You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Vitamin D is the collective name for cholecalciferol (vitamin D3) and ergocalciferol (vitamin D2). Cholecalciferol is formed in the skin by ultraviolet radiation inducing a photochemical reaction. Ergocalciferol is derived from plants. In order to be activated, both undergo hydroxylation at the 25 position in the liver by cytochrome P450 enzymes and then are further hydroxylated in the kidney in the 1a position, yielding the active metabolites: 1,25(OH)2D3 and 1,25(OH)2D2. These molecules render their biological effects by binding to the vitamin D receptor (VDR), which is a nuclear receptor highly expressed in organs involved in calcium homeostasis. The net effect of vitamin D is to increase both serum calcium and serum phosphate by stimulating intestinal absorption, bone resorption, and renal reabsorption.1,2 Vitamin D also facilitates phagocytosis by monocytes and monocyte differentiation. Epithelial cells and macrophages increase the expression of antimicrobial peptides (AMPs) on exposure to microbes, which is dependent on the presence of vitamin D.3 Vitamin D also has anti-inflammatory effects by suppressing pro-inflammatory cytokines, IFN-γ, TNF-α, and IL-12. It controls more than 200 genes that are responsible for the regulation of cellular proliferation, differentiation, apoptosis and angiogenesis.4
This paper will address the various roles vitamin D plays in reference to chronic diseases, including periodontitis. It will also discuss how vitamin D deficiency is diagnosed and treated, as well as its effects on other parts of the body besides the skeletal system.
Vitamin D Levels
Vitamin D is a fat-soluble vitamin that acts like a steroid hormone. Although it has long been known that it is essential for bone health, it also affects other organ systems as well as the immune and cardiovascular systems, muscles, and brain. Sources of vitamin D come from sun exposure and food; selected food sources of vitamin D are listed in Table 1.5 Factors such as skin color (amount of melanin that is expressed), age, fat content (being overweight or obese), and living in northern latitudes where sun exposure is usually minimal affect how much vitamin D the body makes and requires.6
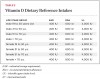
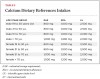
A report by the Institute of Medicine (IOM) in November 20107 reviewed the dietary reference intakes (DRI), the first since the 1997-2004 DRIs. The DRIs include statistical components of distribution. Estimated average requirement (EAR) reflects the estimated median requirement. Recommended dietary allowance (RDA) is derived from the EAR and meets or exceeds the requirement for 97.5% of the population. Tolerable upper intake level (UL) is the highest average daily intake that is likely to pose no risk of adverse effects to almost all individuals in the general population. As intake increases above this, the potential risk of adverse effects may increase.
The IOM’s report recommendations are shown in Table 2 (vitamin D DRI) and Table 3 (calcium DRI). Both sets of recommendations are based on the predicted intakes that meet requirements for the other nutrient. Values were listed for infants and children as well as women who are pregnant or lactating. There is less uncertainty concerning calcium because there is more evidence base and the physiology and metabolism are better understood. Two major limitations of estimating vitamin D dietary requirements are: 1) it is also synthesized following sun exposure of the skin; and 2) because it acts like a hormone it undergoes metabolic feedback loops with endocrine and autocrine functions. In the IOM report, it was assumed that sun exposure was at a minimum and sources of vitamin D came from diet.
The measure of serum 25(OH)D has served as a reflection of total vitamin D exposure and is used to determine adequacy or deficiency. The IOM committee’s review of data suggests the following regarding serum 25(OH)D levels:
- < 30 nmol/L = at risk of deficiency
- 30 nmol/L to 50 nmol/L = some but not all potentially at risk for inadequacy
- 50 nmol/L = all persons sufficient
- > 75 nmol/L = not consistently associated with increased benefit
- > 125 nmol/L = reason for concern
Subtle symptoms such as loss of appetite, diarrhea, insomnia, and muscular weakness may indicate a mild deficiency, while symptoms such as nausea, vomiting, and sleepiness may indicate too much vitamin D. Rickets and osteomalacia are well-known diseases relating to vitamin D deficiency. Vitamin D deficiency has now been associated with other diseases such as cancer, cardiovascular disease (CVD), diabetes, and periodontitis, as well as fractures from falls. But just as there is reason for concern about vitamin D deficiency, there is also a need to determine levels of excess and toxicity and to avoid misclassification of vitamin D deficiency.
