You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Drugs that act directly on the heart can influence any of three cardiac activities, including contractility (force), rate, and intrinsic neural conduction. A number of mechanisms can be exploited but the most familiar are those that mimic or inhibit autonomic innervations to the heart. For example, anticholinergic agents and beta-adrenergic agonists increase most cardiac functions. It must be pointed out, however, that many additional drugs can influence cardiac function by mechanisms that are unrelated to autonomic innervations. Digitalis glycosides and calcium channel blockers are examples.
Inotropic drugs influence the contractility of the heart and, therefore, specifically target the myocardial cells. Positive inotropic agents increase myocardial strength, while those decreasing myocardial strength are described as negative inotropic agents. Chronotropic drugs influence heart rate by altering firing rates of specialized cells comprising the sinoatrial (SA) node. Tachycardia and bradycardia can be managed respectively using drugs having either negative or positive chronotropic effects. Dromotropic drugs influence conduction along neural conductive tissues. Drugs that decrease the speed of conduction through the cardiac neural conduction system are said to produce a negative dromotropic effect. They are vital in the management of arrhythmias in which these tissues are overly excitable.
DIGITALIS GLYCOSIDES
Digitalis, an extract from the foxglove leaf, was described as early as 1250 AD in the writings of Welsh physicians. Additional extracts have since been isolated from various species of foxglove and several other plants as well. Today, “digitalis” is used in a generic sense when referring to any of these preparations.
Digoxin is the only derivative of digitalis in current use. It produces three cardiotonic influences. Most familiar is an ability to increase myocardial contractility, which is the basis for its efficacy in managing patients with congestive heart failure (CHF). In addition to this positive inotropic influence on myocardial cells, digitalis also depresses specific electrical tissues of the heart. It slows the firing rate of the SA node producing a negative chronotropic effect, and delays conduction through the atrioventricular (AV) node imparting a negative dromotropic effect. These electrical influences not only provide a basis for treating atrial arrhythmias, such as atrial fibrillation, but they also introduce a significant potential for cardiac toxicity.
In recent years, the use of digoxin for heart failure has declined because a greater understanding of the condition has revealed a better response with vasodilators that “unload” stress and strain on the heart. Chronic atrial fibrillation is the remaining indication for digoxin. Atrial fibrillation is a condition in which numerous ectopic foci in the atria attempt to take over control of rate from the SA node. Collectively these foci fire as many as 300 times per minute and initiate impulses that spread throughout atrial tissues and toward the ventricles. Fortunately, the AV node blocks most of these impulses from entering the ventricles; otherwise, the ventricles would also fibrillate. In some cases, however, too many impulses reach the ventricle causing a rapid ventricular response. This rapid ventricular rate reduces the time for the ventricles to fill between beats and cardiac output suffers. The patient then experiences lightheadedness and syncope. The negative dromotropic effect of digoxin depresses the AV node, which allows fewer impulses to reach the ventricle. This is described as “slowing the ventricular response” to atrial fibrillation. The patient remains in atrial fibrillation, but is no longer symptomatic. Other drug classes that have negative dromotropic effects are becoming more popular for this condition because they are less toxic. These include the beta-blockers and calcium channel blockers, which will be addressed subsequently.
Digoxin has a low therapeutic index; as many as 25% of patients experience some degree of toxicity. The most common side effects are gastrointestinal related, but neural and visual disturbances also occur. Cardiac side effects are less common but can be life-threatening. Depression of the SA and AV nodes can produce bradycardia and a variety of heart blocks. In addition, digitalis increases intracellular calcium ion concentration and heightens cardiac excitability, which may lead to a variety of tachyarrhythmias. It is safe to say that digitalis intoxication can produce virtually any arrhythmia.1 Risk for cardiac toxicity is enhanced when potassium levels are low (hypokalemia) and may be a consequence of diuretic therapy or dialysis. These therapies may also produce low serum concentrations of magnesium or elevated calcium concentrations, which also predispose patients to cardiac arrhythmias. A summary of adverse effects associated with digitalis is presented in Table 1.
