You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Obstructive sleep apnea (OSA) is a life-threatening condition that affects an estimated 26% of adults in the United States aged 30 through 70 years,1 an estimate that has risen significantly over the past decade and is likely still low. OSA is characterized by recurrent episodes of upper airway obstruction that result in interrupted breathing during sleep. Such obstructions often result in sleep fragmentation and may also lead to blood oxygen desaturations. Left untreated, OSA can be associated with long-term health risks, including hypertension, stroke, heart failure, diabetes, and depression.2 Although doctors have been diagnosing OSA for decades, it is estimated that only 10% to 20% of patients are diagnosed and appropriately treated.3
Dentists can play a critical role in the identification and management of OSA. Dentists are often the first healthcare professionals to recognize symptoms or signs related to sleep-disordered breathing, because as they are examining the teeth and mouth they are also looking into the oropharyngeal airway; moreover, patients often visit their dentist more frequently than a doctor. Dentists have an important role in the interdisciplinary care of patients with OSA and are the only healthcare providers qualified to provide oral appliance therapy.4
Definition of Obstructive Sleep Apnea
OSA is a disorder characterized by repeated pauses in breathing during sleep due to partial or complete obstruction of the airway. These breathing pauses can last from several seconds to minutes at a time. The cessation in airflow often causes significant drops in blood oxygen saturation levels, which can place strain on other organs such as the heart and brain. Untreated OSA can have serious long-term consequences, including an increased risk for hypertension, stroke, diabetes, depression, cardiovascular disease, and even death.2
Epidemiology and Pathophysiology
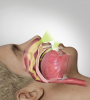
As mentioned earlier, OSA is one of the most common types of sleep disorders, affecting more than one-fourth of adults aged 30 through 70 years in the United States alone. While more prevalent among men than women (29% versus 19%),1 OSA can also affect certain racial/ethnic groups more commonly than others; African Americans have been shown to have higher rates compared to Caucasians or Hispanics.5 Risk factors associated with OSA include male sex, age >35 years, body mass index (BMI) measurement >25 kg/m2, large tonsils or adenoids, alcoholism, smoking, and hypertension.6 All of these risk factors contribute to upper airway narrowing or closure during sleep, which creates obstructions to breathing throughout sleep (Figure 1).
An apnea is technically defined as a cessation of airflow for 10 seconds or more. A hypopnea is a reduction of airflow of at least 50% with a desaturation of 4% or more.7 The apnea hypopnea index (AHI) has been used for decades as the measure of severity of OSA, although some clinicians and researchers now question if this is the correct metric, as the oxygen desaturation index and even the effects of daytime sleepiness may have a greater impact on overall health and safety.8 Currently, mild OSA is defined as having an AHI of 5 to <15; moderate OSA, an AHI of 15 to <30; and severe OSA, an AHI of ≥30.9 OSA episodes can lead to fragmented sleep resulting in daytime fatigue and impairments in cognitive functioning, which may possibly be due in part to related oxygenation desaturations during sleep.6
The Dentist's Role in Evaluating and Diagnosing OSA
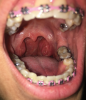
Dentists possess unique skillsets that make them well-suited to screen and evaluate their patients for potential sleep-related breathing disorders.4 Through their training in evaluating facial anatomy, bite relationships, and soft-tissue structures, dentists are able to identify physical signs indicative of an increased risk for developing OSA such as large tonsils (Figure 2), narrowed upper airways, retrognathia, a narrow maxillary arch, and high arched palates. In addition to physical examination findings, dentists commonly order imaging tests such as panoramic radiographs or cone-beam computed tomography scans, which assess head and neck hard and soft tissues and could be used to indicate increased OSA risk.10
Clinical Presentation
The classic signs and symptoms associated with OSA include snoring, often loud, followed by periods where breathing stops for 10 seconds or longer, which is often accompanied by gasping sounds on resumption thereof. Symptoms also include morning headaches, dry mouth upon awakening, and excessive daytime fatigue that interferes with daily activities such as work or school performance. In addition, other common symptoms reported include night sweats, insomnia, restlessness, irritability, poor concentration, decreased libido, erectile dysfunction, depression, difficulty staying asleep, frequent nighttime urination, awaking feeling unrefreshed, memory problems, and difficulty falling asleep again after waking up during nighttime hours. All of these symptoms may contribute to an impaired quality of life due to chronic drowsiness during daytime activities, often leading to further complications such as motor-vehicle or work-related accidents.11