Evaluation and Therapy
Diagnosing vitamin D deficiency is accomplished by measuring serum 25(OH)D levels. However, because the kidney tightly regulates serum 1,25(OH)2D levels, they can be normal even though the levels of 25(OH)D are low. Therefore, even with high levels of the active hormone, the patient would be vitamin D deficient. The serum 1,25(OH)2D is a measure of the endocrine function and does not indicate the body stores vitamin D or the autocrine functions of vitamin D.
Treatment of vitamin D comes from sunlight, artificial ultraviolet B (UVB), or supplements, each with their drawbacks. With both sunlight and artificial UVB, patients should realize that exposure will age the skin and increase the risk of nonmelanoma skin cancers. Toxicity is unlikely with sun exposure. Treatment with oral supplements is more difficult than with light because high doses are required to get adequate serum levels of 25(OH)D. The amount needed varies due to sunlight exposure, body fat, age, and skin color, and potential toxicity is possible, although rare.
Colecalciferol is available in the United States over the counter and via the Internet in capsules of 400, 1,000, 2,000, 5,000, 10,000, and 50,000 international units (IU). Colecalciferol 1,000 IU/day will result in about 10 ng/ml elevation of serum 25(OH)D in a 3- to 4-month period. Prescription ergocalciferol is available in a 50,000 IU capsule. Physicians can give 1 to 2 doses of 50,000 IU weekly for 8 to 16 weeks and then maintain > 40 ng/ml levels of 25(OH)D with 50,000 IU doses every 1, 2, or 4 weeks. Dosing is dependent on the other aforementioned variables, but ergocalciferol may be less effective than cholecalciferol in raising 25(OH)D levels.8,9 Cod liver oil is not recommended because of the possible risk of vitamin A toxicity. Regular consumption of recommended amounts of vitamin D in a multivitamin or in fortified foods effectively prevents vitamin D deficiency. Since vitamin D is dependent on the cytochrome P450 enzymes, drugs that are also dependent on those enzymes may interact with vitamin D metabolism.
Besides deficiency, toxicity, although rare, can also occur. Urine calcium and then serum calcium will increase when 25(OH)D levels exceed > 150 ng/ml, and then true toxicity occurs when hypercalcemia calcifies internal organs. Toxicity cannot occur through skin production, because any excess previtamin D3 or vitamin D3 is destroyed by sunlight.4
The only absolute contraindication to vitamin D supplementation is vitamin D toxicity or allergy. Other contraindications to sun vitamin D are dermatological conditions. Liver disease is not a contraindication. Relative contraindications are vitamin D hypersensitivity, which occurs when extrarenal tissues produce 1,25(OH)2D in an unregulated manner causing hypercalcemia, and hypercalcemia itself.3
Vitamin D and Systemic Diseases
Vitamin D has anticancer properties by decreasing cell proliferation, inducing differentiation, inducing apoptosis, stopping angiogenesis, and having anti-inflammatory effects. Lappe et al10 found that 1,100 IU of vitamin D and 1,500 mg of calcium per day dramatically reduced the relative risk for incident cancers over a 4-year period as compared to the placebos. Other studies have not been as positive; one study found that women taking 400 IU of vitamin D3 plus calcium did not have a lower risk of breast cancer over placebo.11 Another study of male smokers found those with higher vitamin D concentration had an increased risk for pancreatic cancer with smoking not as a confounding factor,12 while a follow-up study of males and females who were mostly nonsmokers did not find this association with pancreatic cancer except in those with low sun exposure.13 With these conflicting results, the IOM has concluded that at this time the evidence is too weak to make recommendations regarding vitamin D as it relates to cancer and that it is still a “work in progress.”14
In reference to CVD, calcitriol, which is the natural ligand to the vitamin D receptor, has been shown to inhibit vascular smooth muscle cell proliferation, regulate the renin-angiotensin system, decrease coagulation, and exhibit anti-inflammatory properties.15 A review by Wang et al16 suggests that moderate to high doses of vitamin D supplements may reduce CVD risk while calcium supplements have minimal effects. Several other studies have found that patients with low vitamin D levels had a higher risk for heart disease, being diagnosed with hypertension, or having a heart attack.17-19 Even though vitamin D may have a promising effect on cardiovascular disease, no large-scale randomized trials with CVD as the primary prespecified outcome have been completed. However, new randomized trials examining the role of vitamin D supplementation in CVD are in progress.15
Concerning fractures from falls, autoimmune diseases, and diabetes, several studies exist. With muscle weakness associated with vitamin D deficiency, fractures often result from falls. Researchers have concluded that “fall risk reduction begins at 700 IU and increases progressively with higher doses.”20 It has been theorized that vitamin D may contribute to autoimmune diseases because it is an immunomodulator and plays a role in regulating the immune system. A study by Munger et al21 found that people with the highest vitamin D concentrations had a 62% lower risk of developing multiple sclerosis than those with the lowest concentrations. Lastly, some studies have shown a lower risk of type 2 diabetes with vitamin D,22 but more studies need to be done to elucidate if there is a definite link.6
In January 2010, recruitment began for the Vitamin D and Omega-3 Trial (VITAL) study at Brigham and Women’s Hospital and Harvard Medical School in Boston, Massachusetts, which investigated whether taking daily dietary supplements of vitamin D (about 2,000 IU) or fish oil (about 1 gram of omega-3 fatty acids) reduces the risk of developing cancer, heart disease, and stroke in people who do not have a prior history of these illnesses. The primary outcome is incidence of these diseases after 5 years. With a goal of 20,000 recruitments, this study may give more insight on vitamin D supplementation.
