You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Dental erosion is a common clinical finding, defined as the permanent loss of tooth structure related to acid exposure, either extrinsic (eg, diet) or intrinsic (eg, endogenous acid, typically stomach acids, often from vomiting or gastroesophageal reflux disorder [GERD]), that does not involve microorganisms.1-6 Typically, erosion does not occur by itself but rather with coexisting factors. Due to similarities with other categories of tooth wear, the condition can be a challenge to diagnose. It is difficult to define exactly how common dental erosion is because the prevalence has been understudied, with percentages in various countries ranging from 5% to 97%.7 This article will explore dental erosion, its coexisting contributors, and prevention and management strategies available to oral healthcare providers to address the condition in adult patients.
Why Diagnosis Can Be Challenging
Dental erosion is common and irreversible. However, other types of tooth wear can make diagnosing erosion more difficult. One type is attrition, which is the loss of enamel and dentin due to mechanical wear, such as grinding or clenching. Another type is abrasion, or the loss of enamel and dentin due to frictional force between a person's teeth and an external object, such as toothbrushing with an abrasive dentifrice.8,9 There is also abfraction, which is caused by normal and abnormal tooth function.10,11
To properly diagnose a patient for dental erosion alone or in combination with another type of tooth wear, it is important to recognize the symptoms a patient may present. They include temperature sensitivity to liquids, solids, or air-typically with cold but also with heat, or no symptoms at all. Other symptoms may include toothbrush sensitivity; rapid tooth structure loss, which would result in more sensitivity; slow tooth structure loss, which would cause less or no sensitivity; or pulpal pathology where the sensitivity varies. These symptoms may mimic sensitivity from other sources, such as exposed root surfaces from gingival recession.
Clinical Signs
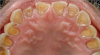
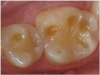
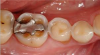
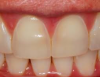
Figure 1 shows an example of lost enamel and dentin from the palatal surface of the teeth. This clinical manifestation is often found with repeated and/or prolonged exposure of the teeth to acidic substances, such as is found in gastric esophageal reflux disease or purging as part of an eating disorder. Repeated exposure of the enamel to acidic gastric contents may lead to the dentin exposure, similarly to what is seen in the figure. Gastrointestinal reflux disorders, pregnancy vomiting, and purging as found in some eating disorders are a few examples of how gastric contents can enter the oral cavity on a repeated and/or prolonged basis. Chipping, fracturing, and signs of pulpal involvement are also present. The surfaces that are susceptible to dental erosion include the facial/buccal, palatal/lingual, and occlusal/incisal. Lesions will often present as a smooth, hard surface with sharply defined margins. Cupping lesions of cusp tips may also occur, leading to exposure of dentin (Figure 2). Preexisting restorations may have the appearance of floating above the surrounding tooth structure (Figure 3). Presence of other tooth wear and confusion with other types are also possible clinical signs of dental erosion.12
One of the earliest signs of erosive lesions is dull or matte enamel. This softened enamel is more easily affected by attrition. If erosion worsens, it creates concave lesions, and eventually the yellow dentin becomes exposed. This also causes uneven wear on the surrounding enamel, higher porosity, and higher solubility.12
Risk Factors
Some patients may be more likely to suffer from dental erosion based on diet and nutrition, lifestyle, and oral health habits. For example, the duration and frequency of which a person exposes the oral cavity to different types of food and drink, as well as the types of food and drink (eg, solid, liquid, chemical components of the substance), can contribute to dental erosion.13,14 Healthy eating is recommended more strongly today than it was several decades ago. However, eating multiple meals throughout the day, including fresh fruits and juices, exposes the oral cavity more frequently to sugar and often more acidic substances. Holding liquids in the mouth, especially acidic substances, can also contribute to erosive risk.14
