You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Endodontists are tasked with the diagnosis of painful pulpal and periapical disease with the eventual goal of alleviating their patient's pain via dental treatment. Taken together, the information gathered from the diagnostic triad of patient history, clinical examination, and radiographic examination is usually sufficient to diagnose endodontic disease states. The patient history will often include reported pain to thermal stimuli, pain during mastication, intraoral or extraoral swelling, and/or drainage from a sinus tract. The clinical examination highlights both existing dental conditions and presumed etiologies of pulpal disease. This exam should also include pulp sensitivity testing and periapical testing, which is meant to replicate symptoms.1 Pulp sensitivity testing includes thermal testing to cold and heat and sometimes electric pulp testing, when indicated. Periapical testing should include percussion, biting, and palpation testing.
Patients experiencing pulpal disease, like pulpitis or necrosis, or periapical disease, such as apical periodontitis or apical abscess, frequently present with symptoms that are easily recognizable and replicable by the diagnosing clinician.2 These patients often present with well-localized pain replicated by cold, heat, or pressure or associated soft-tissue swelling or drainage. The information gathered from the history and clinical exam is augmented by radiographic data. Two-dimensional radiography, including both bitewings and periapical radiographs, is the standard of care in endodontic diagnosis.1 These imaging modalities are limited in their ability to detect pathology due to issues associated with structural overlap and distortion.3 Newer technologies, such as cone-beam computed tomography (CBCT) imaging, offer greater accuracy and confirmation of endodontic pathology.4
Not all patients, however, fit neatly within this diagnostic paradigm. Pain that is difficult to localize is common for patients experiencing degenerative pulpal disease, which is often associated with an acutely symptomatic irreversible pulpitis. These patients frequently present with complaints of radiating pain, often poorly localized between the maxillary and mandibular arches. They may also report referral of pain to other orofacial structures like the ear, lateral neck, and temple.5 Upon clinical testing, exaggerated responses may be noted in multiple teeth, without replication of the patient's chief complaint. These patients often experience pain of such severity that they consume large doses of over-the-counter analgesics, including ibuprofen, to alleviate symptoms. These medications further complicate diagnosis, as they have a measurable negative impact on the accuracy of clinical testing results.6 Many of these patients often experience sleep disruption, which has been shown to further exacerbate the perception of pain.7 Certain patients may present with multiple possible etiologies for pain. Extensive dental restorations can limit the use of currently available pulp sensitivity testing devices and may, in fact, create artifacts rendering CBCT images nondiagnostic.4
In order to properly diagnose patients with diffuse and poorly replicable pain, the conscientious diagnostician must possess additional armamentaria when traditional means are insufficient to determine the source or, in some cases, sources of a patient's pain. Local anesthetics can be readily utilized for these patients to both provide immediate pain relief and serve as a diagnostic aid. The process by which local anesthetics are utilized to narrow down a source of pain is referred to as "selective anesthesia."1,8 Local anesthetics can be useful to both localize sources of pain by elimination of symptoms and rule out other potential sources of pain where the anesthesia fails to affect symptoms.9 This article reviews selective anesthesia techniques and presents cases where selective anesthesia facilitated challenging endodontic diagnoses.
Applying Selective Anesthesia
A convenient feature of pain of dental origin is its elimination by local anesthetics.10 The definitive diagnosis of pulpal and periapical disease necessitates the replication of a patient's chief complaint. In patients with poorly localizable pain associated with the inability to replicate symptoms, selective anesthesia can facilitate endodontic diagnosis by eliminating pain, confirming its source and dental origin. Furthermore, unlike pain of dental origin, several non-odontogenic pain sources are not eliminated by local anesthesia. These include myofascial pain, pain of sinus origin, and neuropathic pain.11-13 The use of selective anesthesia techniques can facilitate the differentiation between odontogenic and non-odontogenic pain sources.
