You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Traditional orthodontic therapy relies on the preexisting alveolar bone volume present, and when the boundaries of the alveolus are exceeded during tooth movement, gingival recession may occur. In fact, the odds ratio for gingival recession in adolescent patients undergoing orthodontic therapy compared with patients that were not treated was 4.48.1 This consequence is a result mainly of a discrepancy involving the tooth and available alveolar bone, where bone volume needed to accommodate these teeth in their final position is limited.2,3 Generally, de-crowding in a deficient jawbone results in teeth being moved into a more labial direction, further compromising the facial bony plate, associated blood supply, and long-term gingival health and integrity.4 Mandibular incisor teeth seem to be the most vulnerable to this occurrence.5 In an attempt to avoid this phenomenon and enable an increased envelope of orthodontic movement, combined surgical-orthodontic therapies have been proposed.
Since the original surgical-orthodontic technique was described by Wilcko,6 several variations have been reported that have encompassed osseous injury and bone augmentation procedures with active tooth movement thereafter. These pre-orthodontic surgical techniques include, among others, surgically facilitated orthodontic treatment (SFOT), pre-orthodontic periodontal augmentation, piezocision, and periodontally accelerated osteogenic orthodontics. They are aimed at increasing volume of bone, into which the teeth can move, and may reduce the incidence of induced gingival recession. The objective of SFOT is two-fold: enhance and thicken the periodontal biotype (both hard and soft tissue), and facilitate accelerated tooth movement through the induction of the regional acceleratory phenomenon (RAP).7
Although enhancing the patient's tissue biotype is a primary goal of SFOT, in order to avoid possible complications and failed outcomes, special considerations should be made to address and overcome anatomical limitations and etiological factors related to thin biotype and its associated compromised blood supply. Historically, the term "regeneration" has been applied when grafting is performed around teeth; it implies a re-formation of periodontal ligament, bone, and cementum. In this article, however, the authors instead will use the term "generation" to describe the formation of alveolar bone that was not originally present or is significantly thin. For all of the surgical-orthodontic combined techniques described in the literature, there is very little information discussing specific factors associated with enhanced regenerative outcomes.
The aim of this article is to review different variables and critical factors that may affect the predictability of generating buccal bone during SFOT. In addition, optional modifications to enhance regenerative outcomes of SFOT will also be discussed.
Based on experience, from the authors' perspective the various factors impacting the final regenerative outcome can generally be classified into three main categories: patient related, orthodontically related, and surgically and biomaterial-related. Each category includes critical variables that should be considered when planning and executing this type of procedure, which is intended to augment alveolar housing. An outline of each category, its main variables, and their impact on the outcome, as described in the literature, is provided. (Editor's note: This article discusses patient-related and orthodontic-related factors associated with thin alveolar bone and tissue; a second article, tentatively scheduled to be published later this year, will describe surgically and biomaterial-related factors linked to increased predictability of bone augmentation during SFOT.)
Patient-Related Factors
There are multiple factors related to a patient's maxillofacial and dentoalveolar features that not only impact alveolar dimension but also may play an important role in reducing treatment predictability if left unaddressed. These include type of malocclusion, vertical facial pattern, and profile.
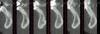
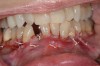
Vertical facial pattern has been shown to be a determining factor in mandibular symphysis alveolar morphology as well as lower incisor positioning for both class I and class III patients.8 Also, it has been shown that short-faced class III patients have a widened alveolar bone.8 However, for long- and normal-faced class III patients, there is a natural compensation that elongates the symphysis and influences incisor position.8 Often this compensation is not only associated with thin alveolar bone but also may influence the strain and pull of the mentalis (Figure 1 through Figure 6).
Specific impact of muscle pressure on bone resorption has previously been shown where, after growth completion, muscle pressure exerted a resorptive effect on bone.9 Patients that were undergoing a forward-upward genioplasty, and therefore having the muscle pull released, showed an increased thickness of and more bone apposition above the symphysis.10
Regarding malocclusion, a relationship also exists between the inclination of the lower incisors and the alveolar bone morphology.11 For example, patients with proclined lower incisors that are attempting to compensate for a class II relationship would show thin buccal bone. Therefore, if during orthodontic treatment an upright movement is executed without consideration of bone availability and root angulation, a thin, compromised bone thickness may result. In addition, the predictability of any augmentation procedures to increase thickness of bone may be reduced because of the lack of blood supply and cells.
