You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Disclosure: The authors had no disclosures to report.
The nasopalatine canal can present both difficulties and opportunities with respect to placement of dental implants in the anterior maxilla. The canal and its foramen are located in the midline of the palate where the maxilla abuts the premaxilla. The foramen is lingual to the central incisors (if they are present).1 The position of the nasopalatine foramen with respect to the teeth or the buccal plate of bone, the size of the foramen, and the type of planned prosthesis are factors to consider when treatment planning dental implant placement in this area. This article reviews issues related to implant insertion in the nasopalatine region for a fixed or removable dental prosthesis.
Anatomy: Nasopalatine Foramen and Canal
The nasopalatine canal connects the roof of the oral cavity (nasopalatine foramen) with the floor of the nasal cavity (nasal foramen). The nasopalatine foramen also is referred to as the incisive foramen.2 On flap reflection of the soft tissue overlying the foramen, two lateral canals are often observed. The paired incisive canals frequently fuse before exiting the foramen to form a shared canal in a “Y” shape.3 Occasionally, one to four canals may be present.2 The nasopalatine canal transmits the anterior branches of the descending palatine vessels and the nasopalatine nerve.4 The origin of the nasopalatine nerve is the maxillary division of the trigeminal nerve. It enters the posterior nasal cavity via the sphenopalatine foramen, traverses the roof of the nose, and proceeds along the nasal septum in between the periosteum and mucosa and enters the nasopalatine foramen. The incisive canal also contains fibrous connective tissue, fat, and small salivary glands.5
Mraiwa et al2 reported that the buccopalatal bone width of the anterior maxillary ridge varies from 2.9 mm to 13.6 mm and the nasopalatine foramen is located approximately 7.4 mm from the labial surface of an unresorbed maxillary anterior ridge. This distance varies depending on the initial size of an individual’s maxillary ridge and the amount of resorption that occurs after extractions.
The mean diameter of the foramen is around 4.6 mm (range, 1.5 mm to 9.2 mm).2 Others reported the average diameter was less: 2.59 mm,6 3.4 mm,5 and 3.61 mm.7 The length of the nasopalatine canal ranges from 3 mm to 14 mm (mean, 9 mm).8 Similarly, it was reported that the average height was 10.08 mm +/- 2.25 mm7 and 10.86 mm.6 With respect to a dentulous maxilla, the height varies from 8.4 mm to 15.8 mm (mean, 12 mm).9 Over time, resorption of the alveolar ridge results in the canal becoming shorter.6,10 Conflicting results regarding the height or width of the nasopalatine canal or width of the foramen could be attributed to different study populations, extent of resorption, or the imaging techniques used.
The angle of the nasopalatine canal varies from 46 to 99 degrees with respect to the horizontal plane. Its mean angulation is 66 degrees.7 The canal is usually slanted, but it can be vertical. Whether slanted or vertical, it may have a curved part at the nasal end.7 The outline of the canal is usually cylindrical6 but can be shaped like a funnel, hourglass, or spindle.7
The following should be noted with regard to surgical flap design in the premaxilla when an implant is not being placed in the nasopalatine canal: When performing surgery in the nasopalatine area, some clinicians create a crestal incision labially around the incisive papilla to avoid transecting contents of the nasopalatine canal.11 An incision through the canal region does not usually have a detrimental effect12; however, it may occasionally result in some numbness of the anterior palatal tissue.11
Management of Dental Implants Close to the Nasopalatine Canal
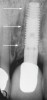
Several clinical approaches can be taken to replace a lost maxillary central incisor with a dental implant. An implant can be inserted to replace a tooth if there is sufficient bone height and width and the osteotomy can be created without encroaching on the nasopalatine canal. However, if the nasopalatine canal is close to the site of implant placement and it is desirable to avoid entering the canal, it may be possible to place the implant at an angle to avoid penetrating the canal (Figure 1). In this regard, if a misangulation of the implant is created intentionally, increased sink depth (distance from the implant platform to the gingival margin) will provide additional running room to facilitate creating a correct emergence profile for the restoration and tissue sculpting.
