You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Despite advances in scientific knowledge, improved hygiene regimens, and the ever-increasing availability of new commercial preventative formulations, dental caries continues to be a major public health problem in most countries. The process of enamel demineralization begins at the atomic level on the hydroxyapatite crystal surface and can continue, unless halted, with the endpoint being evidence of caries and cavitation of enamel.1,2 Left unchecked, early demineralization at the atomic and molecular levels can progress to changes in enamel morphology and enamel lesion development, followed by dentinal involvement, and, ultimately, obvious cavitated lesions.2,3 The pathophysiology of dental caries underlies a process whereby the mineral components of the tooth—largely calcium and phosphate—are demineralized and removed by the action of organic acids produced by bacteria, which reside in the biofilm, adhering to the tooth surface.3 As a result, calcium and phosphate ions are lost from the subsurface enamel, resulting in an early, subsurface enamel lesion. As illustrated in Figure 1, this early carious lesion is reversible via the process of remineralization, whereby “calcium and phosphate ions are supplied from a source external to the tooth to promote ion deposition into crystal voids in demineralized enamel to produce net mineral gain.”4
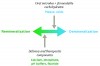
In the past two decades, caries research has been focused on the development of methodologies for remineralization of carious lesions. Therapeutic treatment approaches to caries can now be divided into three basic phases, based on degree of lesion involvement: early treatment – using remineralization techniques; mid or intermediate treatment – using a combination of remineralization and minimally invasive restorative treatment; and late stage treatment – using definitive restorative techniques. While new and developing caries diagnostic devices have emerged,5,6 systematic reviews of these devices appear to indicate some limitations in their detection of early or incipient carious lesions.7 However, as the sensitivity and specificity of the diagnostic information of these new devices continues to improve, clinicians will have additional longitudinal patient data concerning the level of demineralization and the caries status of individual lesions. This will serve as an important information feedback loop (Figure 2) in the clinical application of new remineralizing agents and will provide a basis for the development of improved remineralizing agents. Additionally, devices and products for caries risk assessment continue to emerge and be refined, offering clinicians important information about individual caries risk and susceptibility. The availability of improved diagnostic information concerning the presence of early caries and demineralization, as well as caries risk, complements and enables the use of the remineralization technology discussed in this review.
Further Rationale for Remineralization Technology
Saliva’s natural ability to remineralize demineralized enamel crystals relates to its ability to supply bioavailable calcium and phosphate ions to the tooth.4 At physiological pH, normal saliva is supersaturated with respect to both ionic and non-ionic forms of calcium (phosphate and bicarbonate, hydroxyapatite, and fluorapatite), with ionic calcium comprising about 50% of the total calcium concentration.8,9 The presence in saliva of specialized salivary proteins, such as salivary phosphoproteins, enables binding of calcium phosphate ion clusters, retarding their growth to the critical size necessary for precipitation and transformation into a crystalline phase.8 This critical stabilization of calcium and phosphate ions by salivary phosphoproteins ensures that ions remain bioavailable to diffuse into mineral-deficient lesions to allow for remineralization of demineralized crystals while preventing surface deposition in the form of calculus or nonuseable surface agglomerates. Thus, it appears that natural mechanisms of remineralization use not only the inorganic mineral elements of calcium and phosphate, but also organic components such as phosphopeptides and phosphoproteins to mediate and control the remineralization process.
Nevertheless, early on, researchers discovered that the net mineralization by saliva is small and appears to be limited mainly to the surface layer of the lesion.10 Clinical support for this early in-vitro finding has developed recently based on evidence that white-spot lesions present after removal of orthodontic appliances and, detected and measured by quantitative light-induced fluorescence, remained stable, with no evidence of regression, even after 2 years.11,12 These researchers concluded that new remineralization systems would be needed to achieve more pronounced lesion regression.
Developments continue in technology, methods, and fundamental scientific knowledge regarding the ability to promote remineralization. Fluoride is known to promote remineralization but is dependent on calcium and phosphate ions from saliva as critical ion sources. Recent investigations, technologies, and new products have primarily focused on various calcium phosphate-based compositions, which are designed to supplement and enhance fluoride’s ability to restore tooth mineral.13
Before examining several of these new compositions for remineralization, an attempt to organize or classify the basic categories of remineralizing agents can be helpful. In examining several recent reviews of new and emerging approaches to tooth remineralization,2,4,14 a basic classification of remineralizing agents can be suggested. Mineral- or ion-based remineralizing agents can be divided into these categories: exclusively fluoride-based; non-fluoride-based, ie, calcium-, phosphate-, or calcium-phosphate-based [organic, inorganic, crystalline, amorphous]); or combination products containing both a fluoride-based and non-fluoride-based component. Recent research indicates that an additional mechanism provided by these new non-fluoride remineralizing agents may be the ability to modify or buffer the acid pH of the biofilm microenvironment present in demineralization to a more alkaline pH range.15
Before considering both non-fluoridated remineralizing agents and compositions that combine non-fluoridated remineralizing agents with fluoride, a review of the role of fluoride as a remineralizing agent is helpful.
