You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Periodontitis is chronic and inflammatory. Approximately 35% of US adults older than 30 years of age have periodontitis.1 According to a recent assessment of the prevalence of periodontal disease from 1999 to 2004 using the National Health and Nutrition Examination Survey, the overall prevalence of moderate and severe forms of periodontal disease was 0.82% to 18.3% in adults 20 to 34 years of age and 0.06% to 2.9% in adults 75 years or older.2 For aggressive periodontitis, the prevalence is approximately 2.1% to 2.6% in US African Americans and approximately 0.09% to 0.17% in US Caucasians.3,4 Chronic periodontitis is characterized by an oral bacterial infection leading to inflammation within the supporting tissues of the teeth, which often leads to the destruction of the periodontal tissues and alveolar bone that support the teeth.5,6 This oral inflammation often has systemic effects in which there is an increased concentration of circulating inflammatory markers with the severity of disease being correlated directly with levels of serum inflammatory markers.7 Levels of serum C-reactive protein and other biomarkers of inflammation are higher in patients with periodontal disease than those with no existing periodontal disease.8,9 Periodontal infection has been linked to organ and systemic diseases, such as cardiovascular disease, diabetes mellitus, and adverse pregnancy outcomes.10,11 What is unknown is the link or the possible link between periodontal disease and cancer, and whether this link is associated with overall cancer risk or specific tumor sites. This article reviews the possible link between periodontal infection and solid tumor and hematologic cancers, which include pancreatic cancer, lung cancer, and leukemia.
The Inflammatory Response
The immune system reacts to harmful stimuli by a hyperinflammatory response. Acute inflammation is the initial response in which there is an increase in plasma proteins and leukocytes from the blood to the site of injured tissues. Chronic inflammation is characterized by an increase in mononuclear immune cells, tissue destruction, and attempts at repair with fibrosis and angiogenesis.12 Inflammation must be regulated tightly by the body so that it works to heal as opposed to leading to a host of other diseases, such as atherosclerosis, or inappropriate or unintended tissue necrosis.
Cancer or a malignant neoplasm is marked by the uncontrolled growth of cells, tissue invasion, and metastasis to various organs via the hematologic and lymphatic systems. Nearly all cancers are caused by abnormalities of the genetic material of the transformed cells. These abnormalities often are caused by carcinogens, such as tobacco smoke, radiation, chemicals, or infectious agents. Cancers are classified by the primary type of tissue from which they originate, and prognosis is determined by the type of cancer and the stage or the extent of disease.13
Inflammation is a critical component in the tumor progression. Interaction between the uncontrolled neoplastic cells, defensive host cells, and microenvironment allows the tumor to grow and disseminate. The inflammatory response that is observed near the developing tumor may regulate many aspects of the tumor development by providing mediators needed in maintaining tissue homeostasis.10 Inflammation may enhance cellular proliferation and mutagenesis, reduce adaptation to oxidative stress, promote angiogenesis, inhibit apoptosis, and increase the secretion of inflammatory mediators.6 Therefore, it is prudent to explore the possible relationship between the inflammation of periodontal disease and cancer.