Dental Implications for Patients Medicated with Digoxin
Digoxin is being prescribed less frequently as the paradigms for managing heart failure and atrial fibrillation have shifted to other drug classes. For patients medicated with digoxin, it is always wise to record baseline pulse rate and rhythm before commencing dental treatment. Because its most serious side effects relate to cardiac arrhythmia, it is wise to use caution when administering epinephrine or anticholinergics that may lead to added cardiac excitation. In addition, macrolide and tetracycline antibiotics should be avoided because they elevate digoxin serum concentration and enhance the risk for digoxin toxicity.2
BETA-BLOCKERS
The therapeutic effects of the beta-blockers are largely attributed to their ability to act as antagonists on cardiac beta1-receptors, and thereby inhibit stimulatory effects of the sympathetic system. Beta-blockers are useful in the management of coronary artery disease (angina), arrhythmias, and hypertension. Presumably, a reduction in sympathetic stimulation of the heart accounts for their therapeutic effect in the former two cases. Reduction in heart rate corrects tachycardias and, for patients with ischemic heart disease, a slower heart rate reduces myocardial oxygen consumption and improves time for coronary perfusion. However, an explanation for the antihypertensive effect of beta-blockers has not been clarified. A reduction in contractility would explain a drop in systolic pressure, but over time there is a drop in peripheral resistance and diastolic pressure which has yet to be explained since beta-blockers are not vasodilators. If anything, blockade of vascular beta2-receptors would favor vasoconstriction, not vasodilation. Furthermore, agents having selective action at beta1-receptors are equally effective compared to nonselective beta-blockers. Several modes of action have been proposed, including reduced cardiac output and an inhibition of renin release which reduces subsequent angiotensin-induced vasoconstriction. Most likely, the antihypertensive effect of beta-blockers involves a combination of severalmechanisms.3,4
The efficacy of beta-blockers is confirmed by an impressive number of products that currently are available (Table 2). Propranolol (Inderal) is regarded as the prototype for comparisons that include beta-receptor selectivity, lipid solubility, and elimination half-life. Cardioselective agents have specificity for beta1-receptors, and are less likely to promote bronchospasm, normally attributed to blockade of beta2-receptors on bronchial smooth muscle. This is a particular concern when caring for patients having a history of asthma or chronic lung diseases. Agents having low lipid solubility are less likely to produce central nervous system (CNS) side effects, including lethargy and drowsiness.
One additional feature of certain beta-blockers is intrinsic sympathomimetic activity (ISA). This is a term that is actually synonymous with partial agonist. Agents having this property are not pure antagonists that block the receptor but instead activate it mildly. Nevertheless, their activation of receptors is far less than that provided by the body’s endogenous catecholamines, and they therefore act to inhibit this more excessive stimulation.
Despite these and other subtle differences, there are no compelling data to support superior efficacy of one agent over another in managing hypertension, coronary artery disease, or arrhythmias.3 Selection of a particular agent is essentially empiric. Adjustment in dosage, or selection of an alternative agent, is guided by each patient’s response to the initial agent.
In recent years, beta-blockers have been found useful for managing heart failure. This is a dramatic shift in paradigm since blocking beta-receptors would appear detrimental because they decrease contractility. However, the pathogenesis of heart failure has been found to include excessive sympathetic stimulation of the heart leading to hypertrophy, weakening, and loss of compliance. Although initiating beta blockade in these patients is initially met with a mild decline in ejection fraction, cardiac function actually improves over time.1 In addition, the newest generation of beta blockers has alpha-blocking properties that vasodilate and reduce afterload on the failing heart.
Dental Implications for Patients Medicated with Beta-Blockers
Essential information regarding commonly prescribed beta-blockers is summarized in Table 2. Patients medicated with nonselective beta-blockers have a significant risk for acute hypertensive episodes if they receive vasopressors contained in local anesthetics, ie, epinephrine or levonordefrin. The explanation for this interaction was thoroughly addressed in a previous continuing education article provided in this journal.5 While treating patients medicated with nonselective beta-blockers, it is wise to record blood pressure and heart rate before administering local anesthetic-vasopressor formulations, then reassess those parameters 3 to 5 minutes after each cartridge is administered before giving another.