The use of selective anesthesia requires a robust foundational understanding of maxillary and mandibular innervation. Maxillary teeth receive their pulpal neural afferent supply from the second branch of the trigeminal nerve. The maxillary nerve provides sensory innervation to the molars via the posterior superior alveolar nerve, to the mesiobuccal root of the first molar and the premolars via the middle superior alveolar nerve, and to the anterior teeth via the anterior superior alveolar nerve.14 This innervation extends solely from the buccal aspect of these teeth. Mandibular teeth receive their pulpal neural afferent supply from the third branch of the trigeminal nerve. The inferior alveolar nerve provides sensation to the mandibular molar and premolar teeth. As it courses anteriorly, it splits, with one portion exiting the mandible via the mental foramen and the other continuing anteriorly as the incisive nerve to provide sensation to the mandibular anterior teeth.14 The mandibular anterior teeth may have additional sensation via cross-innervation from the opposite side.15
To be most effective, selective anesthesia should begin with the use of "narrow field" anesthesia techniques capable of producing pulpal anesthesia. Although intraligamentary (PDL) injections have been suggested to differentiate among painful conditions in adjacent teeth, some authors discourage their use for selective anesthesia because they are shown to anesthetize neighboring teeth with regularity.16 When aiming to determine if a pain source is maxillary or mandibular in origin, the maxilla is generally anesthetized first utilizing local infiltration. Complete pulpal anesthesia of maxillary teeth is achievable via local infiltration techniques administered only on the buccal aspects. Administration of local anesthetics on the palatal aspect of a maxillary tooth does not enhance pulpal anesthesia.17 The narrow scope technique of selective anesthesia should begin with anesthesia in the mesial-most suspected location, working distally as needed to broaden the scope of anesthetized tissues. The choice of anesthetic appears unimportant, as both 2% lidocaine and 4% articaine are similarly capable of achieving pulpal anesthesia in the maxilla.18 Therefore, for the practice of selective anesthesia, clinicians can feel comfortable using the anesthetics that are available to them.
Confirmation that pulpal anesthesia has been achieved in the maxillary teeth must be attained prior to the clinician moving to mandibular anesthesia. Soft-tissue signs in the adjacent tissues, namely the lack of sensation to pinprick in the soft tissue in the shared dermatome of anesthesia, can suggest that the adjacent teeth have obtained pulpal anesthesia. That said, pulpal anesthesia can be best confirmed by the absence of a cold response in a previously cold-responsive vital tooth.19 Taken together, the observation of soft-tissue signs of local anesthesia as well as the absence of a cold response in tested teeth provides assurance of adequate pulpal anesthesia.
After ruling out a maxillary source of discomfort, mandibular anesthesia should be employed. The same strategy of moving mesially to distally should be used, with administration of block anesthesia as the last line in the anesthesia in posterior mandibular teeth. Given the thin cortical plate present in the mandibular anterior region, local infiltration anesthesia is usually sufficient for pulpal anesthesia in this area.20 Mandibular premolars may obtain pulpal anesthesia by an incisive nerve block at the mental foramen, wherein the mental foramen provides a portal of entry for local anesthesia to the main branch of the mandibular nerve.21 Local infiltration is considered inadequate for pulpal anesthesia in the mandibular posterior due to the relative thickness of cortical bone prohibiting adequate concentrations of anesthetic to penetrate the deeply seated nerve.22 Therefore, mandibular molars will require block anesthesia targeting the mandibular nerve, via inferior alveolar nerve block or Gow-Gates or Vazirani-Akinosi techniques, with frequent need for supplemental techniques to obtain adequate pulpal anesthesia in this region.23 Like in the maxilla, the choice of anesthetic itself has little impact on achieving predictable success. In the mandible, anesthetics perform similarly; namely, 3% mepivacaine, 2% lidocaine, 4% prilocaine, and 0.5% bupivacaine are equally as effective at achieving anesthesia.24,25
The success rates of inferior alveolar nerve blocks in the posterior mandible are surprisingly low, however. One study utilizing 1.8 mL of 2% lidocaine with 1:100,000 epinephrine administered via inferior alveolar nerve block found that the success rate of pulpal anesthesia in the mandibular first molar was only 51%.26 Consequently, adjunctive techniques are often required. The addition of buccal infiltration utilizing 4% articaine with 1:100,000 epinephrine has been shown to be the most reliable and a safe means of increasing successful pulpal anesthesia, with success rates of 84% when compared to alternative methods like intraosseous anesthesia, PDL injections, and repeat inferior alveolar nerve block.18,27
Figure 1 highlights the sequence a clinician should follow when utilizing selective anesthesia techniques. The disappearance of symptoms following successful anesthesia confirms the dental origin of symptoms and will allow clinicians to localize the maxillary or mandibular origin of pain. Similarly, when a non-odontogenic source of pain is suspected, selective anesthesia is an important tool to rule out an odontogenic source. That said, anesthetic failures should always be ruled out to ensure that the failure of selective anesthesia is not secondary to unsuccessful methodologies or local inflammatory changes.28
The following two cases presentations illustrate the utility of selective anesthesia in endodontic diagnosis.