Pancreatic Cancer Risk
Pancreatic cancer often is diagnosed in a late stage, contributing to the poor survival rate. Cigarette smoking is a consistent risk factor that has been identified.16 Other risk factors that have been suggested include consumption of processed or smoked meats, colonization of Helicobacter pylori, and a history of diabetes.15 A prospective study using data from the Alpha-Tocopherol, Beta-Carotene Cancer Prevention (ATBC) cohort study conducted in Finland reported a positive association between tooth loss and pancreatic cancer. Tooth loss was not found to be associated significantly with Helicobacter pylori seropositivity; however, the subjects of this study were older male smokers. Potential confounders were added to individual statistical models in a stepwise fashion. None of the smoking history variables altered the risk estimates. Other potential confounders were: age, height, weight, body mass index (BMI), urban living, education, history of gallstones, pancreatitis, peptic and duodenal ulcers or diabetes, dietary energy, energy-adjusted carbohydrate, fat, saturated fat, folate, and alcohol. Age confounded the risk estimate of the association, but the other potential confounders did not.16 Another study using data from the National Health and Nutrition Examination Survey (NHANES I) population found that individuals with gingivitis, periodontitis, and edentulism had an elevated risk of death from cancer, with a suggestion of an association for pancreatic cancer. However, adjustment of potential confounders, such as smoking habits and alcohol consumption, reduced this association.17 A prospective study using the data from the Health Professionals Follow-Up Study (HPFS) of male health professionals found an increased pancreatic cancer risk in those with a self-reported history of periodontal disease compared with no history of periodontal disease.6 The number of teeth at baseline was not associated with pancreatic cancer; however, tooth loss during the past 4 years was associated more strongly with pancreatic cancer than cumulative tooth loss. When periodontal disease was analyzed jointly with recent tooth loss, there was a 2.7-fold increase in pancreatic cancer compared with no periodontal disease and no recent tooth loss. This result suggests that timing and severity of periodontal disease play a role in this association. The association with periodontal disease appeared to be stronger among men who were never smokers as well as in men with a BMI less than 25 kg/m2, implying that smoking and obesity are unlikely explanations for the association, but the association seemed similar among younger and older participants, implying that age is an unlikely confounder. When a history of diabetes was excluded from the analysis, a statistically significant association for periodontal disease was still observed. Statistical models were stratified by age and calendar time. They were adjusted for cigarette smoking, diabetes, baseline BMI, race, height, physical activity, non-steroidal anti-inflammatory drug use, multivitamin use, geographic regions, and history of cholecystectomy. Dietary factors were controlled for. Income and socioeconomic status data were not collected and could have confounded the association. The authors acknowledged that a limitation of the study was that the periodontal disease instances were self-reported.6
Despite the data gained by these prospective analyses, more work is needed to confirm the findings and explore the biologic mechanisms. Several authors are exploring the hypothesis that periodontal disease may promote pancreatic carcinogenesis through inflammation or through the generation of carcinogens, such as nitrosamines. Nitrosamines and gastric acidity have been thought to play a role in pancreatic cancer.15,16,20 However, these hypotheses do not imply or prove a causality, but rather there may be a casual relationship.
Lung Cancer Risk
The association between periodontal disease and lung cancer has been controversial because there is a well-established, strong association with cigarette smoking and the risk of lung cancer.31 Through analyzing data from the NHANES I population, Hujoel et al17 found that periodontitis was associated significantly with total cancer death, specifically lung cancer. The association was decreased as the analysis was adjusted for other confounders, such as poverty index and vitamins A and C. When the analysis was limited to never smokers, no association between periodontal disease and lung cancer was identified. Another study looking at the data from the Glasgow Alumni Cohort also did not find an association between tooth loss and lung cancer, both with and without adjustment for baseline smoking status. Participants were adjusted for age at examination, gender, father’s socioeconomic position, smoking status, BMI, and systolic blood pressure.18
The possible association between periodontal disease and lung cancer may be confounding. Periodontal disease may capture an unmeasured aspect of smoking history. Periodontal disease is associated with other smoking-related diseases, such as stroke.17 Plus, the relative magnitude of the periodontitis-systemic disease association mimics the magnitude of the smoking-systemic disease association, suggesting that periodontitis is a surrogate marker for the unmeasured aspect of smoking and mimics the effects of smoking. Because periodontal inflammation is distant from the lung tissues, it is unlikely that the inflammatory infiltrate travels and concentrates into the lung tissues and causes cancer, making the biologic plausibility remote.17 To see if an association can be confirmed, more studies are needed in a population that has a lower smoking prevalence.