Extrinsic Acid
Extrinsic acids may have a critical pH of 5.5 to contribute to caries risk, but it is important to remember that different solutions contribute to dental erosion as opposed to plaque. Titratability and hydrogen ion concentration, calcium, phosphate, and fluoride activity all contribute to the saturation of the compound solution and therefore the erosive effect of an acid. For example, citric acid may be the most erosive because of its characteristic of calcium chelation, whereas sour milk, with its lower pH, is still heavily saturated in calcium, which causes it to have lower erosive potential. Therefore, there are a number of characteristics that contribute to extrinsic acids and cause them to be either more or less prone to erosive potential.4 Although the pH of common beverages ranges from 2.0 to 4.0 for carbonated soft drinks, energy drinks, fruit juice drinks, and some prepared teas and coffee drinks, the absolute pH of a substance is not, in and of itself, the sole indicator of erosive potential, due to the factors discussed previously.13,15
Intrinsic Acid
Intrinsic acid erosion occurs when gastric acids enter the oral cavity and saliva, or when oral health habits cannot adequately buffer it due to excessive frequency.1,16,17 These acids can come from GERD, metabolic or endocrine disorders, autoimmune disorders, drug side effects, stress-induced vomiting, eating disorders, and salivary hypofunction.16,18Reflux caused by GERD can be affected by metabolic, endocrine, and autoimmune disorders, which can make the reflex systems worse and negatively affect the quality and quantity of saliva.19
Oral Habits
Parafunctional activity, such as bruxism, can increase the mechanical chemical component of dental erosion; the resulting tooth-on-tooth wear can cause more tooth wear, especially in a patient who already has risk factors for erosion. Other factors that can increase erosive wear include snacking on hard candies and eating or sucking on citrus fruits13; certain physical activity, such as swimming in chlorinated pools, depending on the amount of chlorination; and the frequent use of sports or energy drinks.3,4
Systemic Comorbidities
Many systemic comorbidities can affect saliva quality and quantity, such as eating disorders/disordered eating; reflux/functional dyspepsia; medication side effects such as dry mouth; and medications themselves, such as chewable vitamins or asthma medications. These comorbidities may contribute to potential erosion. Radiation therapy that specifically targets the head and neck can affect the salivary glands if they are in the path of the radiation; although it is not as common, salivary gland agenesis and removal would cause salivary changes that could increase the potential erosive risk in a patient.20
Occupational Factors
Some occupations have an increased risk of erosion, such as those that expose an individual to acidic gases that may be inhaled through work involving battery factories, dynamite production, galvanized products, or fertilizer manufacturers. Wine tasting as an occupation continually exposes teeth to acids in wine, posing a potential erosive risk. Professional athletes, especially swimmers, or anyone who uses sports or energy drinks regularly during periods of training may also have higher risk.3,4
Oral Hygiene and Home Care
It is important to ask patients how often they practice home hygiene care, as well as how soon after eating and drinking. Although the amount of time it takes to cause an erosive reaction on the tooth surface varies for each individual, it is still important to review this information with the patient and suggest that he or she rinse the mouth out with water and chew sugarless gum to increase the salivary flow, which helps surface remineralization of the tooth enamel.1 The clinician should also review the products a patient uses, such as rinses, pastes, and toothbrushes, to understand the pH levels and abrasive nature of the products. The oral healthcare provider can recommend the use of different oral products according to an individual patient's needs and specify the frequency and duration of using these products to minimize the erosive risk.