Fluorides
An important adjunct in the prevention of dental caries, fluorides are introduced into the oral environment via personal (eg, dentifrices, rinses) or professional (eg, varnishes, foams, gels, fluoride-releasing restorative materials) applications. Fluoride levels of about 3 parts per million (ppm) in the enamel are required to shift the balance from net demineralization to net remineralization.16
Several mechanisms have been suggested to achieve the anti-caries effects of fluoride, including: the formation of fluorapatite, which is more acid-resistant than hydroxyapatite; the enhancement of remineralization; interference of ionic bonding during pellicle and plaque formation; and the inhibition of microbial growth and metabolism.17 Remineralization may be enhanced by providing low levels of calcium and phosphate in conjunction with minimal amounts of fluoride. The difference that a very small amount of fluoride (< 1 ppm) has on demineralization and remineralization is remarkable. This is because fluoride acts as a catalyst and influences reaction rates with dissolution and transformation of various calcium phosphate mineral phases within tooth structure and plaque adjacent to tooth surfaces. Incorporating minimal amounts of fluoride into hydroxyapatite (HAP) yields fluorohydroxyapatite (FHAP), which resists demineralization at a level similar to fluoroapatite (FAP).3 The concentration of fluoride applied intraorally also appears to have a significant influence on the degree of remineralization of advanced (> 150 microns) enamel lesions. This suggests treatments with 5000 ppm F—the concentration found in a fluoride varnish—significantly enhance remineralization and inhibit demineralization when compared to treatments with 1500 ppm F.18
While fluoride is highly effective on smooth-surface caries, it is less so on pit and fissure caries. Moreover, a high-fluoride strategy has potential for overexposure and adverse effects (eg, fluorosis). Also, when environmental sources of fluoride are present, it is suggested that fluoride exposure be limited.2,19 Given the limitations of both natural mechanisms (eg, saliva) and fluorides to induce remineralization,2,19,20 additional new remineralizing agents can be envisioned along with their performance requirements (Table 1).
Non-Fluoride Remineralizing Agents
Non-fluoride-based remineralizing agents can be classified as either calcium-based or non-calcium-based compounds or compositions. The non-calcium-based technologies focus primarily on pH modification through microenvironmental buffering effects within the saliva, surface biofilm, and/or enamel pellicle. An example includes xylitol, which is a non-fermentable, non-acid-producing sugar substitute that serves to maintain biofilm pH above a level that would promote demineralization.2 The use of chewing gum carrying xylitol increases salivary flow rate and enhances the protective properties of saliva. Because the concentration of bicarbonate and phosphate is higher in stimulated saliva, there is a resultant increase in plaque pH and salivary buffering capacity, which prevents demineralization of tooth structure. Moreover, the resultant higher concentration of calcium, phosphate, and hydroxyl ions in saliva may also enhance remineralization. Likewise, the action of the trimetaphosphate ion (TMP) to enhance enamel and dentin remineralization is also currently an area of active research interest.2 The focus of this review, however, will be the following calcium-based remineralizing agents: amorphous calcium phosphate (ACP); casein phosphopeptide (Recaldent); functionalized tricalcium phosphate (fTCP); particulate bioglass (NovaMin); and nano-hydroxyapatite.
Calcium-Based Remineralizing Agents
Amorphous Calcium Phosphate (ACP)
Amorphous calcium phosphate (ACP) is the initial solid phase that precipitates from a highly supersaturated calcium phosphate solution and can convert readily to stable crystalline phases such as octacalcium phosphate or apatitic products.21 The ACP technology was developed by Dr. Ming S. Tung at the ADA Paffenbarger Research Center/National Institute of Standards & Technology in Gaithersburg, Maryland.2 In 1999, ACP was incorporated into a toothpaste called Enamelon, which is no longer available commercially, and was reintroduced in 2004 in Enamel Care™ toothpaste (Church & Dwight). The initial ACP technology required a two-phase delivery system to keep the calcium and phosphorous components from reacting to each other before use.2 The calcium and phosphates salts, usually calcium sulfate and dipotassium phosphate, were supplied in separate compartments prior to mixing. When the two salts were mixed, they rapidly formed the ACP complex that could then precipitate onto the tooth surface and be incorporated in the surface dental plaque. This precipitated ACP could then readily dissolve in the saliva, providing calcium and phosphate for tooth remineralization.22 Single-component formulations of ACP were subsequently developed, most often through the use of anhydrous, yet water-soluble, carrier polymers for delivery of the calcium and phosphate salts to the saliva and tooth surface.
