You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
For the last few years, with the development of bioactive regenerative materials, the author has believed it to be important to choose materials according to the following premise: All things being nearly equal with clinical characteristics, if filling materials can have a positive influence on the oral environment, shouldn't they? The goal for this author in his clinical setting is to use materials that have exemplary clinical properties but that also make a positive impact on the oral environment through ion release that may reduce sensitivity, decrease plaque formation, and increase clinical performance.
For many years, restorative care mainly involved treating teeth damaged from decay by merely filling the hole or replacing missing tooth structure with a material without regard to its positive regenerative effects. The primary restorative goal of dentistry was to remove decay. Today, the goal of research and many clinicians is to not just fill the hole but instead to provide therapy by placing materials that cause regeneration of tissues, or to provide an environment where tissues can heal. If the main physical properties and esthetics of various materials are relatively equal, it makes sense to use materials that provide positive effects on the tissues instead of negative effects.
Dental literature and research today includes a strong focus on development of products that are bioactive in nature. Dworkin et al define a bioactive restorative as a material that forms a layer of hydroxyapatite when immersed in a simulated body fluid or a solution containing inorganic phosphate.1 In regard to restorative materials, the goal is to provide a therapeutic environment that reduces microbes, stimulates secondary dentin tooth repair, and ultimately enables a less sensitive, longer-lasting restoration.
Not everyone has the same definition of bioactive, however. In a survey conducted by Nathaniel Lawson, DMD, director of the division of biomaterials at the University of Alabama at Birmingham School of Dentistry, along with the American Dental Association, 400 dentists were asked about the definition of the word in relation to modern dentistry.2 Most of the dentists surveyed believed bioactive meant that a material was either encouraging the formation of reparative dentin or releasing an ion. To this point, glass-ionomers and fluoride-releasing materials have been releasing ions for many years without being directly affiliated with dentin repair.
Bioactive Materials and Healing
Bioactive materials differ from traditional materials by being biologically tolerant while working with existing tissues to encourage the formation of hydroxyapatite.2 Bioactive restorative materials are generally available in two chemical classes: calcium aluminates and calcium silicates. These materials have demonstrated their ability to stimulate the depositing of hydroxyapatite and therefore tissue regeneration from living tissue (pulp, blood vessels, dentin, and bone).3 Bioactive restorative materials are moisture tolerant, help control microbes, and help to stimulate the development of apatite at the material/tooth interface. Dentists can now find materials that will have a favorable response to the tissues in the mouth and, when possible, those that stimulate tooth repair.2,3
Bioactive Restoratives and Acid Neutralization
Bioactive restorative materials rely on acid neutralization. Both chemical classes of bioactive restorative materials are non-resin based and are set with an acid-base reaction; they each produce an alkaline pH after they have set. This is important because most patients do not adequately brush their teeth, bacteria grow in plaque, and acids are formed, resulting in a potential demineralization of hard dental tissues. Consequently, longevity of a restoration and remineralization are concerns. This demineralization is a factor in tooth degradation and restoration failure. Regular flowable composites maintain acidity. However, with the new materials working to bring acid neutrality, this issue can be avoided while striving for regeneration.4,5
The clinical outcome of bioactive regenerative techniques depends on three separate factors.6Advanced regenerative technologies for periodontal tissue repair are affected by:
• patient-associated factors (such as smoking, infection, plaque, and membrane exposure)
• effects of occlusal forces that deliver intermittent loads in axial and transverse dimensions
• factors associated with the clinical skills of the operator, such as lack of primary closure of the surgical wound
The Preventive Model
Bioactive materials have the potential to release ions that the dentinal tissues can use to help remineralize hard dentinal tissues.7 The process of ion release by a dentin material, uptake and use by the tissues, and repeating of this process is called ionic exchange.8 Calcium, phosphates, and fluoride are some of the ions available through Giomer materials that can participate with oral fluids and tooth structure, which can help in remineralization and a reparative environment.9 Therefore, these bioactive materials not only serve to fill in the space between the tooth, but also help to repair the dentin and sustain a healthier tooth structure, thus creating a preventive model.10 In summary, these materials can be used for several purposes:
• pulp-capping material
• permanent restorations
• dentinal tubule occlusion
• regeneration of live bone tissue to promote tooth vitality
• tooth remineralization and strengthening through fluoride release and/or the release of other minerals
Types of Bioactive Regenerative Materials
Materials placed in the mouth will have an influence on the tissues in the oral environment. Different types of bioactive regenerative materials have different advantages and disadvantages. Various types of bioactive regenerative materials are described below.