The use of nonsteroidal anti-inflammatory drugs (NSAIDs) for more than 5 days may diminish the antihypertensive efficacy of most drug classes used to manage hypertension. This includes any of the beta-blockers.2
CALCIUM CHANNEL BLOCKERS
As their title suggests, calcium channel blockers inhibit the cellular entry of calcium ions. Calcium influx is particularly significant for the generation of impulses in SA and AV nodal cells of the heart, and for the contractile process of smooth muscle cells comprising the walls of arteries and large veins. Calcium channel blockade results in vasodilation and a reduction in heart rate. These are desired outcomes when managing angina, atrial arrhythmias, and hypertension. The precise structure and function of calcium channels vary among body tissues, and the various calcium blockers differ in their relative influences on channel subtypes located throughout the heart and in vascular smooth muscle.2 Verapamil (Calan) produces a greater influence on nodal tissues of the heart, whereas nifedipine (Procardia) has a greater influence on vascular smooth muscle. Because they are prescribed so often as vasodilators, newer generations of calcium channel blockers are derivatives of nifedipine and are more selective for so-called L-channel subtypes found in vascular smooth muscle (Table 3).
Dental Implications for Patients Medicated with Calcium Blockers
Verapamil and diltiazem compete with macrolide antibiotics for the same family of metabolizing enzymes in the liver—CYP3A4. Elevated levels of macrolides may result in cardiac toxicity related to prolonged QT intervals. Elevated levels of the calcium blockers may lead to bradycardias and atrioventricular block. Also, verapamil and diltiazem may inhibit metabolism of benzodiazepines, especially midazolam and triazolam, resulting in greater bioavailability following oral administration and a longer duration of sedation. These interactions do not occur with the remaining calcium channel blockers. Unlike other drug classes used to manage hypertension, the efficacy of calcium channel blockers is not reduced by concurrent use of NSAIDs.2
Calcium channel blockers are known to produce gingival enlargement, resembling that observed with phenytoin (Dilantin). While many agents have been implicated, isradipine (DynaCirc) is a notable exception and may be suggested to the patient’s physician if this problem evolves.6
ANTIARRHYTHMIC DRUGS
The heart is composed of two principal cell types—working cells and specialized cells. Working cells comprise the muscle or myocardium of the atria and ventricles. Specialized cells include the SA node, AV node, and the His-Purkinje system. These cells initiate and conduct electrical impulses throughout the myocardium, and this regulates the rhythm of a cardiac cycle. In order to initiate impulses, specialized cells have a property called “automaticity,” which reflects an ability to depolarize spontaneously. This is a key feature that distinguishes specialized cells from working cells.
An arrhythmia is an abnormality in rate or regularity of the cardiac cycle. Because the cycle is regulated by the heart’s specialized neural conductive tissues, it follows that antiarrhythmic drugs must act on these special cells. A common convention by which antiarrhythmic drugs are categorized is predicated on the primary mechanism by which they produce an antiarrhythmic effect (Table 4). Depolarization of atrial neural conductive tissues is initiated by calcium influx and the calcium channel blockers are most effective in managing supraventricular arrhythmias. The benefit of beta-blockers lies primarily in their ability to offset excessive sympathetic stimulation which renders them particularly useful for arrhythmias triggered by stress. The pharmacology of Class I and III antiarrhythmics is among the most complex of all cardiovascular drugs. They are used for managing both atrial and ventricular arrhythmias.3,4
Dental Implications for Patients Medicated with Antiarrhythmic Drugs
Epinephrine, anticholinergics, and any other drug that has excitatory influences on the heart should be used with caution in patients who present with a history of any cardiac arrhythmia. Careful monitoring of blood pressure and heart rate should be performed before and after any of these medications are administered. Continuous electrocardiographic monitoring is encouraged, especially when moderate or deep sedation is performed.