ACP has been used in tooth-whitening agents to prevent demineralization and reduce post-bleaching sensitivity. It has also been incorporated into prophy pastes and fluoride varnish.2 The incorporation of ACP into a fluoride varnish (Enamel Pro® Varnish, Premier Dental, www.premusa.com) appears to significantly enhance fluoride uptake by both normal and demineralized enamel.23 ACP is also used in the Aegis® product line (Harry J. Bosworth Co., www.bosworth.com), including its pit and fissure sealant.2 An experimental ACP composite has shown to efficiently establish mineral ion transfer throughout the body of the lesion and restore the mineral lost due to acid attack.24 Therefore, incorporation of ACP in composite resins of various types may assist in the remineralization process; however, more evidence is needed.
Casein Phosphopeptide (CPP)/CPP-ACP
As noted earlier in this review, certain natural organic molecules play a key role in the remineralization process. Analogues molecules in the naturally occurring phosphopeptides and phosphoproteins in saliva have been discovered in the form of casein derivatives, which are phosphopeptides found in milk and milk products.25 Caseins are a heterogeneous family of proteins predominated by alpha 1 and 2 and b-caseins. Casein phosphopeptides (CPPs) are phosphorylated casein–derived peptides produced by tryptic digestion of casein.2,4,14,15,20 The CPP component, which contains the amino acid cluster sequence -Ser (P)-Ser (P)-Ser (P)-Glu-Glu-, has the ability to bind and stabilize calcium and phosphate in solution. CPP also can bind dental plaque and tooth enamel. CPP-ACP technology was developed by Prof. Eric Reynolds of the University of Melbourne. CPP-ACP, trademarked Recaldent™, has been launched in sugarless chewing gum and candy. More recently, a sugar-free, water-based cream/paste containing Recaldent (CPP-ACP) has been made available to dental professionals in the form of several products by GC Corp. ( www.gc-dental.com), including GC Tooth Mousse and ProSpec MI Paste, which are also available in the fluoride-containing form of CPP-ACP, ie, CPP-ACFP.2,14,25,26
Casein phosphopeptides have significant ability to bind both positively and negatively charged ions. Each molecule of CPP can bind up to 25 calcium ions, 15 phosphate ions, and five fluoride ions. The calcium phosphate in these complexes is biologically available for remineralization of subsurface lesions in tooth enamel.27,28 Complexes of CPP-ACP exist at nanoscale dimensions in the form of nanoclusters that are only 2 nm in diameter, as compared to larger nano-sized particles of calcium fluoride, which have been prepared with a diameter of some 41 nm.20 The ability to bind multiple ions of calcium, phosphate, and fluoride serves as the basis to combine CPP with ACP (and optionally fluoride as well) in the form of CPP-ACP, and CPP-ACFP. The role of ACP is also said to control the precipitation of CPP with calcium and phosphate ions.27 The advantage of CPP-ACFP is the availability of calcium, phosphate, and fluoride in one product.25
Through their multiple phosphoryl residues (terminal phosphate ion groups), the CPPs can bind to amorphous calcium in ACP to form clusters of ACP in metastable solution, thus preventing their growth to the critical size required for nucleation and precipitation. This binding mechanism prevents the premature loss of solubility of the calcium-phosphate complex, which thereby allows diffusion of calcium and phosphate within the surface layers of tooth structure. CPP-ACP is the acronym for a complex of casein phosphopeptides (CPPs) and amorphous calcium phosphate (ACP).