Bioactive Resin-Modified Glass-Ionomer Cement
Glass-ionomers are used to temporarily fill deciduous teeth and are also well-tolerated for the purpose of permanently sealing bridges and crowns. They are somewhat soluble in saliva, allowing the surface to become porous and therefore pick up debris and stain. Glass-ionomers have a chemical bond with tooth structure and may aid in remineralization of the remaining dental structures by the release of fluoride, and they have a chemical bond with dental tissue. They also assist in the prevention of bacterial microleakage through adhesion with ion exchanges.11
Giomers
Bioactive materials elicit a response from living tissue in that they are dynamic, not passive, and release ions that can result in changes at the tooth surface by stimulation of remineralization.12Bio-interactive materials are those that release ions promoting a change in the environment near the tooth but that aren't necessarily used by the tooth for remineralization.13
Giomer is a relatively recently developed group of restorative materials based on surface pre-reacted glass (S-PRG) technology, where special fillers are added in the resin matrix. These are bio-interactive, bio-compatible materials that have the physical and esthetic properties of composite resins but also with high radiopacity, an anti-plaque effect, fluoride release, and recharge.14 Their long-term clinical performance has been favorable.
Bioactive Composites
Dental composites, also known as white fillings, are one of the first choices in conservative dentistry. However, often composite restorations fail because of microleakage and its decomposing effect on the layer of adhesive. Generally, this causes marginal staining. Recently, a new generation of composites have been introduced that are much more likely to effectively seal to adequately prevent micoleakage and slow or prevent the progression of secondary caries. The goal of these newer composites is to combine antibacterial and regenerative action. Much systematic work has been completed to accomplish this. In addition to quaternary ammonium dimethacrylate (QDAM) and silver nanoparticles (AgNP), calcium phosphate nanoparticles have been added to stimulate regenerative properties. By adding Ag-doped bioactive glass in the resin composite, the total bond strength of the new composites, either before or after treatment, encourages remineralizing and antibacterial properties.15
Bioactive Ceramics
Bioactive ceramics have been clinically used as a substitute for bone. Therefore, some bioactive ceramics are used to repair bone defects. Bioactive ceramic-based materials with designed reactivity for bone-tissue regeneration have shown osteoconduction by formation of biologically active bone-like apatite through chemical reaction of the ceramic surface with surrounding body fluid.16 Bioactive ceramics also solve the problem of the body's reaction (encapsulation in fibrous tissue) to a foreign object. New bone formation on the surface of bioactive ceramics is referred to as osteoconductivity.
Bioactive Materials in Action
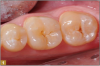
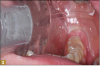
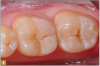
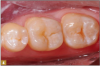
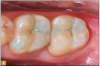
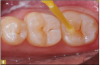
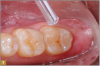
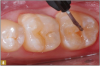
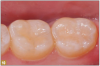
Giomer restoratives are bioactive regenerative materials that can be used where traditional direct composite materials are indicated. In the case presented in Figure 1, decay was noted on the occlusal surfaces of a first and second molar. What often looks like a very small amount of decay on the surface can lead to the discovery of significant dentin destruction. The quadrant was isolated and air abrasion was performed using 50 µm aluminum oxide (Figure 2). The occlusal groove enamel was abraded, revealing obvious decay to the dentin (Figure 3). Decay was identified with caries indicator and removed with a #2 round bur and slow-speed handpiece, with the goal of preserving as much healthy tooth structure as possible (Figure 4). Caries indicator stains denatured collagen and debris and helps to identify areas needing attention by the dentist, but equally importantly, it identifies areas without damage that can be preserved.
The conservative nature of these preparations allows complete restoration with a flowable Giomer. The teeth were etched with 37% phosphoric acid for 15 seconds (Figure 5). After thorough rinsing, a universal bonding material was applied with several coats and air thinned (Figure 6). Air thinning is important to remove the solvents and to provide and thin the film. The surface at that point should be visibly shiny, signifying complete, effective preparation coverage by the bonding agent (Figure 7).