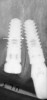
If an osteotomy is created that inadvertently results in a large perforation into the nasopalatine canal, then the contents of the canal should be removed.13 Subsequently, the implant should be inserted, and if a significant gap exists around the implant, it should be filled with bone graft material. In general, an implant should not be left in contact with soft tissue as this may interfere with osseointegration.13,14 If, however, a small mid-canal fenestration is accidentally created when preparing the osteotomy, it may go unnoticed and have little clinical effect (Figure 2).
Chatriyanuyoke et al15 reported that after assessing 120 cone-beam computed tomography (CBCT) scans, the mean distance from the nasopalatine canal to the maxillary incisor at the midroot and apex levels were 3.05 mm +/- 1.64 mm and 5.22 mm +/- 1.56 mm, respectively. They suggested that when placing immediate implants in the incisor region caution should be exercised to avoid perforating into the canal, especially in women and young adults, because their canal is closer to the central incisors than in adult men. Therefore, they suggested using tapered implants.
Bone Grafting the Nasopalatine Canal Before Implant Placement
If the nasopalatine canal is too large to obtain initial stability with an implant, or the position of the canal precludes placing an implant in a restoratively useful position, the nasopalatine canal can be enucleated, a particulate bone graft can be placed, and the site can be allowed to heal for several months before an implant is inserted.13 The concept of bone grafting the nasopalatine canal before implant placement was introduced by Rosenquist and Nystrom.13 They reported that patients (N = 4) who lost maxillary incisors due to trauma and underwent nasopalatine site preparation healed uneventfully. The procedure included enucleating the contents of the nasopalatine canal and bone grafting (autogenous bone from the chin). Implants were placed into the grafted sites after 4 to 5 months. During a 15-month monitoring period, none of the implants were lost.
Subsequently, Scher et al14 also noted successful outcomes with respect to grafting the nasopalatine canal after using alloplast bone (demineralized). Another variation of therapy for an atrophic maxilla was presented by Verardi and Pastagia.16 They bone grafted the nasopalatine canal and performed a guided bone regeneration procedure to restore maxillary ridge width to facilitate ideal implant placement at a later timepoint (N = 2).
Placement of an Implant Into the Nasopalatine Canal at Time of Surgery
A severely resorbed maxilla may also manifest advanced pneumatization of the maxillary sinus, nasal cavities close to the alveolar crest, and type 3 or 4 bone.17 A syndrome associated with an atrophic maxilla is referred to as combination syndrome. This occurs when the maxillary edentulous area is opposed by mandibular anterior teeth. It is coupled with maxillary ridge atrophy, tuberosity overgrowth, mandibular extrusion of teeth, and resorption of the mandibular posterior ridge.18 Different approaches to manage an atrophic maxilla have included sinus lifts, bone grafting, placement of implants in the pterygoid region, and placement of an implant in the nasopalatine canal as part of the overall therapy.13-18
When an atrophic maxilla is present, it may be desirable to place an implant directly into the nasopalatine canal. Several techniques can be used to accomplish this task. The nerve tissue can be displaced and an implant can be placed, or the canal can be enucleated and an implant placed with or without an accompanying bone graft. Each technique is briefly described here.
If an implant will be inserted within the nasopalatine canal, Artzi et al recommended that the contents of the nasopalatine canal be displaced (no debridement of the contents of the canal). They suggested that an implant could be inserted into the osteotomy and a bone block graft should be placed in between the implant and the soft-tissue contents of the canal (N = 1).19 It is the opinion of the present authors that this type of procedure would require a very large foramen. It also may be difficult to keep intact and displace the nerve and blood vessel complex that resides within this canal. Additionally, if the separation between the nerve tissue and the dental implant is not maintained during wound healing, then the bone-to-implant contact in that area would not be established. Others also successfully performed partial lateralization of the nasopalatine nerve at the incisive foramen when executing a ridge augmentation in the premaxilla before inserting dental implants.20
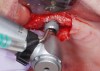
Peñarrocha et al21 assessed the survival rate of implants placed into the nasopalatine canal in patients who underwent rehabilitation of their atrophic maxilla. The nasopalatine canal was enucleated before implant placement and no concurrent bone grafting was performed. They monitored 13 implants placed in the incisive canal over a 9-year period. Six patients indicated they had a slight decrease in sensitivity in the anterior part of the palate that resolved within a few weeks. Two early failures before prosthetic loading were noted. Similarly, Spin-Neto et al22 reported successful placement of an implant into the canal without concurrent bone grafting (N = 1) (Figure 3).
