for that process.
In either the fully edentulous or partially edentulous/dentate patient scenario, because of beam-hardening artifacts17 theanatomy of the teeth will be distorted and the teeth will have a shrapnel-like appearance radiographically in the horizontal plane. This makes it difficult or impossible to align the CBCT to the intraoral scan because anatomical landmarks that are used in the alignment process will not be visible. The use of a scan appliance with fiducial markers is recommended for any patient with five or less unrestored teeth to compensate for this limitation. While this is an added step and requires the fabrication of a separate appliance, it is the optimal way to ensure an accurate alignment of a defined tooth model to the CBCT image. Fabrication of a scan appliance can be done economically chairside in a few minutes without the need for a second appointment through the use of a standard impression tray with fiducial markers attached and common polyvinyl siloxane (PVS) impression material.
After data acquisition, alignment, and design of the various surgical guides and provisional prosthetics, the digital models are exported as industry standard STL (standard tessellation language) files and printed on in-office 3D printers using resins that are US Food and Drug Administration (FDA) certified for each appliance to ensure biocompatibility for class I or class IIa dental appliances. The FDA considers surgical guides to be class I medical devices (to be used for less than an hour in the mouth), while it deems temporary restorations as class IIa and able to be used long term in the mouth. Practitioners must be sure they are using FDA-compliant materials and follow the manufacturer's guidelines for post-processing and clinical use to ensure biocompatibility.18 Depending on the implant system, metal sleeves may need to be bonded into the surgical guide. This can be easily done using the same resin that is used for 3D printing the guide.
The provisional prosthesis is characterized with pink composite, stains, and glaze. The 3D printing process includes post-processing the appliances after they are printed by washing them in isopropyl alcohol, drying them, and then curing them in an ultraviolet (UV) chamber. Automated wash systems are available that greatly simplify the process by automatically washing and drying the appliances. The appliances are then post-cured in a UV light oven for the correct amount of time, which varies by resin type. Attention to detail is important throughout the process of data acquisition, planning, and fabrication because errors are additive through each step.19
Digitally planning a case can be challenging because there are many decisions to make and various ways to treat a case using surgical guides. This can be simplified if the design process mimics the surgical treatment. One suggestion is for the clinician to think about the clinical steps involved and create a digital analog for each step. For example, if a surgical flap is planned the surgical guide will need to sit on the bone. Or if bone reduction is planned a bone reduction guide will be necessary. Another consideration may be whether the bone reduction guide will have to be removed in favor of a separate drilling guide, or whether the drilling guide can be seated on the fixated bone reduction guide. Most cases, especially dentate ones, are best treated with a bone reduction/stacked drilling guide for accuracy, speed, and ease during surgery.
Clinical Case
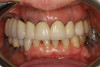
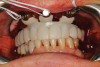
A clinical case of a 76-year-old female patient with a terminal maxillary dentition (Figure 1 and Figure 2) will be used to illustrate this concept of the appliance design mimicking the clinical workflow. The treatment plan for this patient consisted of full-arch extractions, bone reduction, immediate placement of six dental implants, and an immediate fixed provisional restoration. Data acquisition consisted of CBCT imaging a scan appliance (a PVS impression with fiducial markers due to the presence of numerous porcelain-fused-to-metal crowns in the maxillary teeth) in the mouth and a CBCT of the scan appliance by itself. In this case a CS 8100 CBCT system (Carestream Dental, carestreamdental.com) was used. Other comparable CBCT systems include: i3D, Vatech, vatechamerica.com; PreXion 3D Excelsior, PreXion, prexion.com; Planmeca ProMax®, Planmeca, planmeca.com.
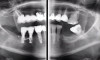
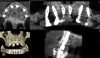
Once the data sets were acquired they were imported into implant planning software and merged by matching the fiducial markers. In this case, the implant planning software used was Blue Sky Plan 4 (BlueSkyBio, blueskybio.com). Other comparable implant planning software products include: co-DiagnostiX®, Dental Wings, codiagnostix.com; Simplant, Dentsply Sirona, dentsplysirona.com; 3Shape Implant Studio, 3Shape, 3shape.com. The scan of the scan appliance in the mouth is shown in the upper left quadrant of Figure 3 and the scan of the scan appliance by itself is shown in the lower left quadrant of Figure 3, while the same scan of the scan appliance is shown in the upper right quadrant of Figure 3 with the visibility adjusted to show only the fiducial markers. The lower right quadrant of Figure 3 shows the result of aligning the fiducial markers.