Periodontal Biotype
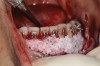
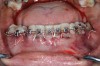
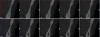
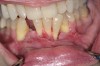
The association between gingival biotype (thickness) and buccal bone thickness has already been demonstrated through the use of direct measurement and cone-beam computed tomography (CBCT) analysis in cadaver heads.12 The importance of biotype as it relates to the outcome of SFOT has to do with its inherent available osseous and mucosal blood supply (Figure 7 through Figure 9). Osseous blood supply associated with various bone thicknesses and its impact on final outcome and predictability will be discussed in the next section (Alveolar Bone Thickness and Tooth Position). As to gingival/mucosal blood supply, it has been demonstrated that the major blood supply to the gingival tissue originates both from the supra-periosteal region and within the alveolar process itself.13 Therefore, if gingival blood supply is compromised due to deficient alveolar bone in a thin biotype, not only will there be an increased risk for future gingival recession but also compromised blood supply to support the regenerative process and wound healing. From classic literature associated with root coverage, thicker flaps bearing more favorable gingival blood supply have shown an increased predictability for complete root coverage.14
Based on these studies, predictability of generating the buccal plate and enhancing bone and soft-tissue thickness in thin biotype throughout SFOT may be reduced due to a limited mucosal blood supply to nourish both the biomaterial barrier and the flap itself. The use of multiple biomaterials should be considered in compromised thin tissue to avoid tissue sloughing and graft exposure. (Selection of biomaterials related to predictability will be discussed further in the aforementioned tentatively scheduled second article.)
Alveolar Bone Thickness and Tooth Position
For any regenerative procedure, the provision of cells, nutrients, and oxygen through existing blood supply is detrimental.15,16 Blood supply, in contrast to other important though modifiable factors, is the only critical factor that is pre-determined and directly associated with baseline alveolar anatomy. Thus, it is crucial to assess baseline trabecular bone versus cortical thickness and density using a CBCT, especially in the anterior mandible.17,18
Previous classifications of alveolar bone thickness and bony dehiscences have been offered to predict future risk of recession and its association with potential tooth movement. Classifications by both Richman and Evans have suggested a 1 mm to 2 mm bone thickness to minimize risk.2,19 Another classification system differentiates between crestal and radicular bone, allowing for proper planning for orthodontic treatment and future tooth positioning and identifying specific areas of need for augmentation to maximize post-therapy buccal bone thickness.20 This approach can also be adopted for case selection and predicting maximum alveolar bone post-SFOT, while combining grafting procedures with guided tooth movement.19 From a tooth movement perspective, orthodontic-related factors are discussed below. But from a regenerative perspective, a compact bone overlying a thin trabecular layer has a negative effect on blood supply and bone regeneration (Figure 7 and Figure 8).
Case selection related to baseline anatomy of alveolar bone is critical and is the initial step in determining predictability. In the author's opinion, in thin compromised cases modifications may include the use of soft-tissue augmentation only, with a minimally invasive approach, and/or the incorporation of proliferative agents and biologics, such as platelet-rich fibrin or platelet-derived growth factor-ßß, to stimulate or enhance angiogenesis.21
Depth of Vestibule
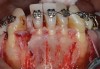
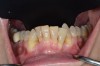
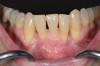
In general, the rate of developing soft-tissue complications related to bone augmentation procedures can range from 0% to 45%.22 In the anterior mandible any regenerative procedure that requires coronal positioning and primary closure can be challenging due to several anatomical factors (Figure 10 through Figure 13). These include the mentalis muscle pull; the depth of the vestibule, which should allow for primary closure without much tension on the lip; and the thickness of tissue, which should enable the primary closure to be maintained without dehiscence that may result in bone and membrane exposure.
In compromised situations where the depth of the vestibule is shallow or a prominent muscle pull exists, several complications can occur, including suboptimal closure with excessive tension around the lower lip area; soft-tissue dehiscence and opening of flaps resulting in loss of biomaterial that may compromise bone augmentation outcome and thickness during the healing phase and tooth movement; risk of infection due to exposure of bone and membrane/acellular dermal matrix, if used; and worsening of gingival recession due to soft-tissue dehiscence and loss of thickness at grafted sites.
Possible modifications in a patient with a shallow vestibule and prominent mentalis include preliminary vestibule deepening with the use of a free gingival graft or a Botox® injection to relieve muscle pull.
Orthodontic-Related Factors
Factors related to orthodontics that affect the regenerative outcome of SFOT primarily have to do with themechanics of tooth movement relative to bone availability. As stated earlier and has been shown in the literature, orthodontic tooth movement can increase a patient's risk for gingival recession.1 Because gingival recession may result from lack of available bone, orthodontic tooth movement that is done without consideration of bone availability, or at the expense of it, can further contribute to an existing risk of thin-tissue biotype. On the other hand, tooth movement that is planned based on the ideal tooth position within ideal bone availability not only can reduce this risk but also enhance stability and bone thickness.
