You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Dental technology is a constantly evolving field that encompasses all facets of the dental industry. Advancements in digital radiography, computerized tomography, optical scanning, 3D printing, and CAD/CAM milling have all factored into the proliferation of technology in dentistry. The clinician's ability to plan cases before ever anesthetizing the patient has led to a significant surge in digital smile designs using prosthetically driven approaches. The implementation of these technologies seems limitless as practitioners continue to embrace new devices, hardware, and software upgrades. These advancements are aimed at providing greater accuracy, enhanced diagnostic quality, improved patient acceptance, and, ultimately, a better patient experience.
Digital technology has long impacted dental implantology. Cone-beam computed tomography (CBCT), which was first introduced in 1999 and found its way into the US market in 2001,1 has become a preferred method for diagnosis and treatment planning in all fields of dentistry, especially dental surgery.2,3 Although advances in CBCT units, including various fields of view, artifact reduction tools and filters, and reduced radiation, have enabled better diagnostic quality, the technology is still not always ideal for tooth-borne surgical templates. The addition of optical scanning, whether it be extra- or intraoral, has allowed planners to combine standard tessellation languages (STL) and digital radiography (digital imaging and communications in medicine [DICOM] data) for improved diagnostics and treatment. This has enabled computer-aided guided implantology through the superimposition of these two components to obviate the inherent noise and scatter with CBCT scans, providing enhanced planning capabilities.
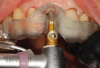
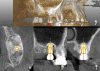
After planning has been completed, a traditional 3D-printed static osteotomy guide may be fabricated (Figure 1).4 Static surgical guides can be tooth-borne, mucosal-borne, or bone-borne. The use of these technologies can help reduce common human errors, eliminating potential harm to the recipient,5 and, in conjunction with diagnostics such as implant stability meters, allow for same-day delivery of prefabricated provisional teeth.
While static 3D-printed surgical guides have risen in popularity, the extent of implant placement done utilizing this technology is still quite small when compared to freehanded surgery. One reason for this may be the amount of time required for planning and printing surgical templates. Dynamic navigation has dramatically altered the state-of-the-art by providing clinicians with the accuracy of static surgical guides without the time and extra cost of printing or milling these templates, and it does not require the use of special drills. Dynamic navigation has been implemented in various medical procedures, including neurological, spinal, orthopedic, and ear/nose/throat surgical procedures. It can be used in various dental applications such as endodontics,6 anesthesia,7 periodontics, and oral surgery.
Dynamic Navigation in Dentistry
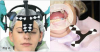
In dentistry, the concept of dynamic navigation involves the utilization of a "virtual template" preplanned in navigational treatment planning software. A specific tracking device is attached to the patient and the surgical handpiece. A micron-tracking camera captures the relation of the surgical handpiece to the patient's jaw (Figure 2 and Figure 3) and provides real-time continuous feedback to the operator. This feedback gives information as to the anterior-posterior-medial-lateral position of the drill as well as its depth, made possible with a special calibration process. The clinician is then able to complete the osteotomy and place the implant based on a preplanned position.
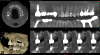
Dynamic navigation is not limited to the drilling of implant osteotomies. Surgical devices, such as piezo surgical units, can be used for sinus window access, bone plate harvesting, ridge splitting, bone reduction, and more. High-speed and low-speed handpieces, as well as subsequent drills, can be calibrated and used to plan specific surgical procedures, such as partial extraction therapy (Figure 4),8 a technique that involves retaining a fragment of tooth while preserving the remaining alveolar bone following tooth extraction.9,10
The use of dynamic navigation offers numerous advantages when compared to static 3D-printed or CAD/CAM-fabricated surgical guides for implant surgical templates. Virtual surgical procedures can be planned and treated in just a few minutes when circumstances permit. The operator's access and vision are unobstructed because the need for the physical surgical template is removed. A standard surgical kit can be used versus having to purchase and learn a system-dependent guided surgical kit. Alterations to the planned surgery can be made at the clinician's discretion on-demand at any time. Costs of surgical guides are eliminated. Finally, dynamic navigation delivers real-time verification of implant position accuracy.11
Many limitations of earlier navigational units have been overcome. Previous versions required a radiographic stent to be taken at the time of radiographic capture. Delays in the feedback or lag time between the computer image and hand movement have been improved as computer processing technology continues to increase exponentially. Software and hardware enhancements to central processing unit (CPU) processors, graphic user interfaces, and memory on graphic cards continue to boost speed. Accessories such as trackers have become more comfortable to use. Tracking unit carts have become more compact and ergonomic. Streamlined digital workflows, including use of prefabricated provisionals from single to full-arch restorations, have been incorporated in many softwares.12
The Process of Dynamic Navigation
The basic process across most dynamic navigation units in dentistry is similar and involves the following steps: radiographic capture utilizing CBCT; intraoral scanning (optional but highly recommended for improved accuracy of calibration) to reduce scatter/noise from CBCT13; planning the implant surgery with the navigation unit's software; attaching the jaw tracking device; and calibrating surgical tracking devices, handpieces, and drills.