A bone segmentation whereby each slice of the CBCT is manually traced to create a 3D model of the bone is necessary for creating a bone reduction guide. In some respects this is like 3D printing in reverse. Manual bone segmentation is tedious and time-consuming and is frequently outsourced. Ongoing research holds the promise of automated bone segmentation techniques.20
Implant positions are planned based on the desired final prosthetic positions of the teeth (whether natural teeth, denture teeth, or a digital wax-up, which are visible in the software) (Figure 4) and available bone. In this case six tapered implants were planned, with the four anterior implants planned parallel to each other and the two distal-most implants planned at 30-degree angles to the four anterior ones. The plan included the use of 30-degree angled multiunit abutments to correct the distal implant angulations. Placing six implants across the arch reduces stress on the implants through enhanced distribution and reduced cantilevers.21,22 By planning the use of angled multiunit abutments and placing the implants using a surgical guide the restorative procedures can be simplified and the try-in or replacement of the multiunit abutments avoided.23
The implant positions may be subcrestal depending on existing bone anatomy and the amount of interocclusal space present. The interocclusal space should be no less than 15 mm and preferably greater to accommodate the prosthetic components and to allow for enough material thickness to prevent flexure and/or fracture.24,25 If the bony anatomy is knife-edged or there is inadequate interocclusal space the use of a bone reduction guide will be necessary as it was in this case.
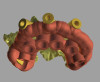
Bone reduction is achieved by designing a guide on the segmented 3D bone model and then virtually cutting both the guide and the 3D bone model simultaneously to the level of the planned implants. The bone reduction guide is then modified to add guide fixation pins (Figure 5 and Figure 6).
From this process two new models are created: a bone reduction guide, which will be fixated to the bone clinically via fixation pins, and a reduced bone model. By combining these two models a stacked drilling guide can now be created that sits on the bone reduction guide. This new drilling guide will be indexed to the bone reduction guide and incorporate guide tubes for each individual implant (Figure 7 and Figure 8).
The provisional prosthesis is also designed to fit on the bone reduction guide by aligning a digital wax-up and a pre-existing dentition intraoral scan or, as in this case, an inverted model created from the CBCT PVS scan. This technique, which was developed by the author and subsequently added to the implant treatment planning software by the manufacturer, takes the place of and eliminates the need for an intraoral scan. The resulting model is merged to indexing wings designed on the bone reduction guide to create the provisional appliance (Figure 9 and Figure 10). The prosthesis will be properly indexed to the occlusion and the bone reduction guide for an easy chairside pick-up. The openings for the implant pick-ups are created by digital (Boolean difference) subtraction of the abutment projections from the prosthetic model in the design software.
The final step is to 3D-print all three appliances: the bone reduction guide, drilling guide, and provisional prosthesis. In this case the appliances were printed on a liquid crystal display (LCD) 3D printer (Phrozen Shuffle, Phrozen Ltd, phrozen3d.com) using 405-nm UV photoinitiated acrylic monomer resins. Similarly capable available printers include: Form 3B, Formlabs, dental.formlabs.com; NextDent® 5100, NextDent, nextdent.com; SprintRay Pro, SprintRay, sprintray.com. A surgical guide resin (NextDent SG, NextDent) was used for the surgical guides, and a microfilled hybrid composite resin (NextDent C&B MFH, NextDent) was used for the provisional. Other similar resins can also be used, such as Formlabs surgical guide resin (Formlabs), SprintRay surgical guide resin (SprintRay), Formlabs temporary C&B resin (Formlabs), and Dentca™ Denture Teeth resin (Dentca, dentca.com). The hybrid composite resin used for the provisional has a flexural strength of 107 MPa, according to the manufacturer, which is similar to CAD/CAM-milled polymethyl methacrylate (PMMA) materials26 that currently are commonly used for this purpose.