To aid in evaluating patients, dentists can use validated screening questionnaires in addition to taking the patient's history and giving a physical examination. Questionnaires such as the Epworth Sleepiness Scale and the STOP-Bang are easy to use and are commonly utilized by medical professionals as well.12,13 Sometimes, however, these screening forms are less likely to detect potential sleep-related breathing disorders in healthier, younger individuals, and particularly women, as the questionnaires are biased toward older, heavier men since this group is at greatest risk.14
Diagnosis
Dentists should work with their medical colleagues to properly diagnose patients with suspected sleep-related breathing disorders. Dentists are encouraged to refer patients to physicians for additional evaluation and to determine if a sleep study is indicated.4 An overnight polysomnogram (PSG) is considered the "gold standard" for the diagnosis of OSA and may provide the dentist with extremely useful information regarding bruxism muscle activity as well as amount of time spent in various sleep stages and how sleeping position and REM (rapid eye movement) sleep affect the patient's OSA.15
So-called home sleep tests (HSTs) have become very com‑mon because they are far less expensive than a PSG. Many US insurance companies require that a patient "fail" an HST before they will authorize a PSG. HSTs can only rule in OSA; they cannot rule it out. It is, therefore, important for dentists to review what type of sleep study patients have received, particularly if they've been told they do not have sleep apnea.16 The data provided by HSTs varies dramatically by the device used. Some are capable of measuring brain waves, while most are not, and as such they cannot actually measure sleep or sleep stages accurately.17 Once a patient has had a PSG or HST, a board-certified sleep physician will read and interpret it and the physician who ordered the study will make a diagnosis.
OSA Treatment Options and Dentistry
Continuous positive airway pressure (CPAP) is the most commonly used treatment for OSA today due to its proven efficacy. However, its long-term clinical efficacy recently has been called into question, as published evidence mostly does not support that a CPAP prescription affects long-term, clinically important outcomes, such as all-cause mortality, various cardiovascular outcomes, and clinically important changes in psychosocial measures, to name a few.18 During sleeping, CPAP uses positive air pressure (regular room air) from a machine connected to a mask worn over the patient's nose, or nose and mouth, to keep the collapsible portion of the patient's upper airway open throughout sleep, acting as a pneumatic splint. Studies have shown that the use of CPAP results in improved outcomes in the treatment of many associated conditions and improves numerous areas of health and wellness.12 Unfortunately, a relatively high percentage of patients abandon the use of CPAP within the first year.19 This lack of compliance is multifactorial, but reasons for it include but are not limited to: patients not fully understanding their disease; durable medical equipment providers inadequately assisting the patient in trying different masks; the patient not feeling significantly better; and bed partner complaints related to the noise from the machine itself or the mask leaking.
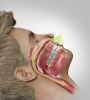
Oral appliance therapy (OAT) involves the use of an oral appliance in an attempt to lower the collapsibility of the oropharyngeal airway while the patient sleeps. Mandibular advancement devices (MADs) are typically custom-made mouthpieces fabricated primarily out of plastic, acrylic, or nylon that fit snugly on the patient's teeth. They anchor the lower jaw to the fixed upper jaw, which results in limiting the retrusive movement of the lower jaw and/or bringing the lower jaw forward with some type of advancement mechanism (Figure 3 and Figure 4).20 Several studies have indicated good success rates using MADs with results ranging from moderate reduction to complete elimination of OSA depending on the severity level being treated.20-22 The American Academy of Sleep Medicine (AASM) has recommended OAT since 2005 as a treatment for mild to moderate OSA, and in 2015 the AASM in conjunction with the American Academy of Dental Sleep Medicine updated guidelines to support the use of oral appliance therapy for any level of OSA, if medically indicated.23,24
While all MADs are designed to limit retrusive movement of the mandible and provide protrusion of the mandible when needed, various basic mechanisms are employed to accomplish this mandibular anchorage and advancement (Figure 5):
Anterior pull/push: This design typically has an adjustment mechanism in the anterior of the appliance that will "pull" the mandible forward to the point that the occlusion is end to end, and then "push" the lower jaw forward of that point as needed and depending on the level of adjustability of the mechanism. Commercially available examples of this design are the TAP® (Thornton adjustable positioner) (Airway Management, tapintosleep.com), MDSA® (medical dental sleep appliance) (ChairsideSplintStore.com [US distributor]), and Silencer® (The Silencer, the-silencer.com) appliances, among others.