Once these steps have been accomplished, the clinician can begin the operation while viewing the computer monitor for directional improvements. At this point, the procedure is similar to using a global positioning service (GPS) in a car or on a mobile device. In short, dynamic navigation could be considered "Google Maps" for implants.
Regarding ease of use of virtual dynamic navigation units during surgery, the general recommendation is for clinicians to find a unit that fits their needs, including size, flexibility within software, and support; cost may also be a factor. Training can be completed in as little as a day, or, depending on the user's comfort level, it might take a few weeks to gain confidence and security in trusting the technology. For those who are experienced in the gaming community, picking up this technology may come more naturally and be quickly adapted. For some, the use of the technology can become second nature after just a few surgeries.14
The following two clinical cases illustrate the use of dynamic navigation in dental implant placement.
Case 1: Maxillary Posterior Implant
A 63-year-old healthy man presented with an edentulous site in the maxillary right second molar region. The patient initially had elected to not have the tooth replaced when extracted. After 1 year he developed a parafunctional habit of chewing only with his left side and reconsidered his earlier decision.
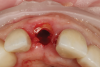
Upon evaluation of the CBCT using a metal artifact reduction (MAR) tool it was determined that either a short implant be placed or an internal sinus elevation procedure be performed to allow placement of a more robust implant. The CBCT showed less than 6 mm of residual bone height (Figure 5) available before the Schneiderian membrane of the maxillary sinus would be breached. After a review of available options, the patient elected to have the sinus elevation done and a longer implant placed with the use of guided surgery. Due to the limited access in the posterior of the oral cavity, the use of a static surgical guide might have proved difficult because of spatial and visual constrictions. Therefore, dynamic virtual navigation was chosen for this procedure.
After anesthetization of the patient and elevation of a full-thickness flap, the navigational unit was introduced with a calibrated pilot bur using the trace and place (TAP) technique.15 (Editor's note: The dynamic navigation system described here is the Navident system [ClaroNav, claronav.com]. Other dynamic navigation systems include X-Guide Dynamic 3D Navigation [X-Nav, x-navtech.com], Image Guided Implant [IGI] Dentistry System [Image Navigation, image-navigation.com], and YOMI® [Neocis, neocis.com].) Utilizing the feedback from the navigational unit the pilot drill was stopped short of the sinus membrane by 1 mm. To accomplish the internal sinus elevation, the concept of osseodensification (OD) was employed.16,17 OD is a dynamic nonsubtractive bone instrumentation method that enhances bone density through compaction autografting while preserving mineral content. This concept involves the use of specially designed burs with flutes in a counterclockwise fashion to develop a hydrodynamic compression wave. After the maxillary sinus autografting was completed, the osseodensification sinus lift protocol was utilized as outlined in the OD protocols.18 In short, the protocol involves measuring bone height to the sinus floor, using the 2 mm bur in OD mode until reaching the sinus floor, using the 3 mm bur in OD mode to enter up to 3 mm past the sinus floor, and, if necessary, using the 4 mm and 5 mm burs in OD mode to further expand the osteotomy. Burs are used in full-step increments, eg, 2 mm, 3 mm, 4 mm, and 5 mm, and the last bur is used in low speed to gently propel well-hydrated allograft material. The compression wave allows for autogenous bone to be compacted apically into the sinus cavity without perforating the Schneiderian membrane.
In the present case, successive burs were used in counterclockwise fashion continuing navigational guidance 1 mm short of the sinus floor until the 3 mm diameter bur was reached. Using the 3 mm diameter bur, the haptic feedback of the bur reaching the dense sinus floor was felt. Pressure was modulated with a pumping motion advancing past the sinus floor in 1 mm increments, up to but not exceeding 3 mm (Figure 6). The osteotomy was filled with synthetic putty bone and widened to 4 mm with slight apical pressure, with care taken not to exceed 3 mm past the sinus floor. Approximately 0.75 cc of synthetic material was used and the membrane was elevated allowing for placement of a 4.5 mm x 10 mm implant. Torque of 45 Ncm was achieved, and an implant stability quotient (ISQ) unit was used to determine implant stability; a reading of 71 was recorded. Based on these readings the implant was subsequently loaded with a healing abutment and sutured.