Protective Factors
The human body is not defenseless against dental erosion; there are inherent protective factors, such as saliva. The appropriate quantity and quality of saliva helps dilute acids in the oral cavity, clear acid from food or drink when it is swallowed, and buffer acidic substances with bicarbonate. In a healthy individual, saliva is typically saturated with respect to the tooth in calcium, phosphate, and fluoride, which is essential for remineralization of enamel.21-28 Additionally, acid-induced stimulation occurs in saliva when a person ingests an acidic food or beverage item to help clear the erosive substance.28
The actual surface of the teeth also has a protective factor, called the salivary pellicle, which is an organic film that covers the oral soft and hard tissues. It can act as a diffusion barrier to keep substances out and also as a semipermeable membrane that allows only certain substances through. The salivary pellicle also contains mucins and other organic compounds to slow mineral loss on the tooth surface.28
Assessment and Diagnosis
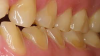
When a patient presents with the clinical signs and symptoms of dental erosion, as described above, a comprehensive examination should be performed, and treatment planning should be explored. Various phases of dental erosion will affect the treatment recommendations for an individual patient. If the patient is in the urgent phase, there may be pain or symptoms of erosion. In the systemic phase, any comorbidities or habits that could increase the erosive risk for the patient should be assessed. Dental erosion is primarily a surface phenomenon. Enamel erosion is characterized by a bulk loss of tooth structure with a partly demineralized surface layer left behind with a decreased microhardness. The erosion is mostly diffusion controlled in the dentin. Increasing exposure of the organic matrix to the source of erosive potential slows ion diffusion, which, in turn, slows the progression of dentin erosion. Figure 4 displays a buccal view of dental erosion that has progressed through the enamel, thus exposing dentin. The lesions have sharp, clearly delineated borders with a defined bulk loss of enamel. Erosion that has not yet progressed into dentin is shown in Figure 5. The outline is sharp and well-defined, but the tissue loss is more subtle, without the obvious color change of exposed dentin.29
Disease control is another consideration. If there are carious lesions, it is important to control caries risk, active caries, and any periodontal concerns before evaluating the oral cavity in the corrective phase, which is the replacement of lost tooth structure and overall rehabilitation to maintain functional pairs of teeth so that the patient can chew comfortably.12
A reevaluation ensures that the treatment prognosis remains favorable and that nothing is introduced to negatively affect the treatment. Prevention is crucial. Establishing a baseline for future follow-up should include radiographs and clinical photographs. It is vital to use study models to determine which treatment options may be appropriate for the patient.12
As part of the assessment and diagnosis, when suspicious lesions are present, it should be established whether the erosion is ongoing or historical. This determination can be challenging during a patient's initial appointment; therefore, it is important to assess the oral health literacy of the patient. What is his or her diet and lifestyle? What behaviors, habits, or occupation affect the oral health of the patient? What diagnoses are present that can contribute to higher erosive potential, such as over-the-counter and prescription medications?30
The oral healthcare provider is responsible for determining whether a medical consultation is necessary. If there are no obvious revelations from the drug history or diagnoses, is the presentation indicative of a reflux disorder? In that case, a medical consultation with the patient's primary care physician should be considered. If low-quality protective factors, such as rope-like or foamy saliva or dry mouth are present, the oral healthcare provider should examine any dental or medical history suggestive of decreased quality or quantity of saliva.28,31
Prevention and Management
After the initial assessment is complete, if there is erosive potential but no clinical evidence of erosion, it is important to educate the patient about potential risky behavior that could cause erosion, such as eating citrus fruits often, consuming energy drinks and soda frequently, and other eating and drinking habits.1,18 The patient should also be informed that there are preventive practices to help with erosive risk, such as certain oral hygiene habits. This is also true for caries risk assessment. The frequency of fluoride treatment should be considered, along with how it is applied-by either an over-the-counter rinse or in the dental office as a topical treatment to remineralize the enamel surface of teeth.21-28 Current evidence suggests that conventional fluorides may offer some limited protection against erosion, mainly from the CaF2 precipitates formed on the surface of enamel. High-concentration fluoride products, as well as frequent applications, are potential effective approaches in the prevention of dental erosion. The evidence is less supportive of the effect of fluoride on the dentin organic matrix. Mineral repair and precipitation agents have also been investigated as alternatives to fluoride in addressing dental erosion. However, products designed to deliver calcium and/or phosphate have not been shown to be successful or significant against dental erosion. In fact, casein phosphopeptide-amorphous calcium phosphate (CPP-ACP) has been shown to be less effective than fluoride in anti-erosion efforts, albeit in a limited number of studies.12,29