After enucleating the contents of the canal, it is possible to place an implant into the canal and concurrently place particulate bone graft material in any gaps, then cover the graft material with a barrier to ensure bone fill around the implant. In this regard, Waasdorp et al23 used a bone particulate graft at the time of implant placement (N = 1). In addition, they placed a demineralized cancellous allograft block and a collagen barrier over the bone grafts. Another clinical scenario involves placing an implant in a desired location and, due to the position of the nasopalatine canal, the canal is intentionally entered.18-21 In other words, the osteotomy is created and it partially overlaps the position of the canal. In this situation, the canal contents should be enucleated, the implant inserted, and a graft placed into the gap between the implant and adjacent bone.
Potential Problems Associated With Implant Placement in or Near the Nasopalatine Canal
Potential Loss of Sensation in the Palatal Soft Tissue
Enucleating the nasopalatine canal results in severing the nasopalatine nerve, and there may be loss of sensation in the anterior palatal region. Pertinently, Rosenquist and Nystrom13 indicated that seven implants placed in four patients did not result in any patients experiencing loss of sensation. Similarly, others also reported that there were no residual sensory alterations after implant placement in the nasopalatine canal.16,21-23 This was probably due to collateral enervation provided by the greater palatine nerve to the anterior part of the palate.16 In contrast, Peñarrocha et al noted transient alteration of sensation in this region for several weeks.21 Raghoebar et al24 also reported a transient loss of feeling, which returned within several months. Ultimately, the patients reported no sensory deprivation.
On the other hand, Misch25 suggested that paresthesia or a dysesthesia is a potential complication of enucleating the nasopalatine canal. Therefore, patients must be informed that, if the nasopalatine canal is enucleated, there is a possibility of having reduced sensory innervation in the anterior palate. In the experience of the authors, transient or, on rare occasion, permanent altered sensation may occur; however, affected patients were subjectively unaware of this effect. They were primarily concerned with the functionality of the fixed or removable overdenture prosthesis. Failure to Achieve Osseointegration
An implant that is stable at initial insertion and submerged under the palatal soft tissue may be found to be mobile at its uncovering or before prosthetic loading. In the experience of the authors, this occurred one time (1 of 8 nasopalatine implants). In this case, it is recommended to remove the mobile implant and graft the canal. This will facilitate re-implantation after a healing period of several months.
Neuroma Development After Nasopalatine Severing or Canal Enucleation
Conceptually, after severing the nasopalatine nerve an amputation neuroma could form at the end of injured nerve fibers.26-28 A neuroma is a tumor or mass of unorganized nerve cells and nerve fibers. These lesions occur as a result of unregulated nerve regeneration and are often painful. However, this type of lesion has not been reported after nasopalatine surgery. Nevertheless, written informed consent describing these potential sequelae should be obtained.
Hemorrhage Associated With Enucleation of the Nasopalatine Canal
When enucleating the nasopalatine canal, hemorrhage may occur and its occurrence may be related to the size of the nasopalatine vessels. Injecting epinephrine 1:50,000 directly into the foramen can aid in controlling bleeding. In addition, twisting gauze and inserting it into the canal with pressure can impede oozing. It may be beneficial to saturate a piece of gauze with 1:50,000 epinephrine and insert it into the nasopalatine canal and hold it in place with a periosteal elevator for a few minutes. This is usually adequate to halt bleeding. A cautery unit also can assist in arresting hemorrhage. The cautery tip can be inserted into the nasopalatine canal. Ultimately, the implant will serve as the final device to arrest bleeding.