Oral Health and Vitamin D
Periodontal disease is a chronic inflammatory disease that affects approximately 35% of US adults over the age of 30.23 Alveolar bone loss is a key feature in periodontitis, and research suggests that osteopenia may be a predisposing factor for periodontal disease by increasing susceptibility to the effects of inflammation-mediated oral bone loss.24 Genetic polymorphisms in the VDR gene have been associated with bone homeostasis and diseases in which bone loss is manifested.
Genetics
Genetic variants at multiple loci associated with periodontitis synergistically contribute to the overall disease process. There may be candidate genes that play a role in both chronic and aggressive periodontitis. Many of these gene polymorphisms play a role in immunoregulation or metabolism.25
Many studies have looked into the different polymorphisms in the VDR in different ethnic groups. Some studies found a positive association between the tt genotype and t allele in what was then referred to as early onset periodontitis (EOP).26,27 However, there are also contradicting studies that found the T allele significantly associated with chronic periodontitis28,29 and the Tt and tt genotype more prevalent in controls compared to those chronic periodontitis patients.30 With reference to the B or b genotype, several studies did not find a significant difference between the periodontitis population and the controls.31-33 Inagaki et al34 found in their study that loss of alveolar bone, clinical attachment, and teeth occurred highest in AA genotype, while Li et al35 found the FF genotype and the frequency of the F allele significantly higher in the generalized aggressive periodontitis (GAP) group. In relation to GAP, Park et al36 found the short VDR associated with increased risk. However, a common limitation of many of these studies was the small size or the homogeneity of the population in terms of either ethnic group or sex or both.
Periodontal Disease and Vitamin D
Other groups looked at the intake or concentrations of vitamin D in relation to periodontitis. One study found that lower serum 1,25(OH)D3 concentrations were associated with higher attachment loss, which may be explained by the anti-inflammatory effects of vitamin D.37 Krall did two studies. One38 showed no association between vitamin D intake from foods and supplements and the number of teeth with progression of periodontal bone loss. The other one39 stated that although the number of studies on the effects of calcium or vitamin D intake on oral outcomes is limited, they suggest that higher intake levels are associated with reduced prevalence of clinical attachment loss and lower risk of tooth loss. Data from a prospective study of oral health in men show a similar association between higher calcium intake and reduced alveolar bone loss. In agreement with one of Krall’s studies, Miley et al40 showed a trend for better periodontal health in patients receiving periodontal maintenance treatment and vitamin D and calcium supplementation. A recent study found that periodontal disease is more common in women with osteoporosis and is associated with lower vitamin D and higher RANKL and osteoprotogerin.41
Periodontal disease is a multifactorial disease initiated by a bacterial infection leading to a response by the host. Hallmarks of this disease are bone loss and an inflammatory, immune reaction. Vitamin D plays a role in both calcium and bone homeostasis as well as in the immune function. Vitamin D and calcium deficiencies lead to bone loss and increased inflammation, both well-known symptoms of periodontal disease.42 Susceptibility to periodontal disease varies among patients as displayed by their onset, extent, and severity of the disease. Further studies are needed in gender-, ethnic-, and age-specific groups because prior studies utilized groups of test individuals with limited and narrow characteristics.