The proposed mechanism of anti-cariogenicity for the CPP-ACP is that it localizes ACP in dental plaque. This buffers the free calcium and phosphate ion activities, thereby helping to maintain a state of supersaturation with respect to tooth enamel, thereby depressing demineralization and enhancing remineralization. Casein phosphopeptides are used alone or, as noted above, as CPP-ACP. If the CPP-ACP is further complexed with fluoride, CPP-ACFP (casein phosphopeptides with amorphous calcium fluoride phosphate) results. With respect to its interaction with fluoride, the CPPs have been shown to keep fluoride ions in solution, thereby enhancing the efficacy of the fluoride as a remineralizing agent, with the end result being the formation of fluoroapatite.29,30 Nevertheless, the main function of casein phosphopeptides appears to be to modulate bioavailability of calcium phosphate levels by maintaining ionic phosphate and calcium supersaturation to increase remineralization.31,32 As such, CPP regulates the behavior of calcium and phosphate and stabilizes calcium phosphate compounds.20
CPP-ACP has shown to reduce demineralization and enhance remineralization of the enamel subsurface carious lesions.25 For example, in a study utilizing removable palatal appliances with four human-enamel, half-slab insets containing subsurface lesions, the incorporation of CPP-ACP into oral lozenges significantly increased enamel subsurface lesion remineralization by 78% to up to 176% (depending on dosage levels), relative to sugar-free lozenge control, when administered four times per day over a 14-day period.33 With respect to depth changes in subsurface enamel lesions due to remineralization induced by CPP-ACP, an in-vitro study demonstrated (as compared to controls cited below) statistically significant lesion depth reductions on the order of 10% when topically applied or used as a toothpaste, and 13% when topically applied in conjunction with a fluoridated toothpaste. A positive control using a fluoridated toothpaste decreased lesion depth by about 7%, and a negative control using a non-fluoridated toothpaste increased lesion depth significantly by 23%.34
CPP also is believed to have an antibacterial and buffering effect on plaque and interferes in the growth and adherence of Streptococcus mutans and Streptococcus sobrinus.15,32,35 CPP inhibits adherence of oral bacteria to saliva-coated hydroxyapatite beads (S-HA). By selectively inhibiting bacterial adhesion to teeth, conventional concepts concerning oral bacterial dynamics would suggest an alteration of the microbial composition of dental plaque, thus favoring establishment of fewer cariogenic species. This activity could also control acid formation (buffering) in dental plaque, in turn reducing hydroxyapatite dissolution from tooth enamel. The incorporation of CPP into the pellicle in exchange for albumin appears to inhibit the adherence of S. mutans and S. sobrinus, which may also cause both neutralization and enhancement of remineralization.32 Nevertheless, recent findings concerning the complexity of the dynamics of oral biofilms may to some extent mitigate or reduce the significance of the role of selective adherence of known cariogenic microorganisms. This changing view is due in part to more current caries biofilm theory that proposes the hypothesis that the genetic composition and the metabolic output of the biofilm should be the focus rather than individual species presence in the biofilm.36 Supportive of this new perspective is recent research suggesting that the adaptive ability of species previously considered commensal have been demonstrated to adapt and become acidogenic within 30 minutes of being exposed to low pH in the biofilm.37 In an animal caries model, it has been observed that CPP-ACP significantly reduced caries activity in a dose-dependent manner, as 1% CPP-ACP produced about a 55% reduction in smooth surface caries and a 46% reduction in fissure caries activity, which is similar in effect to that produced by 500 ppm of fluoride.14,38,39
Combined with fluoride, CPP-ACP has an additive effect on caries activity.27,28 Use of CPP-ACP along with a fluoride-containing dentifrice has proven to be beneficial in reducing the demineralization around orthodontic brackets and remineralizing white spots caused by demineralization.40,41Ramalingam et al42 found that adding CPP-ACP to soft drinks can reduce their erosion capacity. CPP-ACP has also been added to dentifrices, mouthrinses, chewing gums, lozenges, and bovine milk. A study by Walker et al43 found that although milk contains casein phosphate, the addition of CPP-ACP results in enhanced remineralization. A dose of 5 gm of CPP-ACP produced 148% more remineralization compared to 2 gm of CPP-ACP per liter of milk.