If there is active erosion, management should be somewhat different. If the erosion is correctly identified as ongoing, there are preventive strategies, as noted above; other management strategies include identification and mitigation of risk factors, patient education, medical consultation if there is suspicion of GERD, caries risk assessment and adjunctive fluoride therapy, and restorative care if appropriate for a specific patient. It may be necessary to postpone definitive corrective care until risk factors have been managed, depending on the extent of tooth loss and the existence of caries or sensitivity in the patient.12
Treatment
After historical and/or a significant amount of tooth loss has been identified and the dental erosive potential has been decreased or mitigated, it is then appropriate to address the lost tooth structure. At this point, disease would be controlled, and any pulpal involvement would be addressed. The degree of tooth structure loss should be analyzed, and the patient should be asked about sensitivity and esthetic concerns. Additionally, the tooth structure must be assessed for any diminished vertical dimension. If this is the case, definitive corrective restorative care would be delayed until risk factors have been mitigated and the erosion arrested.11,32
Pulpal Involvement
When pulpal involvement is revealed during the assessment, endodontic therapy is likely needed to rehabilitate the tooth. It is possible that there is such extensive tooth structure loss that not enough tooth will remain to use an indirect restoration, and endodontic therapy would be necessary to appropriately restore the tooth. If caries is also present, the caries must be removed, and risk must be addressed in conjunction with erosion. This situation may present as an urgent case because the erosion may have been occurring and then worsened by affecting the pulp, creating the urgency that made the patient seek professional attention.32
Degree of Structure Loss
If structure loss compromises the integrity of the clinical crown, it may suggest restorative care, which could also be dictated by patient sensitivity, esthetic concerns, or the presence of caries. The loss of vertical dimension manifested in functional loss may determine the need for more comprehensive restorative care. Increasing the vertical dimension will allow corrective therapy, but the clinician must be certain that the dental erosion is mitigated successfully before extensive rehabilitation.33
Restorative Replacement
If the vertical dimension is not problematic or causing functional issues in the patient, an indirect restoration, such as a crown or onlay, may be useful-or in cases where the structure loss is not very significant, a direct restorative material, such as composite, resin, or a resin-modified glass-ionomer, may be used instead. The patient's esthetic concerns and sensitivity, as well as caries risk and type of tooth loss, will also be factors that help determine the best way to restore the tooth structure.33
Functional Loss of Tooth Structure
If the degree of tooth structure loss is so great that there is diminished function manifesting as vertical dimension, this would indicate a complex rehabilitation, including opening the vertical dimension with transitional restorations or appliances to evaluate the patient's ability to tolerate the increase in space. Dental erosion typically occurs over time and slowly enough that the patient does not realize the change of space between the jaws. Risks must be mitigated before investing the time and resources required for this type of complex rehabilitation. The patient may need a referral to a prosthodontist or specialist, depending on the extent of the erosion.33
Summary
Patients are best served by correct diagnosis of dental erosion and identification of its contributing factors and risk factors. All possible types of tooth wear should be considered, and any coexisting conditions should be treated to allow each patient the best chance of conserving his or her tooth structure. Prevention and reduction are the best possible defenses against this irreversible loss of hard tissue.
References
1. Dental erosion. American Dental Association. https://www.ada.org/en/member-center/oral-health-topics/dental-erosion. Updated June 14, 2017. Accessed February 1, 2019.
2. Kanzow P, Wegehaupt FJ, Attin T, Wiegand A. Etiology and pathogenesis of dental erosion. Quintessence Int.2016;47(4):275-278.
3. Imfeld T. Dental erosion. Definition, classification and links. Eur J Oral Sci.1996;104(2 pt 2):151-155.
4. Zero DT. Etiology of dental erosion--extrinsic factors. Eur J Oral Sci.1996;104(2 pt 2):162-177.
5. Schlossman M, Montana M. Preventing damage to oral hard and soft tissues. In: Spolarich AE, Panagakos FS, eds. Prevention Across the Lifespan: A Review of Evidence-Based Interventions for Common Oral Conditions.Charlotte, NC: Professional Audience Communications, Inc.; 2017:97-120.
6. Mandel L. Dental erosion due to wine consumption. J Am Dent Assoc.2005;136(1):71-75.
7. Chrysanthakopoulos NA. Prevalence and associated factors of dental erosion in a population of Greek adults. Acta Stomatologica Croatica.2012;46(4):263-272.
8. Arnold WH, Gröger C, Bizhang M, Naumova EA. Dentin abrasivity of various desensitizing toothpastes. Head Face Med. 2016;12:16. doi: 10.1186/s13005-016-0113-1.
9. Barbour ME, Rees GD. The role of erosion, abrasion, and attrition in tooth wear. J Clin Dent. 2006;17(4):88-93.
10. Shellis RP, Addy M. The interactions between attrition, abrasion and erosion in tooth wear. Monogr Oral Sci. 2014;25:32-45.
11. Nascimento MM, Dilbone DA, Pereira PN, et al. Abfraction lesions: etiology, diagnosis, and treatment options. Clin Cosmet Investig Dent. 2016;8:79-87.