Conclusion
In conclusion, larger randomized control trials must be performed in both the prevention and treatment of vitamin D deficiency. Although classically thought of as a “bone hormone,” vitamin D plays a role in other parts of the body. It is a predictor for bone health but is also a potential independent predictor of risk for cancer and other chronic diseases. It was thought that when foods were fortified with vitamin D and rickets was no longer a major problem, that the issue with vitamin D was resolved. But now it appears that vitamin D has a greater role in not only skeletal health but nonskeletal health as well. With the definition of < 20 ng/ml of 25(OH)D, approximately 1 billion people worldwide are vitamin D deficient or insufficient.4 Its effects as well as its uses are still to be explored and elucidated, which may help in the treatment of various chronic diseases, including periodontitis. There needs to be a randomized control trial exploring supplementation of vitamin D with periodontal disease measures as the primary outcome to further determine a possible cause-and-effect relationship. Until then, the relationship between vitamin D and periodontitis remains unknown.
References
1. Amano Y, Komiyama K, Makishima M. Vitamin D and periodontal disease. J Oral Sci.. 2009;51(1):11-20.
2. Yagiela JA, Dowd FJ, Neidle EA. Pharmacology and Therapeutics for Dentistry. 5th ed. St Louis, MO: Mosby; 2004.
3. Cannell JJ, Hollis BW, Zasloff M, Heaney RP. Diagnosis and treatment of vitamin D deficiency. Expert Opin Pharmacother. 2008;9(1):107-118.
4. Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357(3):266-281.
5. U.S. Department of Agriculture, Agricultural Research Service. USDA National Nutrient Database for Standard Reference, Release 24. Washington, DC: 2011. http://www.ars.usda.gov/Services/docs.htm?docid=8964. Accessed January 23, 2012.
6. Gonzalez C. Vitamin D Supplementation: An Update. US Pharm. 2010;35(10):58-76.
7. Institute of Medicine. Dietary reference intakes for calcium and vitamin D. Washington, DC: Institute of Medicine of the National Academies; 2010.
8. Trang HM, Cole DE, Rubin LA, et al. Evidence that vitamin D3 increases serum 25-hydroxyvitamin D more efficiently than does vitamin D2. Am J Clin Nutr. 1998;68(4)854-858.
9. Armas LA, Hollis BW, Heaney RP. Vitamin D2 is much less effective than vitamin D3 in humans. J Clin Endocrinol Metab. 2004;89(11):5387-5391.
10. Lappe JM, Travers-Gustafson D, Davies KM, et al. Vitamin D and calcium supplementation reduces cancer risk: results of a randomized trial. Am J Clin Nutr. 2007;85(6):1586-1591.
11. Chelbowski RT, Johnson KC, Kooperberg C, et al. Calcium plus vitamin D supplementation and the risk of breast cancer. J Natl Cancer Inst. 2008;100(22):1581-1591.
12. Stolzenberg-Solomon RZ, Vieth R, Azad A, et al. A prospective nested case-control study of vitamin D status and pancreatic cancer risk in male smokers. Cancer Res. 2006;66(20):10213-10219.
13. Stolzenberg-Solomon RZ, Hayes RB, Horst RL, et al. Serum vitamin D and risk of pancreatic cancer in the prostate, lung, colorectal and ovarian screening trial. Cancer Res. 2009;69(4):1439-1447.
14. Nicholas J. Vitamin D and cancer: uncertainty persists; research continues. J Natl Cancer Inst. 2011;103(11):851-852.
15. Shapses SA, Manson JE. Vitamin D and prevention of cardiovascular disease and diabetes: why the evidence falls short. JAMA. 2011;305(24):2565-2566.
16. Wang L, Manson JE, Song Y, Sesso HD. Systematic review: vitamin D and calcium supplementation in prevention of cardiovascular events. Ann Intern Med. 2010;152(5):315-323.
17. Wang TJ, Pencina MJ, Booth SL, et al. Vitamin D deficiency and risk of cardiovascular disease. Circulation. 2008;117(4):503-511.
18. Giovannucci E, Liu Y, Hollis BW, Rimm EB. 25-Hydroxyvitamin D and risk of myocardial infarction in men: a prospective study. Arch Intern Med. 2008;168(11):1174-1180.
19. Forman JP, Giovannucci E, Holmes MD, et al. Plasma 25-hydroxyvitamin D levels and risk of incident hypertension. Hypertension. 2007;49(5):1063-1069.
20. Liebman B. From sun & sea: new study puts vitamin D & omega 3s to the test. Nutrition Action Healthletter. November 1, 2009:3-7.
21. Munger KL, Levin LI, Hollis BW, et al. Serum 25-hydroxyvitamin D levels and risk of multiple sclerosis. JAMA. 2006;296(23):2832-2838.