Though casein derivatives have been investigated as a remineralizing agent, some authors have reported insufficient clinical trial evidence to make a recommendation regarding the long-term effectiveness of casein derivatives in preventing caries in vivo.44 More recent trials also suggest a mixed picture with respect to the clinical effectiveness of CPP-ACFP in preventing or remineralizing white-spot lesions. Several clinical trials have demonstrated the ability of the CPP-ACFP technology to prevent and, to some degree, reverse spot lesions during orthodontic treatment.45,46 Conversely, some studies suggest no clinical advantage for the use of CPP-ACFP paste supplementary to normal oral hygiene over limited (several months) time periods.47,48
Surface-Activated Tricalcium Phosphate with Fluoride
Beta-tricalcium phosphate (β-TCP) can be coupled with organic and/or inorganic agents such as carboxylic acids and surfactants to create a “functionalized” tricalcium phosphate (f-TCP).49 β-TCP serves as a bioactive and relatively stable source of calcium and phosphate ions in fluoride-containing, water-based compositions. This technology has been licensed (3M ESPE) for use in high-concentration fluoride toothpastes and a fluoride varnish. Functionalized TCP is a unique technology involving mechano-chemical ball milling of tricalcium phosphate with simple organic ingredients, resulting in a functionalized or bioactive tricalcium phosphate.50 Tricalcium phosphate was selected because it appears as a transitional phase in hydroxyapatite conversion, is biocompatible and bioactive in the oral cavity, and contains sites within its structure that can be activated with, for instance, simple organic molecules. Research findings suggest that low levels of functionalized TCP are needed to produce mineral nucleation without negatively interacting with fluoride. Additionally, tricalcium phosphate has been formulated for several oral care products. For example, Vanish™ White Varnish (3M ESPE, www.3MESPE.com) contains 5% sodium fluoride with a functionalized tricalcium phosphate in an alcohol-based solution of modified rosin.51 In this product, which is sweetened with xylitol, the tricalcium phosphate is prepared by mechano-chemical ball milling of tricalcium phosphate with fumaric acid. The result is free phosphate and functionalized calcium oxide protected by fumaric acid.52 Similar tricalcium-based materials and their incorporation into topically applied oral hygiene products have been described in detail by Karlinsey and Mackey.53
In a published clinical study using human enamel blocks, acid-induced, artificial enamel lesions were bonded in place in human subjects using either a 225 ppm fluoride (F) mouthrinse or a mouthrinse containing 225 ppm F plus a functionalized tricalcium phosphate complexed with β-TCP that was surface-modified with silica (Si) and urea (Ur) (TCP-Si-Ur).54 The results of this 28-day study indicated that the mouthrinse containing 225 ppm F plus TCP-Si-Ur provided significantly greater remineralization relative to 225 ppm F only or saliva alone.
Calcium Sodium Phosphosilicate (Bioactive Glass)
Calcium sodium phosphosilicate is an inorganic compound that reacts in aqueous environments to release calcium, sodium, and phosphate ions over time. Originally developed as a bone regenerative material, this compound has been shown to be effective at physically occluding dentinal tubules through the development of a hydroxyapatite-like mineral layer.55,56 NovaMin® (NovaMin Technology Inc., a subsidiary of GlaxoSmithKline, www.gsk.com), a commercial trade name for the synthetic mineral composed of calcium, sodium, phosphorous, and silica, is a form of calcium sodium phosphosilicate that releases deposits of crystalline hydroxycarbonate apatite (HCA) structurally, like tooth mineral composition. Clinical evaluations of NovaMin for the treatment of dentin hypersensitivity have shown statistically significant and clinically positive results.57
When bioactive glass comes in contact with saliva, there is a rapid release of sodium, calcium, and phosphorous ions into the saliva that are available for remineralization of the tooth surface. The released ions form hydroxycarbonate apatite directly. They also attach to the tooth surface and continue to release ions and remineralize the tooth surface after the initial application. These particles have been shown to release ions and transform into HCA for up to 2 weeks. Ultimately, these particles completely transform into HCA.58,59
A recent investigation examined the potential remineralization effect of topical NovaMin and sodium fluoride gel on caries-like lesions in permanent teeth. NovaMin dentifrice (Oravive revitalizing toothpaste containing NovaMin) appeared to have a greater effect on remineralization of carious-like lesions in permanent teeth when compared to that of a fluoride-containing dentifrice (Topex® Take Home Care® 1.1% neutral sodium fluoride gel [Sultan Healthcare, www.sultanhc.com]) containing 0.5% fluoride ion (which is equal to 5000 ppm fluoride).60
There is also limited evidence that the NovaMin technology displays antimicrobial activity, with possible anti-gingivitis clinical effects.61 Currently available products are marketed under the following trade names: NovaMin®; Topex® Renew™ Paste (Sultan Healthcare); DenShield™ (NovaMin Technology Inc.); NuCare™ Root Conditioner with NovaMin® (Sunstar Butler, www.sunstar.com); NUPRO® Sensodyne® Prophylaxis Paste with NovaMin® and NUPRO® Sensodyne® Professional Toothpaste with NovaMin® (a dentist-dispensed toothpaste with 5000 ppm fluoride and NovaMin for use at home) (DENTSPLY Professional, www.nupro-sensodyne.com); and Sylc™ therapeutic prophy powder (OSspray Ltd., www.osspray.com).