Case Presentations
Case 1
A 75-year-old man presented with the chief complaint of severe, poorly localized pain in his right jaw and face beginning 1 week prior to presentation. He described pain as throbbing and indicated that it extended from the midline to his right ear in both the maxilla and mandible. The patient reported that the application of heat exacerbated his symptoms and that he could relieve pain by holding cold water in his mouth. Little relief was noted with the combination of 600 mg ibuprofen plus 500 mg acetaminophen taken orally.
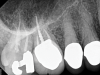
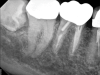
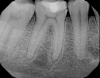
Although the patient's chief complaint was consistent with a degenerative pulpitis, the clinical and radiographic examination was initially unremarkable. All teeth in the maxillary and mandibular right quadrants were normally responsive to pulp sensitivity testing, and they were neither percussion nor palpation sensitive. Neither swelling nor sinus tracts were present, and the periodontal examination revealed normal probing depths and lack of mobility throughout the quadrant. Initial periapical radiographs revealed normal PDL spaces surrounding teeth Nos. 30 and 31, as well as a horizontally impacted No. 32 with a radiolucency within the crown in close proximity to the distal root surface of No. 31 (Figure 2 and Figure 3). Because the reported pain was severe, and in an effort to not only diagnose the issue but also offer some respite from the severe pain the patient was experiencing, an inferior alveolar nerve block was administered using 3.4 cc 0.5% bupivacaine with 1:200,000 epinephrine. Within 5 minutes of administration, the patient reported complete resolution of his pain.
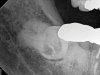
Given the confirmation of a likely odontogenic source of pathology since pain was relieved by the block anesthesia, secondary periapical imaging was taken, which showed intracoronal resorption in the unerupted tooth No. 32 (Figure 4). Because of the visualizable pathology and signs and symptoms consistent with a symptomatic irreversible pulpitis, the patient was referred to an oral and maxillofacial surgeon for extraction of tooth No. 32. The patient reported complete resolution of symptoms following extraction of the affected tooth. Communication from the oral surgeon confirmed the diagnosis of intracoronal resorption on tooth No. 32.
Case 2
A 35-year-old man presented for evaluation of severe right-sided jaw and facial pain. He reported poorly localized, intense pain of several days' duration. His initial symptoms seemed localized to the lower jaw, although over the previous 24 hours the symptoms, he said, seemed more closely associated with the maxilla. He reported that waves of pain kept him from sleeping, and he experienced no relief from 220 mg naproxen. His dental history was relevant for deep restorations completed on teeth Nos. 2 and 30 within the past 10 years.
On clinical examination, marginal leakage was noted around the large composite restoration on the occlusal surface of tooth No. 2. Recurrent caries was noted on the buccal margin of the bucco-occlusal composite restoration on tooth No. 30. Tooth No. 1 was not visible within the oral cavity. Teeth Nos. 2 and 30 were both nonresponsive to pulp sensitivity testing, whereas neighboring control teeth responded normally to cold and electric pulp testing. All posterior teeth in the maxillary and mandibular right quadrants were normally responsive to percussion and palpation testing. No swellings, sinus tracts, or periodontal defects were noted.