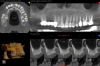
Upon completion of the surgery a postoperative CBCT was taken to evaluate the sinus membrane and ensure integrity. The navigational unit's software was used to determine accuracy of the planned implant position versus the surgical outcome (Figure 7). A small deviation consisting of 0.32 mm on entry, 0.34 mm at apex, and angle of 2.28 degrees was observed. The postoperative period was uneventful.
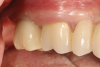
After 2 months of healing allowing for osseointegration, the ISQ was rechecked and a significant increase in value of 80 was achieved, indicating the implant was ready for restoration. The implant fixture was then scanned using an intraoral scanner and scanbody. A final screw-retained crown was milled and delivered 1 week later. At a 3-year follow-up (Figure 8 and Figure 9), a CBCT was taken due to additional treatment required on the contralateral side. Evaluation showed evidence of a new sinus floor and a healthy surrounding alveolar housing.
Case 2: Fixed Prosthesis
A 57-year-old healthy man returned to the author's office with four existing implants that had been placed in the maxilla 5 years prior for an implant-retained overdenture. The patient now desired to improve his quality of life with a fixed prosthesis. Because the fixed option originally was declined 5 years ago, the implants were placed in positions that were ideal for a removable appliance instead of a fixed prosthesis.
CBCT evaluation indicated that all four implants were healthy, and a decision was made to place two more implants. The plan called for one implant to be placed on each side to create an all-on-six solution. In the maxillary right quadrant, an implant would be placed between the two existing implants. The complexity of maintaining 3 mm of adequate space between each implant in the right quadrant for hygiene maintenance and adequate blood supply necessitated the use of guided surgery, as failure to maintain the 3 mm inter-implant spacing can result in severe bone loss and peri-implantitis.19In the left quadrant an implant was planned posterior to the most distal implant, thus requiring the implementation of the OD sinus lift protocol described in Case 1 (Figure 10).
Using the TAP method for calibration, the existing implants were used as markers for the navigational unit (Figure 11). Full-thickness flaps were elevated on both planned implant site locations. Pilot drills and all subsequent drills were calibrated for navigational surgery. Two implants were placed with adequate torque of 40+ Ncm and ISQ values of 80 and 79, respectively, enabling second staging of implants with healing abutments (Figure 12). The surgical sites were sutured and allowed to heal.
After 7 weeks the ISQ values were retested and substantial increases in values of 83 and 82, respectively, were noted. The patient's existing denture was used for a traditional conversion, which was accomplished utilizing multi-unit abutments and temporary cylinders while attempting to maintain the patient's current vertical dimension of occlusion (VDO). Approximately 3 weeks were allotted for the patient to adjust to his new fixed provisional. After determining that VDO, phonetics, and comfort were acceptable to the patient, the process for a final prothesis was initiated.
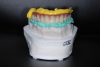
An iJig (a combined fit verification jig and final prosthetic proposal designed for easy, digital transfer of full-arch records)20 (Figure 13) was created by scanning with an intraoral scanner the converted provisional with scan analogs along with a tissue scan and counter model scan. Ten days later a 3D-printed polymethyl methacrylate (PMMA) try-in was delivered, and adjustments were made as needed. Vinyl polysiloxane (VPS) impression material was used to capture the intaglio surface and record occlusal registration. The case was returned to the laboratory for processing.
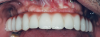
The final prosthetic was evaluated using a case check 3D viewer tool (Figure 14). After 2 weeks a final prosthesis was delivered (Figure 15 and Figure 16). At a follow-up visit the patient recorded proper occlusion, excursive movements, and comfort with the new prosthetics.
Conclusion
Due in large part to committed dedication to technological advancements, dynamic navigation has become a proven alternative to traditional static guided surgery modalities. Based on the same diagnosis and treatment planning foundation provided by CBCT 3-dimensional imaging, dynamic navigation can deliver the precision and accuracy of conventional static guided surgery. The many benefits of the "virtual template" include enhanced speed and delivery of implants without the added time and cost related to production of physical guides or the need to purchase a guided surgical drill kit. A standard surgical drill kit can be used regardless of manufacturer. Complex procedures such as partial extraction therapy, full-arch reconstruction, and sinus elevation can be completed accurately using this technology. In fact, dynamic navigation may be preferable for these procedures due to improved access and planning. The ability to receive real-time feedback and immediate verification of the surgical planning is the key to improving surgical outcomes by "staying on target," thus helping to enrich the overall patient experience.