Nano-Hydroxyapatite (Nano-HA)
A number of studies have been conducted to determine the effect of nano-hydroxyapatite-containing toothpastes on initial enamel lesions under dynamic pH-cycling conditions.62-64 These studies concluded that nano-HA has the potential to remineralize initial enamel lesions. A concentration of 10% nano-HA may be optimal for remineralization of early enamel caries.65 Likewise, some preliminary evidence suggests that the addition of nano-HA to sports drinks may enhance resistance to demineralization as well as remineralization of early enamel lesions.66
Summary and Conclusion
Both opportunities and challenges lie ahead in the implementation of new remineralization technology. The availability and continued development of new agents and methods for remineralization of early and incipient enamel lesions offers new therapeutic options and benefits to the dental profession. Critical to further growth will be enhanced devices and techniques for diagnosing and monitoring incipient carious lesions and areas of early tooth demineralization. These devices and methods will need to be highly efficacious and reliable, as well as cost-effective. As improved remineralization techniques are clinically established, modes of reimbursement should provide improved financial incentives for the increased time and expertise allocated to such advanced preventive services. Likewise, additional viable treatment options will become available to clinicians as more effective modes of therapeutic delivery of remineralizing agents are developed and new products are designed to deliver multiple agents simultaneously to increase both the quantity and quality of remineralized apatite mineral.
DISCLOSURE
The author has no affiliation with any products mentioned in this article.
REFERENCES
1. Featherstone JD. Dental caries: dynamic disease process. Aust Dent J. 2008;53(3):286-291.
2. Goswami M, Saha S, Chaitra TR. Latest developments in non-fluoridated remineralizing technologies. J Indian Soc Pedod Prev Dent. 2012;30(1):2-6.
3. Hicks J, Garcia-Godoy F, Flaitz C. Biological factors in dental caries: role of remineralization and fluoride in the dynamic process of demineralization and remineralization (part 3). J Clin Pediatr Dent. 2004;28(3):203-214.
4. Cochrane NJ, Cai F, Huq NL, et al. New approaches to enhance remineralization of tooth enamel. J Dent Res. 2010;89(11):1187-1197.
5. Guerrieri A, Gaucher C, Bonte E, Lasfargues JJ. Minimal intervention dentistry: part 4. Detection and diagnosis of initial caries lesions. Br Dent J. 2012;213(11):551-557.
6. Rochlen GK, Wolff MS. Technological advances in caries diagnosis. Dent Clin North Am. 2011;55(3):441-452.
7. Twetman S, Axelsson S, Dahlén G, et al. Adjunct methods for caries detection: a systematic review of literature. Acta Odontol Scand. 2013;71(3-4):388-397.
8. Hay DI, Moreno EC. Statherin and the acidic proline-rich proteins. In: Tenovuo JO, ed. Human Saliva: Clinical Chemistry and Microbiology. Boca Raton, FL: CRC Press; 1989:131-150.
9. Fejerskov O, Kidd E. Dental Caries: The Disease and its Clinical Management. Oxford, England: Blackwell Munksgaard; 2003:56.
10. Silverstone LM. Remineralization of human enamel in vitro. Proc R Soc Med. 1972;65(10):906-908.
11. van der Veen MH, Mattousch T, Boersma JG. Longitudinal development of caries lesions after orthodontic treatment evaluated by quantitative light-induced fluorescence. Am J Orthod Dentofacial Orthop. 2007;131(2):223-228.
12. Mattousch TJ, van der Veen MH, Zentner A. Caries lesions after orthodontic treatment followed by quantitative light-induced fluorescence: a 2-year follow-up. Eur J Orthod. 2007;29(3):294-298.
13. Fejerskov O. Changing paradigms in concepts on dental caries: consequences for oral health care. Caries Res. 2004;38(3):182-191.
14. Rao A, Malhotra N. The role of remineralizing agents in dentistry: a review. Compend Contin Educ Dent. 2011;32(6):26-33.
15. Nongonierma AB, Fitzgerald RJ. Biofunctional properties of caseinophosphopeptides in the oral cavity. Caries Res. 2012;46(3):234-267.
16. Summit JB, Robbins JW, Schwartz RS, et al. Fundamentals of Operative Dentistry: A Contemporary Approach. 2nd ed. Chicago, IL: Quintessence Publishing; 2001:377-385.
17. Niessen LC, Gibson G. Oral health for a lifetime: preventive strategies for the older adult. Quintessence Int. 1997;28(9):626-630.
18. ten Cate JM, Buijs MJ, Miller CC, Exterkate RA. Elevated fluoride products enhance remineralization of advanced enamel lesions. J Dent Res. 2008;87(10):943-947.
19. Brown WE. Physicochemical mechanisms in dental caries. J Dent Res. 1974;53(2):204-216.
20. Walsh LJ. Contemporary technologies for remineralization therapies: a review. Int Dent SA. 2009;11(6):6-16.
21. Zhao J, Liu Y, Sun WB, Zhang H. Amorphous calcium phosphate and its application in dentistry. Chem Cent J. 2011;5:40.
22. Tung MS, Eichmiller FC. Dental applications of amorphous calcium phosphates. J Clin Dent. 2003;10(1 spec no):1-6.
23. Schemehorn BR, Wood GD, McHale W, Winston AE. Comparison of fluoride uptake into tooth enamel from two fluoride varnishes containing different calcium phosphate sources. J Clin Dent. 2011;22(2):51-54.