22. Pittas AG, Harris SS, Stark PC, Dawson-Hughes B. The effects of calcium and vitamin D supplementation on blood glucose and markers of inflammation in nondiabetic adults. Diabetes Care. 2007;30(4):980-986.
23. Albandar JM, Brunelle JA, Kingman A. Destructive periodontal disease in adults 30 years of age and older in the United States, 1988-1994. J Periodontol. 1999;70(1):13-29.
24. Jeffcoat MK, Chesnut CH III. Systemic osteoporosis and oral bone loss: evidence shows increased risk factors. J Am Dent Assoc. 1993;124(11):49-56.
25. Yoshie H, Kobayashi T, Tai H, Galicia JC. The role of genetic polymorphisms in periodontitis. Periodontology 2000. 2007;43:102-132.
26. Hennig BJ, Parkhill JM, Chapple IL, et al. Association of a vitamin D receptor gene polymorphism with localized early-onset periodontal disease. J Periodontol. 1999;70(9):1032-1038.
27. Sun JL, Meng HX, Cao CF, et al. Relationship between vitamin D receptor gene polymorphism and periodontitis. J Periodontal Res. 2002;37(4):263-267.
28. Tachi Y, Shimpuku H, Nosaka Y, et al. Vitamin D receptor gene polymorphism is associated with chronic periodontitis. Life Sci. 2003;73(26):3313-3321.
29. Wang C, Zhao H, Xiao L, et al. Association between vitamin D receptor gene polymorphisms and severe chronic periodontitis in a Chinese population. J Periodontol. 2009;80(4):603-608.
30. Brett PM, Zygogianni P, Griffiths GS, et al. Functional gene polymorphisms in aggressive and chronic periodontitis. J Dent Res. 2005;84(12):1149-1153.
31. Yoshihara A. Sugita N, Yamamoto K, et al. Analysis of vitamin D and Fcγ receptor polymorphisms in Japanese patients with generalized early-onset periodontitis. J Dent Res. 2001;80(12):2051-2054.
32. de Brito Júnior RB, Scarel-Caminaga RM, Trevilatto PC, et al. Polymorphisms in the vitamin D receptor gene are associated with periodontal disease. J Periodontol. 2004;75(8):1090-1095.
33. Naito M, Miyaki K, Naito T, et al. Association between vitamin D receptor gene haplotypes and chronic periodontitis among Japanese men. Int J Med Sci. 2007;4(4):216-222.
34. Inagaki K, Krall EA, Fleet JC, Garcia RI. Vitamin D receptor alleles, periodontal disease progression, and tooth loss in the VA dental longitudinal study. J Periodontol. 2003;74(2):161-167.
35. Li S, Yang MH, Zeng CA, et al. Association of vitamin D receptor gene polymorphisms in Chinese patients with generalized aggressive periodontitis. J Periodontal Res. 2008;43(3):360-363.
36. Park KS, Nam JH, Choi J. The short vitamin D receptor is associated with increased risk for generalized aggressive periodontitis. J Clin Periodontol. 2006;33(8):524-528.
37. Dietrich T, Joshipura KJ, Dawson-Hughes B, Bischoff-Ferrari HA. Association between serum concentrations of 25-hydroxyvitamin D3 and periodontal disease in the US population. Am J Clin Nutr. 2004;80(1):108-113.
38. Krall EA, Wehler C, Garcia RI, et al. Calcium and vitamin D supplements reduce tooth loss in the elderly. Am J Med. 2001;111(6):452-456.
39. Krall EA. The periodontal-systemic connection: implications for treatment of patients with osteoporosis and periodontal disease. Ann Periodontol. 2001;6(1):209-213.
40. Miley DD, Garcia MN, Hildebolt CF, et al. Cross-sectional study of vitamin D and calcium supplementation effects on chronic periodontits. J Periodontol. 2009;80(9):1433-1439.
41. Jabbar S, Drury J, Fordham J, et al. Plasma vitamin D and cytokines in periodontal disease and postmenopausal osteoporosis. J Periodontal Res. 2011;46(1):97-104.
42. Hildebolt CF. Effect of vitamin D and calcium on periodontitis. J Periodontol. 2005;76(9):1576-1587.
About the Authors
Suellan Go Yao, DMD
Clinical Instructor
Columbia University College of Dental Medicine
Department of Periodontics
New York, New York
James Burke Fine, DMD
Associate Dean for Postdoctoral Education
Professor of Clinical Dentistry and Postdoctoral Director of the Division of Periodontics
Columbia University College of Dental Medicine
New York, New York