Evans et al discussed different types of mechanics of orthodontic tooth movement that result in various tooth alignments and bone availability. They also offered a subclassification of pre-orthodontic tooth position to allow better diagnosis and risk analysis for long-term stability of tissues post-orthodontic movement.19
In light of this, CBCT examination of the dentoalveolar complex should include evaluation of the pretreatment and projected final positions of the tooth within bone.19 This assessment will serve as a blueprint for identifying patients with anatomic risk factors for attachment loss before the initiation of tooth movement. In addition, this evaluation may help in determining the appropriate orthodontic mechanics and type of movement needed to not only maximize bone availability by the end of treatment but also allow more available bone to provide blood supply and cells when augmentation procedures are performed.
In animal models it has been shown that bone graft material does not impede orthodontic tooth movement, but instead the graft material resorbs more readily with tooth movement.20 Therefore, orthodontic treatment provides a benefit in enhancing metabolism of the bone graft material.
The RAP is another important factor related to bone augmentation around teeth undergoing orthodontic movement.6,23 The RAP not only allows for rapid tooth movement but also is a biological event that will support bone metabolism.6 Aside from mechanics mentioned above, there are two main considerations for orthodontic treatment: the timing of treatment to achieve maximum metabolism of bone biomaterial and the extent of tooth movement needed. As shown in the literature, after the application of orthodontic force, the bone matrix remineralizes, in which tooth movement is induced and bone and graft material is remodeled.6,20 After corticotomies, a demineralization of the alveolus occurs around the involved teeth, and during tooth movement the collagen matrix of bone is transported with the tooth.6 Therefore, it is critical that tooth movement is initiated within 2 weeks of the bone augmentation procedure to maximize enhancement of regenerative potential related to the RAP effect.6
Before the surgery takes place, it is also important to receive input from the orthodontist regarding the extent of tooth movement needed to correct the malocclusion. This will dictate the amount of augmentation necessary to compensate for the expected dehiscence that is associated with the new tooth position.6,24,25
Conclusion
For more than a decade SFOT has been advocated in treatment of patients demonstrating crowded dentition and/or tooth malalignment. However, before orthodontic therapy is initiated, the etiology behind any gingival recession should be evaluated and understood. Also, prior to orthodontic treatment, the spatial limitations of the baseline alveolar anatomy should be considered, and if the projected tooth movement is expected to exceed these boundaries, SFOT with alveolar augmentation may be appropriate. Etiological factors associated with thin alveolar bone and tissue that frequently lead to dehiscences and fenestrations should be diagnosed and addressed before surgical procedures are carried out. The main factors that need to be understood and addressed are the muscle pull, a prominent symphysis, and a short vestibule. Also, the analysis of 3D images is critical not only for tooth movement relative to bone availability, but also for factors that are correlated with thin alveolar bone. Collaboration among the interdisciplinary treatment team is critical for communicating and planning future tooth movement and the extent of it.
Acknowledgment
The authors recognize and thank the orthodontists involved with their team in the treatment of the cases shown in this article: Paul G. Trotter, DMD; Eladio DeLeon, Jr, DMD, MS; and Steven C. Ricci, DDS, MS.
About the Authors
Stuart Beauchamp, DMD
Third Year Resident, Department of Periodontics, Augusta University Dental College of Georgia, Augusta, Georgia
Jacob K. Stern, DMD, MSc
Associate Professor, Department of Periodontics, Augusta University Dental College of Georgia, Augusta, Georgia
Colin S. Richman, DMD
Private Practice, Roswell, Georgia
William Baldock, DMD
First Year Resident, Department of Periodontics, Augusta University Dental College of Georgia, Augusta, Georgia
Brock J. Pumphrey, DMD
Private Practice, Atlanta, Georgia
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Renkema AM, Fudalej PS, Renkema AA, et al. Gingival labial recessions in orthodontically treated and untreated individuals: a case-control study. J Clin Periodontol. 2013;40(6):631-637.
2. Richman C. Is gingival recession a consequence of an orthodontic tooth size and/or tooth position discrepancy? "A paradigm shift." Compend Contin Educ Dent. 2011;32(1):62-69.
3. Richman CS. Dental space deficiency syndrome: an anthropological perspective. Compend Contin Educ Dent. 2017;38(3):180-186.