About the Author
Isaac Tawil, DDS, MS
MINEC Ambassador (Megagen International Network of Educators and Clinicians); Member of MINEC USA; Member of Board of Directors, Digital Dental USA Society; Diplomate, International Academy of Dental Implantology and International Academy for Dental Facial Esthetics; Fellow, International Congress of Oral Implantologists; Private Practice, Brooklyn, New York
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Hatcher DC. Operational principles for cone-beam computed tomography. J Am Dent Assoc. 2010;141(suppl 3):3S-6S.
2. Tyndall DA, Price JB, Tetradis S, et al, American Academy of Oral and Maxillofacial Radiology. Position statement of the American Academy of Oral and Maxillofacial Radiology on selection criteria for the use of radiology in dental implantology with emphasis on cone beam computed tomography. Oral Surg Oral Med Oral Pathol Oral Radiol. 2012;113(6):817-826.
3. Ganz SD. CT scan technology - an evolving tool for predictable implant placement and restoration. Int Magazine of Oral Implantology. 2001;1:6-13.
4. Ganz SD. Presurgical planning with CT-derived fabrication of surgical guides. J Oral Maxillofac Surg. 2005;63(9 suppl 2):59-71.
5. Ganz SD. Brain-guided implant reconstruction: who makes the decisions? CAD/CAM International Magazine of Digital Dentistry. 2017;8(3):26-31.
6. Jain SD, Saunders MW, Carrico CK, et al. Dynamically navigated versus freehand access cavity preparation: a comparative study on substance loss using simulated calcified canals. J Endod. 2020;S0099-2399(20)30578-1.
7. Jain SD, Carrico CK, Bermanis I, Rehil S. Intraosseous anesthesia using dynamic navigation technology. J Endod. 2020;46(12):1894-1900.
8. Chen JT. A novel application of dynamic navigation system in socket shield technique. J Oral Implantol. 2019;45(5):409-415.
9. Ganz SD, Tawil I, Mitsias ME. The root membrane concept: in the zone with the "triangle of bone." DentalCEToday. 2017;36:80-84.
10. Ganz SD, Tawil I. Guided applications for partial extraction therapy. Dentistry Today. September 1, 2020. https://www.dentistrytoday.com/implants/10692-guided-applications-for-partial-extraction-therapy. Accessed June 29, 2021.
11. Block MS, Emery RW. Static or dynamic navigation for implant placement: choosing the method of guidance. J Oral Maxillofac Surg. 2016;74(2):269-277.
12. Stefanelli LV, Mandelaris GA, Franchina A, et al. Accuracy of dynamic navigation system workflow for implant supported full arch prosthesis: a case series. Int J Environ Res Public Health. 2020;17(14):5038.
13. Franchina A, Stefanelli LV, Maltese F, et al. Validation of an intra-oral scan method versus cone beam computed tomography superimposition to assess the accuracy between planned and achieved dental implants: a randomized in vitro study. Int J Environ Res Public Health. 2020;17(24):9358.
14. Jorba-García A, Figueiredo R, González-Barnadas A, et al. Accuracy and the role of experience in dynamic computer guided dental implant surgery: an in-vitro study. Med Oral Patol Oral Cir Bucal. 2019;24(1):e76-e83.
15. Stefanelli LV, Mandelaris GA, DeGroot BS, et al. Accuracy of a novel trace-registration method for dynamic navigation surgery. Int J Periodontics Restorative Dent. 2020;40(3):427-435.
16. Huwais S, Meyer EG. A novel osseous densification approach in implant osteotomy preparation to increase biomechanical primary stability, bone mineral density, and bone-to-implant contact. Int J Oral Maxillofac Implants. 2017;32(1):27-36.
17. Tanello B, Huwais S, Tawil I, et al. Osseodensification protocols for enhancement of primary and secondary implant stability - a retrospective 5-year follow-up multi-center study. Clin Oral Implants Research. 2019;30(S19):414.
18. Huwais S, Mazor Z, Ioannou AL, et al. A multicenter retrospective clinical study with up-to-5-year follow-up utilizing a method that enhances bone density and allows for transcrestal sinus augmentation through compaction grafting. Int J Oral Maxillofac Implants. 2018;33(6):1305-1311.
19. Tarnow DP, Cho SC, Wallace SS. The effect of inter-implant distance on the height of inter-implant bone crest. J Periodontol. 2000;71(4):546-549.
20. Ganz SD, Tawil I. Full-arch implant surgical and restorative considerations: innovative digital workflow using a verification jig with teeth. Dentistry Today. January 1, 2020. https://www.dentistrytoday.com/implants/10640-full-arch-implant-surgical-and-restorative-considerations-innovative-digital-workflow-using-a-verification-jig-with-teeth. Accessed June 29l, 2021.