Upper Gastrointestinal Cancer Risk
Many studies have looked at the association between tooth loss and upper gastrointestinal cancers, including cancers of the upper digestive tract, esophagus, and stomach. A Japanese case-control study, which looked at 242 cases with gastric cancer and 484 cancer-free controls matched for gender, age, and place of residence, observed a significant dose–response relationship between the odds of developing gastric cancer and the number of missing teeth. Stress, long working time, irregularity in sleeping time, and gray hair in men displayed high odds ratios.19 Two studies looked at the people of Linxian, China, which has some of the highest rates of esophageal and gastric cardia cancer in the world. In their first study,20 the authors looked at 29,584 cancer-free individuals aged 40 to 69 years and found a significantly elevated risk of esophageal squamous cell carcinoma, gastric cardia adenocarcinoma, and gastric noncardia adenocarcinoma. They noted this increased risk was associated most strongly with the loss of the first few teeth and primarily was confined to the younger members of the cohort. Age, gender, tobacco use, and alcohol use always were included in the statistical models. They proposed four possible mechanisms in which tooth loss could increase the risk for upper gastrointestinal cancers: 1) tooth loss is a marker for poorer general health and is not in itself a risk factor; 2) loss of teeth leads to alterations in dietary patterns, which can elevate the risk of cancer; 3) tooth loss affects chewing and swallowing, which can contribute to esophageal cancer by causing irritation or damage to the esophageal epithelium; and 4) tooth loss increases the risk of upper gastrointestinal cancers through alternations in the oral bacterial flora, leading to an increased exposure and in vivo production of nitrosamines. A follow-up study of the same cohort21 found a significant relationship between tooth loss and risk of dying from upper gastrointestinal cancer with age as an effect modifier. This elevated risk was present in male smokers, male nonsmokers, and females who were nearly all nonsmokers. The statistical models controlled for age, gender, smoking, drinking, height, weight, and systolic blood pressure. This population has limited access to preventive dentistry and, if further studies show a causal relationship between periodontitis and cancer mortality, dental public health interventions may prevent early death. A third study by Abnet et al22 looked prospectively at the ATBC study cohort, which consisted of 29,133 eligible men aged 50 to 69 years. On comparing edentulous subjects with individuals with < 10 teeth lost, the authors found a significant two-fold increase in the risk of gastric noncardia adenocarcinoma. However, no statistically significant associations were found between tooth loss and esophageal squamous cell carcinoma or esophageal/gastric cardia adenocarcinoma. Adjustments for confounding factors (dietary factors, smoking, and H pylori) did not explain these results. Other oral bacteria also may play a role in this type of cancer through similar inflammatory mechanisms. More studies are needed with better control of smoking and H pylori infection.
Oral Cancer Risk
Oral hygiene has been considered a potential risk factor for oral cancers. Tobacco smoking and alcohol consumption are the main risk factors, and both are associated with inadequate oral hygiene and may be confounders of an association with tooth loss. There are six recent case control studies identifying the possible link between tooth loss, periodontal disease, and oral cancer. One study, conducted in Beijing,23 found that after adjustment for tobacco smoking and alcohol consumption, poor dentition reflected as missing teeth emerged as a strong risk factor and those with leukoplakia and lichen planus showed an elevated risk of oral cancers. They had included the following potential confounders: alcohol drinking, tobacco smoking, and years of education to indicate socioeconomic status. A study by Marshall et al24 confirmed earlier findings that cigarette smoking and alcohol consumption impart a substantial risk to oral cancers, but they also found that poor oral hygiene increases the risk of oral cancers. However, this risk is smaller than those of cigarette and alcohol use. Also, individuals who smoked cigarettes, drank alcohol, and lost teeth without replacements were at an increased risk for oral cancers. A study in Denmark25 looked at the risk of developing intraoral squamous cell carcinoma. The authors found a two-fold increase in cancer risk in persons with fewer than five teeth compared with persons with 15 or more teeth, after correcting for tobacco and alcohol consumption. Also, marital status and dental status remained significantly associated with the risk. Garrote et al,26 studying a population in Cuba, found that the number of missing teeth and poor general oral condition at oral inspection showed a 2.7- and a 2.6-fold increase in oral cancers, respectively. The participants were adjusted for age, gender, area of residence, education, and smoking and drinking habits. But a study in Italy27 did not show a significant association with increased cancer risk and missing 16 or more teeth. They included age, gender, smoking and drinking habits, and intake of fruits and vegetables. A study in Sweden28 investigating oral and oropharyngeal squamous cell carcinoma looked at potential risk factors, such as oral hygiene, dental status, oral mucosal lesions, alcohol and tobacco use, and virus infection, as well as some related to lifestyle. They found that average oral hygiene, poor oral hygiene, more than five defective teeth, more than 20 missing teeth, and defective complete dentures were significant risk factors for development of oropharyngeal squamous cell carcinoma. Regular dental care reduced this risk.28 Tooth loss and oral hygiene may contribute to oral carcinogenesis through interactions with cigarette and alcohol use. Also the magnitude of association with known risk factors may vary at different oral sites, such as the pharynx, larynx, and oral cavity; therefore, further studies should look at these specific sites.