Class III antiarrhythmics may prolong the QT interval triggering Torsades de pointe, a lethal ventricular tachycardia. This potential is enhanced by macrolide antibiotics such as erythromycin. Amiodarone (Cordarone) use has risen in recent years and is also implicated in adverse interactions with two additional drugs frequently used in dentistry—lidocaine and fentanyl.2
Amiodarone inhibits lidocaine metabolism, which increases lidocaine serum levels. Local anesthetic toxicity has occurred during surgical procedures using this anesthetic. It would be wise to reduce maximum doses of any amide local anesthetic to half those normally permitted when treating patients medicated with amiodarone. Articaine would appear to be a safer alternative due to rapid hydrolysis of its ester side chain.
Profound bradycardia, sinus arrest, and hypotension have occurred during cardiac anesthesia using fentanyl. The bradycardia is usually unresponsive to atropine, and large doses of vasopressors have been required. Although these cases have occurred when using doses much higher than those used conventionally for moderate to deep sedation, caution is certainly advised.
DIURETICS
Diuretics induce the loss of water and solutes (diuresis). They may be categorized into three groups; thiazide, loop, and potassium-sparing diuretics (Table 5). All are regarded as natriuretic, in that they inhibit reabsorption of sodium. According to simple principles of osmosis, water accompanies the loss of sodium. Diuresis is also accompanied by excretion of other electrolytes, and this may result in various imbalances, most notably hypokalemia. This problem can be reduced by using one of the so-called “potassium-sparing” diuretics that inhibit the excretion of potassium. These can be used alone, or in combination with traditional thiazide diuretics, when the risk for hypokalemia is significant.
Diuretics are essential for correcting the sodium and water retention consistently associated with congestive heart failure. They are also useful as antihypertensive agents, but their risk-benefit ratio for this purpose is controversial. Electrolyte imbalances, hyperglycemia, and elevated cholesterol are side effects that may outweigh any potential benefit. Their routine use appears justified when managing hypertension within certain defined groups, eg patients who are elderly, obese, or of African-American descent.3 Generally, lower dosages can be used when managing hypertension than for congestive heart failure.
Thiazide derivatives are the most widely prescribed diuretics and are represented by hydrochlorothiazide (HCTZ). Loop diuretics have the greatest efficacy among agents currently available and are therefore referred to as “high ceiling” diuretics. Furosemide (Lasix) is the prototype of these agents and is the standard for managing severe cases of congestive heart failure.
Dental Implications for Patients Medicated with Diuretics
The use of NSAIDs for more than 5 days may diminish the efficacy of diuretics used to manage hypertension.2 Although the dosages of most medications should not be altered prior to dental care, diuretics present an exception, especially loop diuretics such as furosemide. When lengthy appointments under intravenous sedation are planned, it is permissible to withhold the morning dose of diuretic until the appointment is completed. This will not diminish control of the medical condition and will obviate the need to interrupt treatment for micturition.
VASODILATORS
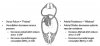
Vasodilators lower blood pressure, improve the patency of coronary arteries and reduce the work of the heart. They are vital for the management of hypertension, CHF, and coronary artery disease (CAD). Their ability to reduce venous return and arterial resistance is an obvious benefit when managing hypertension, but the manner in which vasodilation assists patients with coronary disease and heart failure is more complex. In these cases, vasodilators are described as “unloading” the heart. This concept is illustrated in Figure 1.
Vasodilators act by many ingenious mechanisms, and these offer a basis for subclassification, eg, alpha blockers, calcium channel blockers. Regardless of their precise mechanism, all vasodilators have several side effects in common. These include facial flushing, headaches, dizziness, and postural hypotension. Clinical experience has established the relative preference of each subclass for specific indications. Commonly prescribed agents and their typical indications are summarized in Table 6.