Bilateral pull: The design of this type of MAD usually has bands made from various materials that connect from the canine/first premolar area on the upper jaw to the first molar area on the lower jaw. Bands of different lengths can be used to achieve different amounts of protrusion. Among the commercially available examples of this design are the Silent Nite® and EMA® appliances (available from multiple laboratories).
Bilateral push: Dentists will recognize the look of this design due to its use of Herbst hardware, which has been used in orthodontics for decades. The Herbst bars are placed from the maxillary molar area to the mandibular premolar/canine area and can be adjusted to telescope the bar to provide greater protrusion. There are many commercially available appliances that use this Herbst hardware or have their own variation of Herbst hardware.
Interlocking: Interlocking designs involve some form of "fins" (eg, dorsal fin) on the maxillary and mandibular portions of the appliance that interact in a way that reduces or eliminates posterior movement of the mandible and uses a mechanism-often an expansion screw or system of multiple trays-to allow for greater protrusion. Commercially available examples of this design include but are not limited to the ProSomnus EVO™ (ProSomnus Sleep Technologies, prosomnus.com), SomnoDent® (SomnoMed, somnomed.com), Dorsal® (DynaFlex, dynaflex.com), and WholeYou™ Respire Blue (Respire Medical, wholeyou.com) appliances.
Adjustable monoblock: Monoblock appliances stabilize the mandibular position by rigidly securing the upper and lower jaws, usually restricting movement of the lower jaw in any direction while the appliance is worn. Most modern monoblock appliances include a mechanism for increasing protrusion of the mandible. Commercially available examples of adjustable monoblocks include the Klearway™ (UBC Sleep Apnea Dental Clinic, klearway.com) and Moses® (Moses Appliances LLC, themoses.com) appliances, among others.21
Treatment of obstructive sleep apnea, whether through the use of CPAP or OAT, is often a lifelong therapy as OSA tends to worsen with age. Dentists who provide OAT should offer regular follow-up for their patients.24 It is also important to note that appliances used to protect a patient's teeth from bruxism activity have been shown to potentially increase OSA.25 Therefore, dentists are encouraged to screen their patients for possible sleep-related breathing disorders prior to fabrication of a nightguard.
Conclusion
Dentists play a critical role in the screening, evaluation, diagnosis, and treatment of obstructive sleep apnea as part of a multidisciplinary team. Most people who have OSA have yet to be diagnosed or treated. The public health, social, and financial costs of untreated OSA are substantial, and the impact dentists can have on the health and lives of their patients and patients' families cannot be overstated.
About the Author
Jamison R. Spencer, DMD, MS
Adjunct Faculty, University of Utah School of Dentistry, Salt Lake City, Utah;
Private Practice, Taylorsville, Utah
Queries to the author regarding this course may be submitted to authorqueries@broadcastmed.com.
References
1. Peppard PE, Young T, Barnet JH, et al. Increased prevalence of sleep-disordered breathing in adults. Am J Epidemiol. 2013;177(9):1006-1014.
2. Shahar E, Whitney CW, Redline S, et al. Sleep-disordered breathing and cardiovascular disease: cross-sectional results of the Sleep Heart Health Study. Am J Respir Crit Care Med. 2001;163(1):19-25.
3. American Academy of Sleep Medicine. Hidden Health Crisis Costing America Billions. Mountain View, CA: Frost & Sullivan; 2016. https://aasm.org/resources/pdf/sleep-apnea-economic-crisis.pdf. Accessed March 28, 2023.
4. Addy N, Bennett K, Blanton A, et al; American Academy of Dental Sleep Medicine Board of Directors. Policy statement on a dentist's role in treating sleep-related breathing disorders. J Dent Sleep Med. 2018;5(1):25-26.
5. Dudley KA, Patel SR. Disparities and genetic risk factors in obstructive sleep apnea. Sleep Med. 2016;18:96-102.
6. Mitra AK, Bhuiyan AR, Jones EA. Association and risk factors for obstructive sleep apnea and cardiovascular diseases: a systematic review. Diseases. 2021;9(4):88.