A study by Michaud et al29 looked at whether periodontal disease or tooth loss is associated with cancer risk. Analyzing the HPFS population, the authors found that after adjustment for known risk factors, such as smoking and diet, those with a history of periodontal disease had an increased risk of total cancer compared with those without a history of periodontal infection. The statistical models were stratified by age and calendar time and included ethnic origin, physical activity, smoking history, pack years, history of diabetes, geographical region, height, alcohol, vitamin D score, calcium intake, fruit and vegetable intake, red meat consumption, and total caloric intake. After controlling for smoking and other risk factors, there were significant associations between periodontal disease and lung, kidney, pancreatic, and hematologic cancers. Fewer teeth at baseline (0 to 16) also were associated with an increase risk for lung cancer. In never smokers, periodontal disease was associated with significant increases in total and hematologic cancers, but no association was noted for lung cancer. Interestingly, an inverse association was found for the number of teeth and melanoma of the skin and advanced prostate cancer. However, this relationship should be interpreted with caution because this study looked at many different cancer sites and this finding might have been caused by chance. Also, the study did not adjust for sun exposure, a risk factor for melanoma of the skin, which hypothetically could be related to the number of teeth and be a confounding variable. Limitations of this study included the self-reporting of periodontal disease and the inadequate power to be able to examine the less common cancers. Three other studies looked at the relationship between periodontal disease and total cancer mortality with limited findings. Cabrera et al30 included the following confounders: age, waist-to-hip ratio, BMI, smoking, age at first birth, parity, and socioeconomic variable. Tu et al18 did not find a relation between tooth loss and cancer in their study cohorts. Hujoel et al17 found an increased risk of fatal cancer associated with periodontitis, gingivitis, and edentulism.
Discussion
There are many hypotheses for the possible association between periodontal disease and cancer, total and by site (Table 1). However, residual confounding effects from smoking must be taken into consideration because smoking is a known risk factor for many cancers as well as for periodontal disease and tooth loss. Diet and a history of diabetes also may play a confounding role and are known contributory factors for periodontal disease. But inflammation does play a role in both periodontal disease and cancer. The chronic inflammation induced by the periodontal pathogens may serve to promote initiated cells and may lead to breakdown of normal growth and possible carcinogenesis. The chronic inflammation also may be a sign of a lack of surveillance of tumor growth by the body. Lastly, the formation of endogenous nitrosamines by the bacteria is promoted by inadequate oral hygiene and periodontal disease.31