Horizontal positioning of the appliance on the 3D printer build platform has been recommended to ensure dimensional accuracy.27 This has been this author's experience as well. In this case all three appliances were printed at 100-µm layer heights; at this height, layer lines may be visible but are typically obscured by the use of light-cured glazes during the characterization process. Printing at 50-µm layer heights will reduce the visibility of these lines but adds to the printing time.
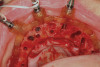
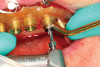
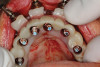
The workflow described in this case is just one example, and there are numerous ways to achieve the same goal. This particular workflow is relatively easy to execute and efficient from the standpoint of planning, fabrication, and clinical application. Figure 11 and Figure 12 demonstrate the accurate placement of the implants as can be seen by the copings exiting directly into the openings in the printed provisional. The provisional was indexed and the copings picked-up by injecting a provisional bisacryl composite resin. The finished 3D-printed provisional was characterized with pink composite and fixated to the implants with abutment screws, and the access holes were sealed with sterilized teflon tape and composite resin (Figure 13).
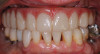
The rigidly fixated provisional was left undisturbed for a minimum of 5 months to allow for osseointegration of the implants. The patient was advised to maintain a soft diet during that time. To prevent fracture of the provisional, cantilevers should be eliminated or minimized and any metal inserts should have at least 4 mm of thickness around them. If any fractures or chips have occurred on the provisional they can be repaired intraorally or extraorally with standard light-cured bisphenol A-glycidyl methacrylate (bis-GMA) or bisacryl composite resins. Figure 14 and Figure 15 illustrate the in-house 3D-printed provisional at 5 months with excellent bone levels on the implants.
Conclusion
Immediate-load full-arch fixed prostheses have been shown to have similar long-term success rates compared to staged approaches.28 Being involved in the design and fabrication process gives clinicians placing the implants an intimate connection to both the prosthetic and surgical use of the printed appliances. The surgical process becomes more efficient, and if changes or alterations are needed during the surgery clinicians can readily make modifications. In the author's experience, the reduced stress and time and cost savings that come with digital planning and 3D printing are certainly worth the preparation time.
About the Author
Rick Ferguson, DMD
Clinical Assistant Professor, Department of Community Dentistry and Behavioral Sciences, University of Florida College of Dentistry, Gainesville, Florida;
Private Practice, Davie, Florida
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Ganz SD. The triangle of bone-a formula for successful implant placement and restoration. Implant Soc. 1995;5(5):2-6.
2. Ganz SD. Presurgical planning with CT-derived fabrication of surgical guides. J Oral Maxillofac Surg. 2005;63(9 suppl 2):59-71.
3. Magrin GL, Rafael SNF, Passoni BB, et al. Clinical and tomographic comparison of dental implants placed by guided virtual surgery versus conventional technique: a split-mouth randomized clinical trial. J Clin Periodontol. 2020;47(1):120-128.
4. Oberoi G, Nitsch S, Edelmayer M, et al. 3D printing-encompassing the facets of dentistry. Front Bioeng Biotechnol. 2018;6:172.
5. Zaharia C, Gabor AG, Gavrilovici AM, et al. Digital dentistry-3D printing applications. J Interdisciplinary Medicine. 2017;2(1):50-53.
6. Malo P, Rangert B, Nobre M. "All-on-Four" immediate-function concept with Brånemark System implants for completely edentulous mandibles: a retrospective clinical study. Clin Implant Dent Relat Res. 2003;5 (suppl 1):2-9.
7. Tonellini G, Saez Vigo R, Novelli G. Double guided surgery in All-on-4® concept: when ostectomy is needed. Int J Dent. 2018;2018:2672549.
8. Whitley D 3rd, Eidson RS, Rudek I, Bencharit S. In-office fabrication of dental implant surgical guides using desktop stereolithographic printing and implant treatment planning software: a clinical report. J Prosthet Dent. 2017;118(3):256-263.