7. Mbata G, Chukwuka J. Obstructive sleep apnea hypopnea syndrome. Ann Med Health Sci Res. 2012;2(1):74-77.
8. Won CHJ, Qin L, Selim B, Yaggi HK. Varying hypopnea definitions affect obstructive sleep apnea severity classification and association with cardiovascular disease. J Clin Sleep Med. 2018;14(12):1987-1994.
9. American Academy of Sleep Medicine. Obstructive Sleep Apnea. AASM website. 2008. https://aasm.org/resources/factsheets/sleepapnea.pdf. Accessed March 28, 2023.
10. Enciso R, Shigeta Y, Nguyen M, Clark GT. Comparison of cone-beam computed tomography incidental findings between patients with moderate/severe obstructive sleep apnea and mild obstructive sleep apnea/healthy patients. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;114(3):373-381.
11. Obstructive sleep apnea in adults: epidemiology, clinical presentation, and treatment options. Adv Cardiol. 2011;46:1-42.
12. Labarca G, Saavedra D, Dreyse J, et al. Efficacy of CPAP for improvements in sleepiness, cognition, mood, and quality of life in elderly patients with OSA: systematic review and meta-analysis of randomized controlled trials. Chest. 2020;158(2):751-764.
13. Corso RM, Petrini F, Buccioli M, et al. Clinical utility of preoperative screening with STOP-Bang questionnaire in elective surgery. Minerva Anestesiol. 2014;80(8):877-884.
14. Dosman JA, Karunanayake CP, Fenton M, et al. STOP-Bang score and prediction of severity of obstructive sleep apnea in a First Nation community in Saskatchewan, Canada. Clocks Sleep. 2022;4(4):535-548.
15. Alzoubaidi M, Mokhlesi B. Obstructive sleep apnea during rapid eye movement sleep: clinical relevance and therapeutic implications. Curr Opin Pulm Med. 2016;22(6):545-554.
16. Collop N. Home sleep testing: appropriate screening is the key. Sleep. 2012;35(11):1445-1446.
17. Hung CJ, Kang BH, Lin YS, Su HH. Comparison of a home sleep test with in-laboratory polysomnography in the diagnosis of obstructive sleep apnea syndrome. J Chin Med Assoc. 2022;85(7):788-792.
18. Agency for Healthcare Research and Quality. Continuous Positive Airway Pressure Treatment for Obstructive Sleep Apnea. AHRQ website. https://www.ahrq.gov/sites/default/files/wysiwyg/research/findings/ta/drafts-for-review/sleep-apnea-draftreport.pdf. Accessed March 28, 2023.
19. Wolkove N, Baltzan M, Kamel H, et al. Long-term compliance with continuous positive airway pressure in patients with obstructive sleep apnea. Can Respir J. 2008;15(7):365-369.
20. Jayesh SR, Bhat WM. Mandibular advancement device for obstructive sleep apnea: an overview. J Pharm Bioallied Sci. 2015;7(suppl 1):
S223-S225.
21. Essick GK, Blank AR, Spencer JR, Parker JA. Part III: Oral appliance therapy for sleep-disordered breathing. CDEWorld website. September 2016. https://cdeworld.com/courses/5023-part-iii-oral-appliance-therapy-for-sleep-disordered-breathing. Accessed March 28, 2023.
22. Verburg FE, Bollen KHA, Donker HJ, Kramer GJC. The effectiveness of two types of MADS for OSA therapy. Clin Oral Investig. 2018;22(5):1995-2003.
23. Kushida CA, Morgenthaler TI, Littner MR, et al; American Academy of Sleep. Practice parameters for the treatment of snoring and obstructive sleep apnea with oral appliances: an update for 2005. Sleep. 2006;29(2):240-243.
24. Ramar K, Dort LC, Katz SG, et al. Clinical practice guideline for the treatment of obstructive sleep apnea and snoring with oral appliance therapy: an update for 2015. J Dent Sleep Med. 2015;2(3):71-125.
25. Gagnon Y, Mayer P, Morisson F, et al. Aggravation of respiratory disturbances by the use of an occlusal splint in apneic patients: a pilot study. Int J Prosthodont. 2004;17(4):447-453.