12. Lussi A, Ganss C, eds. Erosive Tooth Wear. Basel, Switzerland: Karger; 2014. Monographs in Oral Science, vol. 25.
13. Zimmer S, Kirchner G, Bizhang M, Benedix M. Influence of various acidic beverages on tooth erosion. Evaluation by a new method. PLoS One. 2015;10(6):e0129462.
14. Lussi A, Jaeggi T. Erosion-diagnosis and risk factors. Clin Oral Investig.2008;12(suppl 1):S5-S13.
15. Reddy A, Norris DF, Momeni SS, et al. The pH of beverages in the United States. J Am Dent Assoc. 2016;147(4):255-263.
16. Scheutzel P. Etiology of dental erosion--intrinsic factors. Eur J Oral Sci. 1996;104(2 pt 2):178-190.
17.Salas MM, Nascimento GG, Vargas-Ferreira F, et al. Diet influenced tooth erosion prevalence in children and adolescents: results of a meta-analysis and meta-regression. J Dent. 2015;43(8):865-875.
18. Carvalho TS, Colon P, Ganss C, et al. Consensus report of the European Federation of Conservative Dentistry: erosive tooth wear-diagnosis and management. Clin Oral Investig. 2015;19(7):1557-1561.
19. Mikami DJ, Murayama KM. Physiology and pathogenesis of gastroesophageal reflux disease. Surg Clin North Am. 2015;95(3):515-525.
20. Mohan RP, Verma S, Chawa VR, Tyagi K. Non-syndromic non-familial agenesis of major salivary glands: a report of two cases with review of literature. J Clin Imaging Sci. 2013;3(suppl 1):2.
21. West N, Seong J, Macdonald E, et al. A randomised clinical study to measure the anti-erosion benefits of a stannous-containing sodium fluoride dentifrice. J Indian Soc Periodontol. 2015;19(2):182-187.
22. Bellamy PG, Harris R, Date RF, et al. In situ clinical evaluation of a stabilised, stannous fluoride dentifrice. Int Dent J. 2014;64(suppl 1):43-50.
23. Hooper S, Seong J, Macdonald E, et al. A randomised in situ trial, measuring the anti-erosive properties of a stannous-containing sodium fluoride dentifrice compared with a sodium fluoride/potassium nitrate dentifrice. Int Dent J. 2014;64(suppl 1):35-42.
24. Eversole SL, Saunders-Burkhardt K, Faller RV. Erosion protection comparison of stabilised SnF2, mixed fluoride active and SMFP/arginine-containing dentifrices. Int Dent J. 2014;64(suppl 1):22-28.
25. Faller RV, Eversole SL, Saunders-Burkhardt K. Protective benefits of a stabilised stannous-containing fluoride dentifrice against erosive acid damage. Int Dent J. 2014;64(suppl 1):29-34.
26. Faller RV, Eversole SL. Enamel protection from acid challenge--benefits of marketed fluoride dentifrices. J Clin Dent. 2013;24(1):25-30.
27. O'Toole S, Bartlett D, Moazzez R. Efficacy of sodium and stannous fluoride mouthrinses when used before single and multiple erosive challenges. Aust Dent J. 2016;61(4):497-501.
28. Buzalaf MA, Hannas AR, Kato MT. Saliva and dental erosion. J Appl Oral Sci. 2012;20(5):493-502.
29. Buzalaf MAR, ed. Fluoride and the Oral Environment. Basel, Switzerland: Karger; 2011. Monographs in Oral Science, vol. 22.
30. Bruno V, Amato M, Catapano S, Iovino P. J Med Case Rep. 2015;9:250. doi: 10.1186/s13256-015-0738-x.
31. Meurman JH, ten Cate JM. Pathogenesis and modifying factors of dental erosion. Eur J Oral Sci. 1996;104(2 pt 2):199-206.
32. Vailati F, Belser UC. Classification and treatment of the anterior maxillary dentition affected by dental erosion: the ACE Classification. Int J Periodontics Restorative Dent. 2010;30(6):559-571.
33. Schlichting LH, Maia HP, Baratieri LN, Magne P. Novel-design ultra-thin CAD/CAM composite resin and ceramic occlusal veneers for the treatment of severe dental erosion. J Prosthet Dent. 2011;105(4):217-226.