9. Scherer MD. Implementing 3D printing technology in practice. ACP Messenger. Fall 2016;47(4):12-15.
10. Chung YJ, Park JM, Kim TH, et al. 3D printing of resin material for denture artificial teeth: chipping and indirect tensile fracture resistance. Materials (Basel). 2018;11(10):1798.
11. Dementyeva JN, Kashapov R, Kashapov L. Determination of optimal technology for manufacturing dental surgical guides. IOP Conf Series: Mater Sci Eng. 2018;412(1):012096.
12. Deeb GR, Allen RK, Hall VP, et al. How accurate are implant surgical guides produced with desktop stereolithographic 3-dimentional printers? J Oral Maxillofac Surg. 2017;75(12):2559.e1-2559.e8.
13. Yeung M, Abdulmajeed A, Carrico CK, et al. Accuracy and precision of 3D-printed implant surgical guides with different implant systems: an in vitro study. J Prosthet Dent. 2020;123(6):821-828.
14. Scherer MD, Roh HK. Radiopaque dental impression method for radiographic interpretation, digital alignment, and surgical guide fabrication for dental implant placement. J Prosthet Dent. 2015;113(4):343-346.
15. Almog DM, Torrado E, Meitner SW. Fabrication of imaging and surgical guides for dental implants. J Prosthet Dent. 2001;85(5):504-508.
16. Moura GF, Siqueira R, Meirelles L, et al. Denture scanning technique for computer-guided implant-supported restoration treatment of edentulous patients [published online ahead of print. J Prosthet Dent. 2020;S0022-3913(20)30253-30255.
17. Nagarajappa AK, Dwivedi N, Tiwari R. Artifacts: the downturn of CBCT image. J Int Soc Prev Community Dent. 2015;5(6):440-445.
18. Kurzmann C, Janjić K, Shokoohi-Tabrizi H, et al. Evaluation of resins for stereolithographic 3D-printed surgical guides: the response of L929 cells and human gingival fibroblasts. Biomed Res Int. 2017;2017:
4057612.
19. Marlière DAA, Demètrio MS, Picinini LS, et al. Accuracy of computer-guided surgery for dental implant placement in fully edentulous patients: a systematic review. Eur J Dent. 2018;12(1):153-160.
20. Wang L, Chen KC, Gao Y, et al. Automated bone segmentation from dental CBCT images using patch-based sparse representation and convex optimization. Med Phys. 2014;41(4):043503.
21. Silva GC, Mendonça JA, Lopes LR, Landre J Jr. Stress patterns on implants in prostheses supported by four or six implants: a three-dimensional finite element analysis. Int J Oral Maxillofac Implants. 2010;25(2):239-246.
22. Caramês J. A comprehensive classification to full arch implant rehabilitation. Portuguese Journal of Stomatology, Dental Medicine and Maxillofacial Surgery. 2019;60(4):175-188.
23. Wu YL, Wu AYJ. A method of fabricating an accurate repositioning device for relocating multiple multiunit abutments. J Prosthet Dent. 2017;118(4):564-566.
24. Misch CE. Contemporary Implant Dentistry. 3rd ed. St. Louis, MO: Mosby Elsevier; 2008:99-100.
25. Phillips K, Wong KM. Vertical space requirement for the fixed-detachable, implant-supported prosthesis. Compend Contin Educ Dent. 2002;23(8):750-756.
26. Alp G, Murat S, Yilmaz B. Comparison of flexural strength of different CAD/CAM PMMA-based polymers. J Prosthodont. 2019;28(2):e491-e495.
27. Vasques M, Laganá DC. Accuracy and internal fit of 3D printed occlusal splint, according to the printing position. Clin Lab Res Dent. 2018; doi: 10.11606/issn.2357-8041.clrd.2018.148012.
28. Gallardo YNR, da Silva-Olivio IR, Gonzaga L, Sesma N, Martin W. A systematic review of clinical outcomes on patients rehabilitated with complete-arch fixed implant-supported prostheses according to the time of loading. J Prosthodont. 2019;28(9):958-968.