24. Langhorst SE, O’Donnell JN, Skrtic D. In vitro remineralization of enamel by polymeric amorphous calcium phosphate composite: quantitative microradiographic study. Dent Mater. 2009;25(7):884-891.
25. Aimutis WR. Bioactive properties of milk proteins with particular focus on anticariogenesis. J Nutr. 2004;134(4):989S-995S.
26. Llena C, Fomer L, Baca P. Anticariogenicity of casein phosphopeptide-amorphous calcium phosphate: a review of the literature. J Contemp Dent Pract. 2009;10(3):1-9.
27. Cross KJ, Hug NL, Palamara JE, et al. Physicochemical characterization of casein phosphopeptide-amorphous calcium phosphate nanocomplexes. J Biol Chem. 2005;280(15):15362-15369.
28. Reynolds EC. Remineralization of enamel subsurface lesions by casein phosphopeptide-stabilized calcium phosphate solutions. J Dent Res. 1997;76(9):1587-1595.
29. Rose RK. Effects of an anticariogenic casein phosphopeptide on calcium diffusion in streptococcal model dental plaques. Arch Oral Biol. 2000;45(7):569-575.
30. Reynolds EC. Calcium phosphate-based remineralization systems: scientific evidence? Aust Dent J. 2008;53(3):268-273.
31. Cross KJ, Huq NL, Reynolds EC. Casein phosphopeptides in oral health—chemistry and clinical applications. Curr Pharm Des. 2007;13(8):793-800.
32. Schüpbach P, Neeser JR, Golliard M, et al. Incorporation of caseinoglycomacropeptide and caseinophosphopeptide into the salivary pellicle inhibits adherence of mutans streptococci. J Dent Res. 1996;75(10):1779-1788.
33. Cai F, Shen P, Morgan MV, Reynolds EC. Remineralization of enamel subsurface lesions in situ by sugar-free lozenges containing casein phosphopeptide-amorphous calcium phosphate. Aust Dent J. 2003;48(4):240-243.
34. Kumar VL, Itthagarun A, King NM. The effect of casein phosphopeptide-amorphous calcium phosphate on remineralization of artificial caries-like lesions: an in vitro study. Aust Dent J. 2008;53(1):34-40.
35. Rose RK. Binding characteristics of Streptococcus mutans for calcium and casein phosphopeptide. Caries Res. 2000;34(5):427-431.
36. Nyvad B, Crielaard W, Mira A, et al. Dental caries from a molecular microbiological perspective. Caries Res. 2013;47(2):89-102.
37. Takahashi N, Nyvad B. The role of bacteria in the caries process: ecological perspectives. J Dent Res. 2011;90(3):294-303.
38. Reynolds EC, Cain CJ, Webber FL, et al. Anticariogenicity of calcium phosphate complexes of tryptic casein phosphopeptides in the rat. J Dent Res. 1995;74(6):1272-1279.
39. Gupta R, Prakash V. CPP-ACP complex as a new adjunctive agent for remineralisation: a review. Oral Health Prev Dent. 2011;9(2):151-165.
40. Sudjalim TR, Woods MG, Manton DJ, Reynolds EC. Prevention of demineralization around orthodontic brackets in vitro. Am J Orthod Dentofacial Orthop. 2007;131(6):705.e1-e9.
41. Andersson A, Sköld-Larsson K, Hallgren A, et al. Effect of a dental cream containing amorphous cream phosphate complexes on white spot lesion regression assessed by laser fluorescence. Oral Health Prev Dent. 2007;5(3):229-233.
42. Ramalingam L, Messer LB, Reynolds EC. Adding casein phosphopeptide-amorphous calcium phosphate to sports drinks to eliminate in vitro erosion. Pediatr Dent. 2005;27(1):61-67.
43. Walker G, Cai F, Shen P, et al. Increased remineralization of tooth enamel by milk containing added casein phosphopeptide-amorphous calcium phosphate. J Dairy Res. 2006;73(1):74-78.
44. Azarpazhooh A, Limeback H. Clinical efficacy of casein derivatives: a systematic review of the literature. J Am Dent Assoc. 2008;139(7):915-924.
45. Rollings S, Greene L, Borrie F, Lamont T. Small trial finds beneficial effect for MI Paste in preventing white spot lesions during orthodontic treatment. Evid Based Dent. 2012;13(4):117-118.
46. Robertson MA, Kau CH, English JD, et al. MI Paste Plus to prevent demineralization in orthodontic patients: a prospective randomized controlled trial. Am J Orthod Dentofacial Orthop. 2011;140(5):660-668.