Size and Location of the Nasopalatine Canal May Be an Issue
An issue with respect to the proximity of the nasopalatine canal and maxillary central incisors may not be evident on regular radiographs. In this regard, Kraut and Boyden8 reported after assessing 84 scans that 4% of the time the size of the incisive canal suggested that normal preparation of an osteotomy for an implant may invade the nasopalatine canal. Accordingly, it has been suggested that if a CT scan is not available, a full-thickness mucoperiosteal palatal flap should be elevated to visualize the extent and location of the nasopalatine canal.19 However, in the authors’ estimation it probably is better not to raise a large palatal flap unless during drilling of the osteotomy it feels like the bone of the canal was penetrated.
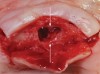
It also should be noted that the palatal aspect of the bone surrounding the nasopalatine canal is more apical than the buccal aspect of the canal (Figure 4 and Figure 5). Therefore, it may be prudent to perform ostectomy on the buccal aspect to create greater parity (the discrepancy may be too large to totally eliminate) between the buccal and palatal aspects. This will facilitate placing the platform of the implant more even with the bone buccally and palatally. Nevertheless, the buccal aspect of the implant platform will probably be 1 mm or more subcrestal and the lingual aspect 1 mm to 2 mm supracrestal. This anatomic disparity may cause difficulty when attaching a healing, interim, or definitive abutment to the implant. If this occurs, it is suggested that a nonflared component be used or judicious osseous profiling be performed to seat the component fully.
Ridge Resorption Effects on Nasopalatine Foramen and Canal
After teeth are removed, resorption of bone in the anterior maxilla occurs vertically and horizontally. Ridge width is lost mostly on the buccal aspect of the ridge. Therefore, the nasopalatine foramen apparently moves closer to the buccal aspect, which reduces the amount of ridge available for implant placement. A vertical height reduction can also shorten the height of the nasopalatine canal, especially on the buccal. The palatal aspect of the canal, which is part of the horizontal plate of the palate, is less likely to undergo extensive alteration. In this regard, Mardinger et al10 reported the average length of the canal after extraction shortened from 10.7 mm to 9 mm and the nasopalatine foramen constituted 36.5% of the ridge width (range, 13% to 58%). They noted that after tooth extraction the nasopalatine canal became larger in all directions. In severely resorbed ridges, the nasopalatine foramen enlarged by 32% (1.8 mm) and reached 5.5 mm in diameter. It was reported that the foramen could occupy up to 58% of the ridge.10 With respect to the buccal ridge, Bornstein et al29 noted a diminishing coronal width of the buccal bone among patients who had maxillary central incisors extracted and this resorption increased with time.
Prosthetic Considerations
In a ridge that is not resorbed, the nasopalatine canal generally is located approximately 7 mm from the buccal plate. This distance will probably be less after an extraction due to horizontal resorption. Furthermore, the ridge may be severely resorbed if a patient wore a maxillary denture. Other contributing factors resulting in a diminished ridge could be trauma or periodontitis. Ultimately, the type of prosthesis that will be fabricated is a critical determinant with respect to how the implant in the nasopalatine canal is used.
At first glance, it might be presumed that lingual implant placement in the nasopalatine canal may contribute to a fixed prosthesis extending too far palatally, thereby compromising phonetics. However, this assumption is not always true. The location of the implant can accommodate a screw-down fixed prosthesis. In general, when a screw-down prosthesis is constructed in the maxillary anterior region, it is desirable to have a horizontal disparity of 5 mm to 7 mm between the placement of the implant’s platform and the facial of the prosthetic tooth to be restored. This horizontal mismatch accommodates the required prosthetic components and a prosthetic tooth (Figure 6). Accordingly, the labial aspect of the prosthesis will be in correct position to provide excellent esthetics. Concerning intraoral comfort, a patient may perceive a slight elevation of the prosthesis on the palatal aspect, coincidental with the site of implant placement; however, compared to a denture, the patients can tolerate this minor protuberance. Conversely, if a removable prosthetic appliance is fabricated, a low-level unsplinted attachment can be used to minimize the acrylic height on the lingual aspect of the prosthesis (Figure 7).