4. Steiner GG, Pearson JK, Ainamo J. Changes of the marginal periodontium as a result of labial tooth movement in monkeys. J Periodontol. 1981;52(6):314-320.
5. Renkema AM, Fudalej PS, Renkema A, et al. Development of labial gingival recessions in orthodontically treated patients. Am J Orthod Dentofacial Orthop. 2013;143(2):206-212.
6. Wilcko WM, Wilcko T, Bouquot JE, Ferguson DJ. Rapid orthodontics with alveolar reshaping: two case reports of decrowding. Int J Periodontics Restorative Dent. 2001;21(1):9-19.
7. Frost HM. The regional acceleratory phenomenon: a review. Henry Ford Hosp Med J. 1983;31(1):3-9.
8. Molina-Berlanga N, Llopis-Perez J, Flores-Mir C, Puigdollers A. Lower incisor dentoalveolar compensation and symphysis dimensions among Class I and III malocclusion patients with different facial vertical skeletal patterns. Angle Orthod. 2013;83(6):948-955.
9. Hoyte DA, Enlow DH. Wolff's law and the problem of muscle attachment on resorptive surfaces of bone. Am J Phys Anthropol. 1966;24(2):205-213.
10. Chamberland S, Proffit WR, Chamberland PE. Functional genioplasty in growing patients. Angle Orthod. 2015;85(3):360-373.
11. Yu Q, Pan XG, Ji GP, Shen G. The association between lower incisal inclination and morphology of the supporting alveolar bone-a cone-beam CT study. Int J Oral Sci. 2009;1(4):217-223.
12. Fu JH, Yeh CY, Chan HL, et al. Tissue biotype and its relation to the underlying bone morphology. J Periodontol. 2010;81(4):569-574.
13. Rateitschak KH, Rateitschak EM, Wolf HF, Hassell TM. Color Atlas of Periodontology. 2nd ed. Stutgart, Germany: Georg Thieme Verlag; 1992.
14. Baldi C, Pini-Prato G, Pagliaro U, et al. Coronally advanced flap procedure for root coverage. Is flap thickness a relevant predictor to achieve root coverage? A 19-case series. J Periodontol. 1999;70(9):1077-1084.
15. Wang HL, Boyapati L. "PASS" principles for predictable bone regeneration. Implant Dent. 2006;15(1):8-17.
16. Susin C, Wikesjö UM. Regenerative periodontal therapy: 30 years of lessons learned and unlearned. Periodontol 2000. 2013;62(1):232-242.
17. Garaicoa C, Suarez F, Fu JH, et al. Using cone beam computed tomography angle for predicting the outcome of horizontal bone augmentation. Clin Implant Dent Relat Res. 2015;17(4):717-723.
18. Mandelaris GA, Neiva R, Chambrone L. Cone-beam computed tomography and interdisciplinary dentofacial therapy: an American Academy of Periodontology Best Evidence Review focusing on risk assessment of the dentoalveolar bone changes influenced by tooth movement. J Periodontol. 2017;88(10):960-977.
19. Evans M, Tanna NK, Chung CH. 3D guided comprehensive approach to mucogingival problems in orthodontics. Semin Orthod. 2016;
22(1):52-63.
20. Araújo MG, Carmagnola D, Berglundh T, et al. Orthodontic movement in bone defects augmented with Bio-Oss. An experimental study in dogs. J Clin Periodontol. 2001;28(1):73-80.
21. Dohle E, El Bagdadi K, Sader R, et al. Platelet-rich fibrin-based matrices to improve angiogenesis in an in vitro co-culture model for bone tissue engineering. J Tissue Eng Regen Med. 2018;12(3):598-610.
22. Lim G, Lin GH, Monje A, et al. Wound healing complications following guided bone regeneration for ridge augmentation: a systematic review and meta-analysis. Int J Oral Maxillofac Implants. 2018;33(1):41-50.
23. Yaffe A, Fine N, Binderman I. Regional accelerated phenomenon in the mandible following mucoperiosteal flap surgery. J Periodontol. 1994;
65(1):79-83.
24. Coscia G, Coscia V, Peluso V, Addabbo F. Augmented corticotomy combined with accelerated orthodontic forces in class III orthognathic patients: morphologic aspects of the mandibular anterior ridge with cone-beam computed tomography. J Oral Maxillofac Surg. 2013;71(10):
1760.e1-e9.
25. Mandelaris GA, DeGroot BS, Relle R, et al. Surgically facilitated orthodontic therapy: optimizing dentoalveolar bone and space appropriation for facially prioritized interdisciplinary dentofacial therapy. Compend Contin Educ Dent. 2018;39(3):146-156.