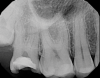
Periapical imaging revealed evidence of a prior pulpotomy on tooth No. 2, without obvious signs of apical pathology (Figure 5). A deep restoration with evidence of periapical pathology was seen on periapical imaging of tooth No. 30 (Figure 6). The diagnosis for tooth No. 2 was previously initiated treatment with normal periapical tissues, while the diagnosis for tooth No. 30 was pulpal necrosis with symptomatic apical periodontitis. Although it was clear that both teeth would eventually require definitive endodontic treatment or extraction, the source of the current severe pain remained unclear.
Selective anesthesia was utilized in order to differentiate the source of the pain to facilitate clinical decision making and triage emergency care. Initial administration of 1.7 cc 4% septocaine with 1:100,000 epinephrine via buccal infiltration on tooth No. 2 resulted in no reduction in pain. However, after 10 minutes, symptoms began to localize to the lower right quadrant. Further administration of 1.7 cc 2% xylocaine with 1:100,000 epinephrine via inferior alveolar nerve block resulted in complete resolution of pain. Given the absence of pain following mandibular anesthesia, tooth No. 30 was clearly the current source of pain and required immediate treatment. A pulpectomy was completed on tooth No. 30 to alleviate the patient's symptoms. Follow-up the next day confirmed resolution of all symptoms.
Conclusion
The use of selective anesthesia should never be the only means of making an appropriate diagnosis. Generally, first-line diagnostics, including a thorough patient history, in-depth clinical examination involving the use of pulp sensitivity testing, and comprehensive radiographic examination involving both 2-dimensional and 3-dimensional methods for detection of pathology will suffice to detect pulpal or periapical pathology. However, in cases where the examination is not definitive yet the suspicion for pathology remains high, selective anesthesia can be a useful diagnostic tool.
About the Authors
Brooke Blicher, DMD, Certificate in Endodontics
Assistant Clinical Professor, Department of Endodontics, Tufts University School of Dental Medicine, Boston, Massachusetts; Clinical Instructor, Department of Restorative Dentistry and Biomaterials Science, Harvard School of Dental Medicine, Boston, Massachusetts; Instructor in Surgery, Dartmouth Medical School, Hanover, New Hampshire; Private Practice limited to Endodontics, White River Junction, Vermont
Rebekah Lucier Pryles, DMD, Certificate in Endodontics
Assistant Clinical Professor, Department of Endodontics, Tufts University School of Dental Medicine, Boston, Massachusetts; Clinical Instructor, Department of Restorative Dentistry and Biomaterials Science, Harvard School of Dental Medicine, Boston, Massachusetts; Private Practice limited to Endodontics, White River Junction, Vermont
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Berman LH, Hartwell GR. Diagnosis. In: Hargreaves KM, Cohen S, Berman LH, eds. Cohen's Pathways of the Pulp. 10th ed. St Louis, MO: Mosby; 2011:2-39.
2. American Association of Endodontists. Glossary of Endodontic Terms. 10th ed. Chicago, IL: American Association of Endodontists; 2020.
3. Patel S, Dawood A, Mannocci F, et al. Detection of periapical bone defects in human jaws using cone beam computed tomography and intraoral radiography. Int Endod J. 2009;42(6):507-515.
4. Patel S, Durack C, Abella F, et al. Cone beam computed tomography in endodontics - a review. Int Endod J. 2015;48(1):3-15.
5. Bender IB. Pulpal pain diagnosis - a review. J Endod. 2000;26(3):175-179.
6. Read JK, McClanahan SB, Khan AA, et al. Effect of ibuprofen on masking endodontic diagnosis. J Endod. 2014;40(8):1058-1062.
7. Krause AJ, Prather AA, Wager TD, et al. The pain of sleep loss: a brain characterization in humans. J Neurosci. 2019;39(12):2291-2300.
8. Littner MM, Tamse A, Kaffe I. A new technique of selective anesthesia for diagnosing acute pulpitis in the mandible. J Endod. 1983;9(3):116-119.
9. Blicher B, Walton RD, Torabinejad M. Endodontic diagnosis and treatment planning. In: Torabinejad M, Fouad AF, Shabahang S, eds. Endodontics: Principles and Practice. 6th ed. St. Louis, MO: Elsevier; 2020:61-82.