Conclusions
The nasopalatine canal is an anatomic structure that can be managed surgically and prosthetically. Knowledge and application of surgical techniques that can be used in the nasopalatine region are needed to provide predictable results with respect to implant survival. Similarly, prosthetic management can accommodate a fixed or removable prosthesis.
Nevertheless, clinicians need to be aware of potential side effects related to manipulation of the nasopalatine canal (eg, loss of sensation, hemorrhage, neuromas). In this regard, managing intraoperative bleeding is within the scope of an experienced clinician, and neuroma formation in the nasopalatine canal due to implant placement has not been documented to date. Furthermore, numerous reports were cited relating predictable, successful placement of an implant into the canal and this rarely caused loss of palatal sensation.
Therefore, before placing an implant into the nasopalatine canal, careful consideration must be given to treatment alternatives and a detailed discussion with the patient must take place concerning the potential loss of some palatal sensation. For an experienced clinician, the nasopalatine canal should not be an impediment to a prosthetic plan if its presence can provide an opportunity to manage a case that might be impossible if the canal was not present. In conclusion, in the deficient or atrophied maxilla, if the benefit of placing an implant in the nasopalatine canal outweighs the potential risks, then this site can be advantageously utilized.
About the Authors
John Cavallaro, DDS
Clinical Professor
College of Dental Medicine
Columbia University
New York, New York
Private Practice
Surgical Implantology and Prosthodontics
Brooklyn, New York
Shota Tsuji, DDS, MS
Recently Graduated Periodontist
Division of Periodontics
Columbia University
New York, New York
Tzu-Shan Chiu, DDS, MS
Recently Graduated Periodontist
Division of Periodontics
Columbia University
New York, New York
Gary Greenstein, DDS, MS
Clinical Professor
College of Dental Medicine
Columbia University
New York, New York
Private Practice
Surgical Implantology and Periodontics
Freehold, New Jersey
Queries to the author regarding the course may be submitted to authorqueries@aegiscomm.com.
References
1. Al-Amery SM, Nambiar P, Jamaludin M, et al. Cone beam computed tomography assessment of the maxillary incisive canal and foramen: considerations of anatomical variations when placing immediate implants. PLoS One. 2015;10(2):e0117251.
2. Mraiwa N, Jacobs R, Van Cleynenbreugel J, et al. The nasopalatine canal revisited using 2D and 3D CT imaging. Dentomaxillofac Radiol. 2004;33(6):396-402.
3. Allard RH, van der Kwast WA, van der Waal I. Nasopalatine duct cyst. Review of the literature and report of 22 cases. Int J Oral Surg.1981;10(6):447-461.
4. Greenstein G, Cavallaro J, Tarnow D. Practical application of anatomy for the dental implant surgeon. J Periodontol. 2008;79(10):1833-1846.
5. Liang X, Jacobs R, Martens W, et al. Macro- and micro-anatomical, histological and computed tomography scan characterization of the nasopalatine canal. J Clin Periodontol. 2009;36(7):598-603.
6. Tozum TF, Guncu GN, Yildirim YD, et al. Evaluation of maxillary incisive canal characteristics related to dental implant treatment with computerized tomography: a clinical multicenter study. J Periodontol. 2012; 83(3):337-343.
7. Thakur AR, Burde K, Guttal K, et al. Anatomy and morphology of the nasopalatine canal using cone-beam computed tomography. Imaging Sci Dent. 2013;43(4):273-281.
8. Kraut RA, Boyden DK. Location of incisive canal in relation to central incisor implants. Implant Dent. 1998;7(3):221-225.
9. Song WC, Jo DI, Lee JY, et al. Microanatomy of the incisive canal using three-dimensional reconstruction of microCT images: an ex vivo study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108(4):583-590.
10. Mardinger O, Namani-Sadan N, Chaushu G, et al. Morphologic changes of the nasopalatine canal related to dental implantation: a radiologic study in different degrees of absorbed maxillae. J Periodontol. 2008;79(9):1659-1662.