The nitrates differ in their selectivity for veins and arteries. Nitroglycerin is primarily a venous dilator, lowering venous pressure and therefore venous return (preload). It is frequently used to lower myocardial oxygen consumption in patients with symptoms of coronary artery disease. In contrast, sodium nitroprusside is an arterial dilator that reduces afterload by lowering the resistance against which the ventricles must eject. It is particularly attractive for managing acute episodes of congestive heart failure.
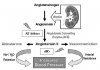
Drugs that interrupt the renin-angiotensin pathway are the most commonly prescribed vasodilators. Angiotensin II is an endogenous vasoconstrictor that also promotes release of aldosterone, leading to sodium and water retention (Figure 2). The angiotensin-converting enzyme (ACE) inhibitors block the formation of angiotensin II and thereby promote vasodilation and limit excessive retention of sodium and water. Enthusiasm for this drug class is understandable considering the role of arterial resistance and sodium retention in the pathogenesis of essential hypertension and congestive heart failure.1,3,7 One is cautioned not to regard ACE inhibitors as diuretics; they do not promote diuresis. Rather, they limit excessive sodium and water retention. Indeed, diuretics are often prescribed in combination with an ACE inhibitor.
Dental Implications for Patients Medicated with Vasodilators
Any vasodilator has the potential for producing postural hypotension, and this may be enhanced when CNS depressants are administered for moderate to deep sedation. This is especially true with opioids and agents having alpha-blocking actions such as promethazine and droperidol. Following dental treatment, always proceed cautiously when preparing the patient for discharge.
The ACE inhibitors and angiotensin-receptor blockers (ARBs) have been implicated in producing severe episodes of hypotension during general anesthesia, and some anesthesiologists prefer that patients withhold these particular medications. It has not been suggested that these agents be withheld prior to moderate or deep sedation, but blood pressure should be monitored closely, especially when propofol is being used.
The most common and troubling side effect of ACE inhibitors is a nagging cough. This is most likely related to elevated levels of bradykinin or other inflammatory mediators that are normally metabolized byACE.8 The same mechanism presumably explains reported cases of angioedema and perioral swelling that may be confused with similar signs attributed to odontogenic infections. The ARBs are used alternatively in such cases and provide comparable efficacy, minus the troubling side effects associated with reduced activity of angiotensin-converting enzyme.
The use of NSAIDs for more than 5 days may diminish the efficacy of ACE inhibitors and ARBs when used to manage hypertension.2
ABOUT THE AUTHOR
Dr. Becker is a Professor of Allied Health Sciences at Sinclair Community College and Associate Director of Education, General Dental Practice Residency, at Miami Valley Hospital in Dayton, Ohio.
REFERENCES
1. Rocco TP, Fang JC. Pharmacotherapy of congestive heart failure. In: Brunton LL, Lazo JS, Parker KL, eds. Goodman and Gilman’s The Pharmacological Basis of Therapeutics. 11th ed. New York: McGraw-Hill, 2006.
2. Olin BR, Hebel SK, Dombek CE, eds. Drug Interaction Facts. St Louis, MO: Facts and Comparisons Inc; 2007.
3. Opie LH, ed. Drugs for the Heart. 4th ed. Philadelphia, PA: WB Saunders Co; 1997.
4. Olin BR, Hebel SK, Dombek CE, eds. Drug Facts and Comparisons, 2007. St Louis, MO: Facts and Comparisons Inc; 2007.
5. Becker DE, Reed KL. Essentials of local anesthetic pharmacology. Anesth Prog. 2006;53(3):98-108.
6. Westbrook P, Bednarczyk EM, Carlson M, et al. Regression of nifedipine-induced gingival enlargement following switch to a same class calcium channel blocker, isradipine. J Periodontol. 1997;68(7):645-650.
7. Braunwald E. ACE inhibitors—A cornerstone of the treatment of heart failure. N Engl J Med. 1991;325(5):351-353.
8. Levey BA. Angiotensin-converting enzyme inhibitors and cough. Chest. 1990;98(5):1052-1053.