10. Malamed SF, ed. Handbook of Local Anesthesia. 6th ed. St. Louis, MO: Mosby; 2012.
11. Friction JR, Kroening R, Haley D, Siegert R. Myofascial pain syndrome of the head and neck: a review of clinical characteristics of 164 patients. Oral Surg Oral Med Oral Pathol. 1985;60(6):615-623.
12. Mattscheck D, Law A, Nixdorf D. Diagnosis of nonodontogenic toothache. In: Hargreaves KM, Cohen S, Berman LH, eds. Cohen's Pathways of the Pulp.10th ed. St. Louis, MO: Mosby; 2011:49-70.
13. Zakrzewska JM. Diagnosis and differential diagnosis of trigeminal neuralgia. Clin J Pain. 2002;18(1):14-21.
14. Fehrenbach MJ, Herring SW. Illustrated Anatomy of the Head and Neck. 4th ed. St. Louis, MO: Elsevier/Saunders; 2012.
15. Pogrel MA, Smith R, Ahani R. Innervation of the mandibular incisors by the mental nerve. J Oral Maxillofac Surg. 1997;55(9):961-963.
16. White JJ, Reader A, Beck M, Meyers WJ. The periodontal ligament injection: a comparison of the efficacy in human maxillary and mandibular teeth. J Endod. 1988;14(10):508-514.
17. Guglielmo A, Drum M, Reader A, Nusstein J. Anesthetic efficacy of a combination palatal and buccal infiltration of the maxillary first molar. J Endod. 2011;37(4):460-462.
18. Kanaa MD, Whitworth JM, Meechan JG. A prospective randomized trial of different supplementary local anesthetic techniques after failure of inferior alveolar nerve block in patients with irreversible pulpitis in mandibular teeth. J Endod. 2012;38(4):421-425.
19. Hsiao-Wu GW, Susarla SM, White RR. Use of the cold test as a measure of pulpal anesthesia during endodontic therapy: a randomized, blinded, placebo-controlled clinical trial. J Endod. 2007;33(4):406-410.
20. Dressman AS, Nusstein J, Drum M, Reader A. Anesthetic efficacy of a primary articaine infiltration and a repeat articaine infiltration in the incisive/mental nerve region of the mandibular premolars: a prospective, randomized, single-blind study. J Endod. 2013;39(3):313-318.
21. Nist RA, Reader A, Beck M, Meyers WJ. An evaluation of the incisive nerve block and combination inferior alveolar and incisive nerve blocks in mandibular anesthesia. J Endod.1992;18(9):455-459.
22. Currie CC, Meechan JG, Whitworth JM, Corbett IP. Is mandibular molar buccal infiltration a mental and incisive nerve block? A randomized controlled trial. J Endod. 2013;39(4):439-443.
23. Goldberg S, Reader A, Drum M, et al. Comparison of the anesthetic efficacy of the conventional inferior alveolar, Gow-Gates, and Vazirani-Akinosi techniques. J Endod. 2008;34(11):1306-1311.
24. McLean C, Reader A, Beck M, Meryers WJ. An evaluation of 4% prilocaine and 3% mepivacaine compared with 2% lidocaine (1:100,000 epinephrine) for inferior alveolar nerve block. J Endod. 1993;19(3):146-150.
25. Fernandez C, Reader A, Beck M, Nusstein J. A prospective, randomized, double-blind comparison of bupivacaine and lidocaine for inferior alveolar nerve blocks. J Endod. 2005;31(7):499-503.
26. Reader A, Nusstein J, Drum M. Successful Local Anesthesia for Restorative Dentistry and Endodontics. Hanover Park, IL: Quintessence Publishing; 2011.
27. Kung J, McDonagh M, Sedgley CM. Does articaine provide an advantage over lidocaine in patients with symptomatic irreversible pulpitis? A systematic review and meta-analysis. J Endod. 2015;41(11):1784-1794.
28. Hargreaves KM, Keiser K. Local anesthetic failure in endodontics: mechanisms and management. Endod Topics. 2002;1(1):26-39.