11. Sclar AG. Surgical techniques for management of peri-implant soft tissues. In: Sclar AG, ed. Soft Tissue and Esthetic Considerations in Implant Therapy. Chicago, IL: Quintessence Publishing; 2003:6-62.
12. Magennis P. Sensory morbidity after palatal flap surgery–fact or fiction? J Ir Dent Assoc. 1990;36(2):60-61.
13. Rosenquist JB, Nystrom E. Occlusion of the incisal canal with bone chips. A procedure to facilitate insertion of implants in the anterior maxilla. Int J Oral Maxillofac Surg. 1992;21(4):210-211.
14. Scher ELC. Use of the incisive canal as a recipient site for root form implants: preliminary clinical reports. Implant Dent. 1994;3(1):38-41.
15. Chatriyanuyoke P, Lu CI, Suzuki Y, et al. Nasopalatine canal position relative to the maxillary central incisors: a cone beam computed tomography assessment. J Oral Implantol. 2012;38(6):713-717.
16. Verardi S, Pastagia J. Obliteration of the nasopalatine canal in conjunction with horizontal ridge augmentation. Compend Contin Educ Dent. 2012;33(2):116-120,122.
17. Lekholm U, Zarb G. Patient selection and preparation. In: Brånemark PI, Zarb GA, Albrektsson T, eds. Tissue-Integrated Prostheses: Osseointegration in Clinical Dentistry. Chicago, IL: Quintessence Publishing; 1985:199-209.
18. Peñarrocha M, Viña JA, Carrillo C, et al. Rehabilitation of reabsorbed maxillae with implants in buttresses in patients with combination syndrome. J Oral Maxillofac Surg. 2012;70(5):e322-e330.
19. Artzi Z, Nemcovsky CE, Bitlitum I, Segal P. Displacement of the incisive foramen in conjunction with implant placement in the anterior maxilla without jeopardizing vitality of nasopalatine nerve and vessels: a novel surgical approach. Clin Oral Implants Res 2000;11(5):505-510.
20. Urban I, Jovanovic SA, Buser D, Bornstein MM. Partial lateralization of the nasopalatine nerve at the incisive foramen for ridge augmentation in the anterior maxilla prior to placement of dental implants: a retrospective case series evaluating self-reported data and neurosensory testing. Int J Periodontics Restorative Dent. 2015;35(2):169-177.
21. Peñarrocha D, Candel E, Guirado JL, et al. Implants placed in the nasopalatine canal to rehabilitate severely atrophic maxillae: a retrospective study with long follow-up. J Oral Implantol. 2014;40(6):699-706.
22. Spin-Neto R, Bedran TB, de Paula WN, et al. Incisive canal deflation for correct implant placement: case report. Implant Dent. 2009;18(6): 473-479.
23. Waasdorp J. Enucleation of the incisive canal for implant placement: a comprehensive literature review and case report. J Oral Implantol. 2016;42(2):180-183.
24. Raghoebar GM, den Hartog L, Vissink A. Augmentation in proximity to the incisive foramen to allow placement of endosseous implants: a case series. J Oral Maxillofac Surg. 2010;68(9):2267-2271.
25. Misch E. Premaxilla surgery: implant insertion, bone spreading, nasal floor elevation, and incisive foramen implants In: Misch CE. Contemporary Implant Dentistry. 3rd ed. St. Louis, MO: Mosby; 2007:769-798.
26. Robinson M, Slavkin HC. Dental amputation neuromas. J Am Dent Assoc. 1965;70:662-675.
27. Lee HT. Amputation neuroma of the maxilla. Dent J Malaysia Singapore. 1971;11(1):17-19.
28. Hegedus F, Diecidue RJ. Trigeminal nerve injuries after mandibular implant placement–practical knowledge for clinicians. Int J Oral Maxillofac Implants. 2006;21(1):111-116.
29. Bornstein MM, Balsiger R, Sendi P, von Arx T. Morphology of the nasopalatine canal and dental implant surgery: a radiographic analysis of 100 consecutive patients using limited cone-beam computed tomography. Clin Oral Impl Res. 2011;22(3):295-301.