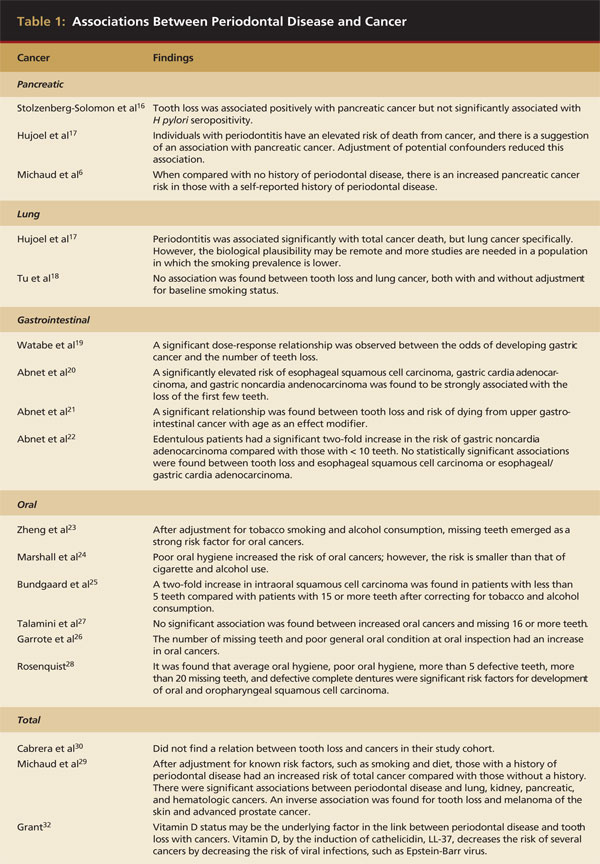
Grant32 has suggested that the underlying factor between the link of periodontal disease and tooth loss with cancer is low serum 25-hydroxyvitamin D (calcidiol) levels. Vitamin D decreases the risk of periodontal disease by the induction of human cathelicidin, LL-37 and, by this, decreases the risk of several cancers by decreasing the risk of viral infections, such as the Epstein-Barr virus.32
Conclusion
Clearly, further research is warranted to confirm many of the positive associations between periodontal disease and various cancers. As the role of periodontal disease in the etiology of cancers is further elucidated, possible biologic mechanisms can be explored. Periodontal disease may be a sign of a deficiency of the overall immune system or it may have a direct cause of cancer. Either way, some studies have shown a potential link between periodontal disease and cancer while others have not. The future challenge is to determine the nature of the link—causal or casual. Finally, the dental professional should advise patients with significant periodontal infection and tooth loss that a thorough evaluation by a physician may be warranted.
References
1. Albandar JM, Brunelle JA, Kingman A. Destructive periodontal disease in adults 30 years of age and older in the United States, 1988-1994 [published erratum appears in: J Periodontol. 1999;70(3):351]. J Periodontol. 1999,70(1):13-29.
2. Eke P, Baker L. Prevalence of periodontal disease in the United States: NHANES 1999-2004 [abstract]. 2007;86(spec iss A): 1843.
3. Melvin WL, Sandifer JB, Gray JL. The prevalence and sex ratio of juvenile periodontitis in a young racially mixed population. J Periodontol. 1991;62(5):330-334.
4. Löe H, Brown LJ. Early onset periodontitis in the United States of America. J Periodontol. 1991;62(10):608-618.
5. Lindhe J, Ranney R, Lamser I, et al. Consensus report: chronic periodontitis. Ann Periodontol. 1999;4(1)38-38.
6. Michaud DS, Joshipura K, Giovannucci E, et al. A prospective study of periodontal disease and pancreatic cancer in US male health professionals. J Natl Cancer Inst. 2007;99(2):171-175.
7. Amabile N, Susini G, Pettenati-Soubayroux I, et al. Severity of periodontal disease correlates to inflammatory systemic status and independently predicts the presence and angiographic extent of stable coronary artery disease. J Intern Med. 2008;263(6):644-652.
8. Loos BG. Systemic markers of inflammation in periodontitis. J Periodontol. 2005;76(11 suppl):2106-2115.
9. Joshipura KJ, Wand HC, Merchant AT, et al. Periodontal disease and biomarkers related to cardiovascular disease. J Dent Res. 2004;83(2):151-155.
10. Persson GR, Persson RE. Cardiovascular disease and periodontitis: an update on the associations and risk. J Clin Periodontol. 2008;35(8 suppl):362-379.
11. Mealey BL, Rose LF. Diabetes mellitus and inflammatory periodontal diseases. Curr Opin Endocrinol Diabetes Obes. 2008;15(2):135-141.
12. Cotran RS, Kumar V, Collins T. Robbins Pathologic Basis of Disease. 6th ed. Philadelphia, PA: WB Saunders Co; 1999:50-88.