47. Huang GJ, Roloff-Chiang B, Mills BE, et al. Effectiveness of MI Paste Plus and PreviDent fluoride varnish for treatment of white spot lesions: a randomized controlled trial. Am J Orthod Dentofacial Orthop. 2013;143
(1):31-41.
48. Beerens MW, van der Veen MH, van Beek H, ten Cate JM. Effects of casein phosphopeptide amorphous calcium fluoride phosphate paste on white spot lesions and dental plaque after orthodontic treatment: a 3-month follow-up. Eur J Oral Sci. 2010;118(6):610-617.
49. Karlinsey RL, Pfarrer AM. Fluoride plus functionalized β-TCP: a promising combination for robust remineralization. Adv Dent Res. 2012;24(2):48-52.
50. Tri-Calcium Phosphate (TCP), Espertise™ - A Collection of Scientific Results 2008–2011. St. Paul, MN: 3M ESPE; 2011.
51. Vanish™ 5% Sodium Fluoride White Varnish with Tri-Calcium Phosphate – Technical Product Profile. St. Paul, MN: 3M ESPE; 2010.
52. Karlinsey RL, Mackey AC, Walker ER, Frederick KE. Preparation, characterization and in vitro efficacy of an acid-modified beta-TCP material for dental hard-tissue remineralization. Acta Biomater. 2010;6(3):969-978.
53. Karlinsey RL, Mackey AC. Solid-state preparation and dental application of an organically modified calcium phosphate. J Mater Sci. 2009;44(1):346-349.
54. Amaechi BT, Karthikeyan R, Mensinkai PK, et al. Remineralization of eroded enamel by a NaF rinse containing a novel calcium phosphate agent in an in situ model: a pilot study. Clin Cosmet Investig Dent. 2010;2:93-100.
55. Andersson OH, Kangasniemi I. Calcium phosphate formation at the surface of bioactive glass in vitro. J Biomed Mater Res. 1991;25(8):1019-1030.
56. Hench LL, Andersson Ö. Bioactive glasses. In: Hench LL, Wilson J, eds. An Introduction to Bioceramics. Singapore: World Scientific Publishing Co.; 1993:45-47.
57. Burwell AK, Litkowski LJ, Greenspan DC. Calcium sodium phosphosilicate (NovaMin®): remineralization potential. Adv Dent Res. 2009;21(1):35-39.
58. Du M,Q Tai BJ, Jiang H, et al. Efficacy of dentifrice containing bioactive glass (NovaMin) on dentine hypersensitivity [abstract]. J Dent Res. 2004;83:13-15. Abstract 1546.
59. Burwell A, Jennings D, Muscle D, Greenspan DC. NovaMin and dentin hypersensitivity—in vitro evidence of efficacy. J Clin Dent. 2010;21(3):66-71.
60. Vahid Golpayegani M, Sohrabi A, Biria M, Ansari G. Remineralization effect of topical NovaMin versus sodium fluoride (1.1%) on caries-like lesions in permanent teeth. J Dent (Tehran). 2012;9(1):68-75.
61. Tai BJ, Bian Z, Jiang H, et al. Anti-gingivitis effect of a dentifrice containing bioactive glass (NovaMin) particulate. J Clin Periodontol. 2006;33(2):86-91.
62. Najibfard K, Ramalingam K, Chedjieu I, Amaechi BT. Remineralization of early caries by a nano-hydroxyapatite dentifrice. J Clin Dent. 2011;22(5):139-143.
63. Tschoppe P, Zandim DL, Martus P, Kielbassa AM. Enamel and dentine remineralization by nano-hydroxyapatite toothpastes. J Dent. 2011;39(6):430-437.
64. Huang S, Gao S, Cheng L, Yu H. Remineralization potential of nano-hydroxyapatite on initial enamel lesions: an in vitro study. Caries Res. 2011;45(5):460-468.
65. Huang SB, Gao SS, Yu HY. Effect of nano-hydroxyapatite concentration on remineralization of initial enamel lesion in vitro. Biomed Mater. 2009;4(3):034104.
66. Min JH, Kwon HK, Kim BI. The addition of nano-sized hydroxyapatite to a sports drink to inhibit dental erosion: in vitro study using bovine enamel. J Dent. 2011;39(9):629-635.
ABOUT THE AUTHOR
Steven R. Jefferies, MS, DDS, PhD
Professor, Donald and Cecelia Platnick Professor, and Director of Biomaterials Research Laboratory, Restorative Dentistry, and Director of Clinical Research, Maurice H. Kornberg School of Dentistry, Temple University, Philadelphia, Pennsylvania
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.