A 0.5 to 1.0 mm layer of Giomer flowable with self-leving properties and low flow was applied to cover the dentin; it was then light-cured for 15 seconds (Figure 8). Uncured resin weakens the restoration at the tooth-restorative interface and may be a pulpal irritant resulting in patient discomfort. It is therefore prudent that the layers closest to dentin be polymerized to their greatest potential. A thin layer of material over the dentin provides the best potential for ultraviolet light-induced polymerization before thicker, harder-to-penetrate layers are added. Sealing the dentin with a thin layer of well-cured Giomer flowable allows ionic release at the restoration-dentin interface. The bond of restorative materials deteriorates with time from many factors, such as enzymes, acids, and mechanical forces, which may decrease sensitivity while increasing restoration longevity. Flowable was then applied to complete the contour of the restoration, and it was then light-cured (Figure 9). The occlusion was checked with articulating paper, adjusted, and polished with a rubber cup (Figure 10). The result was a conservative, esthetic, regenerative restoration.
Conclusion
Excellent clinical performance with esthetic, comfortable, long-lasting restorations is the clinical goal. It is advantageous that the materials put into the mouth have a regenerative influence on fluids and tissues instead of a negative one. Release of ions such as fluoride, silicates, sodium, and calcium can influence the oral environment to be one of regeneration instead of degradation. Some ion release acts to help neutralize acids that come from plaque. If materials are otherwise similar in clinical performance and esthetics, those that have a positive influence on the oral environment should be preferred. All characteristics should be considered to meet the goals of each case. Bioactive properties may provide a clinical advantage for the dentist and patient.
About the Author
Jack D. Griffin, Jr., DMD
Private Practice
Lake St. Louis, Missouri
References
1. Dworkin O, Kugel G, Loo C. What is bioactive dentistry? A review. Dent Today. 2018;37(1):44-46.
2. Elsenpeter R. How bioactive materials are changing restorative dentistry. Dental Products Report. http://www.dentalproductsreport.com/dental/article/how-bioactive-materials-are-changing-restorative-dentistry. Published June 21, 2018. Accessed January 31, 2019.
3.Goldstep F. Bioactivity in restorative dentistry: a user's guide. Oral Health. https://www.oralhealthgroup.com/features/bioactivity-restorative-dentistry-users-guide/. Published January 11, 2018. Accessed January 31, 2019.
4.Nagafuji A, Shinno K, Kimoto K, et al. Acid-neutralizing capacity of resin-based materials containing S-PRG filler. 2010 IADR/PER General Session (Barcelona, Spain);89(B):1437.
5. Ratna AA, Triaminingsih S, Eriwati YK. The effect of prolonged immersion of giomer bulk-fill composite resin on the pH value of artificial saliva and resin surface roughness. J Phys: Conf Ser. 2017;884:012011.
6. Ramseier CA, Rasperini G, Batia S, Giannobile WV. Advanced regenerative technologies for periodontal tissue repair. Periodontol 2000. 2012;59(1):185-102.
7. Gordan VV, Blaser PK, Watson RE, et al. A clinical evaluation of a giomer restorative system containing surface prereacted glass ionomer filler: results from a 13-year recall examination. J Am Dent Assoc. 2014;145(10):1036-1043.
8. Jefferies SR. Bioactive and biomimetic restorative materials: a comprehensive review. Part I. J Esthet Restor Dent. 2014;26(1):14-26.
9. Cao W, Hench LL. Bioactive materials. Ceram Int. 1996;22(6):493-507.
10.Sonarkar S, Purba R. Bioactive materials in conservative dentistry. Int J Contemp Dent Med Rev. 2015. doi: 10.15713/ins.ijcdmr.47.
11. Sidhu SK, Nicholson JW. A review of glass-ionomer cements for clinical dentistry. J Funct Biomater. 2016;7(3):16. doi: 10.3390/jfb7030016.
12. Hench LL, Splinter RJ, Allen WC, Greenlee TK. Bonding mechanisms at the interface of ceramic prosthetic materials. J Biomed Mater Res. 1071;5(6):117-141.
13. Gandolfi MG, Siboni F, Botero T, et al. Calcium silicate and calcium hydroxide materials for pulp capping: biointeractivity, porosity, solubility and bioactivity of current formulations. J Appl Biomater Funct Mater. 2015;13(1):43-60.
14. Jyothi KN, Annapurna S, Kumar AS, et al. Clinical evaluation of giomer- and resin-modified glass ionomer cement in class V noncarious cervical lesions: an in vivo study. J Conserv Dent. 2011; 14(4):409-413.
15. Chatzistavrou X, Lefkelidou A, Papadopoulou L, et al. Bactericidal and bioactive dental composites. Front Physiol.2018;9:103.
16. Ohtsuki C, Kamitakahara M, Miyazaki T. Bioactive ceramic-based materials with designed reactivity for bone tissue regeneration. J R Soc Interface. 2009;6(suppl 3):S349-S360.