13. Cotran RS, Kumar V, Collins T. Robbins Pathologic Basis of Disease. 6th ed. Philadelphia, PA: WB Saunders Co; 1999:260-327.
14. van Kempen LC, de Visser KE, Coussens LM. Inflammation, proteases and cancer. Eur J Cancer. 2006;42(6):728-734.
15. Risch HA. Etiology of pancreatic cancer, with a hypothesis concerning the role of N-nitroso compounds and excess gastric acidity. J Natl Cancer Inst. 20032;95(13):948-960.
16. Stolzenberg-Solomon RZ, Dodd KW, Blaser MJ, et al. Tooth loss, pancreatic cancer, and Helicobacter pylori. Am J Clin Nutr. 2003;78(1):176-181.
17. Hujoel PP, Drangsholt M, Spiekerman C, et al. An exploration of the periodontitis-cancer association. Ann Epidemiol. 2003;13(5):312-316.
18. Tu YK, Galobardes B, Smith GD, et al. Associations between tooth loss and mortality patterns in the Glasgow Alumni Cohort. Heart. 2007;93(9):1098-1103.
19. Watabe K, Nishi M, Miyake H, et al. Lifestyle and gastric cancer: a case-control study. Oncol Rep. 1998;5(5):1191-1194.
20. Abnet CC, Qiao YL, Mark SD, et al. Prospective study of tooth loss and incident esophageal and gastric cancers in China. Cancer Causes Control. 2001;12(9):847-854.
21. Abnet CC, Qiao YL, Dawsey SM, et al. Tooth loss is associated with increased risk of total death and death from upper gastrointestinal cancer, heart disease, and stroke in a Chinese population-based cohort. Int J Epidemiol. 2005;34(2):467-474.
22. Abnet CC, Kamangar F, Dawsey SD, et al. Tooth loss is associated with increased risk of gastric non-cardia adenocarcinoma in a cohort of Finnish smokers. Scand J Gastroenterol. 2005;40(6):681-687.
23. Zheng TZ, Boyle P, Hu HF, et al. Dentition, oral hygiene, and risk of oral cancer: a case-control study in Beijing, People’s Republic of China. Cancer Causes Control. 1990;1(3):235-241.
24. Marshall JR, Graham S, Haughey BP, et al. Smoking, alcohol, dentition and diet in the epidemiology of oral cancer. Eur J Cancer B Oral Oncol. 1992;28B(1):9-15.
25. Bundgaard T, Wildt J, Frydenberg M, et al. Case-control study of squamous cell cancer of the oral cavity in Denmark. Cancer Causes Control. 1995;6(1):57-67.
26. Garrote LF, Herrero R, Reyes RM, et al. Risk factors for cancer of the oral cavity and oro-pharynx in Cuba. Br J Cancer. 2001;85(1):46-54.
27. Talamini R, Vaccarella S, Barbone F, et al. Oral hygiene, dentition, sexual habits and risk of oral cancer. Br J Cancer. 2000;83(9):1238-1242.
28. Rosenquist K. Risk factors in oral and oropharyngeal squamous cell carcinoma: a population-based case-control study in southern Sweden. Swed Dent J Suppl. 2005;(179):1-66.
29. Michaud DS, Liu Y, Meyer M, et al. Periodontal disease, tooth loss, and cancer risk in male health professionals: a prospective cohort study. Lancet Oncol. 2008;9(6):550-558.
30. Cabrera C, Hakeberg M, Ahlqwist M, et al. Can the relation between tooth loss and chronic disease be explained by socio-economic status? A 24-year follow-up from the population study of women in Gothenberg, Sweden. Eur J Epidemiol. 2005;20(3):229-236.
31. Meyer MS, Joshipura K, Giovannucci E, et al. A review of the relationship between tooth loss, periodontal disease, and cancer. Cancer Causes Control. 2008;19(9):895-907.
32. Grant WB. Vitamin D, periodontal disease, tooth loss, and cancer risk. Lancet Oncol. 2008;9(7):612-613.

