You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Asthma is a common chronic disease characterized by repeated episodes of obstruction of the respiratory tract. Susceptibility to this disease appears to be based on an acquired bronchial hyperresponsiveness to either an inhaled antigen to which the patient is allergic (“extrinsic asthma”) or an irritant (“intrinsic asthma”).1 Following exposure to either, a sequence of events is initiated in the airway consisting of an inflammatory response, contraction of the smooth muscles of the bronchioles (bronchoconstriction and bronchospasm), edema of the bronchial lining, and increased secretion of mucous.2 Collectively, these reactions result in an increase in airway resistance and progressive difficulty with breathing.
Symptoms of an acute asthmatic episode, or “asthma attack,” manifest as an abrupt onset of shortness of breath, wheezing, and coughing. Progression of these symptoms is considered to be a respiratory emergency that can lead to hypoxia and even death. In the United States, deaths caused by asthma approached 7,000 in 1952 but decreased to 4,400 by 2000, primarily attributable to the development of new drug therapies for its prevention and treatment.3
The overall prevalence of asthma in the United States is approximately 8%, and more than half of those affected report experiencing an acute asthma episode in the previous 12 months. Hispanics of Puerto Rican ancestry have the highest rate (14.5%), followed by black children (12.5%). Geographically, rates are higher in the Northeast than in other parts of the country. Poverty has been found to be associated with a higher prevalence rate (10.3%).4 Similarly, in a sample of patients seen in the authors’ clinics, which consist primarily of an urban, underserved population, 15% reported a history of asthma. These authors found that in a sample of an urban underserved population seeking urgent dental care at University of Pittsburgh School of Dental Medicine in 2008, 15% reported a history of asthma.
Another feature that characterizes asthma and its ramifications is the cost of this disease. In a study of 67,500 patients with “moderate, persistent asthma,” healthcare costs related to its management were more than $6,000 a year.5
Etiology
Recent evidence shows the development of extrinsic asthma may include a genetically mediated susceptibility to sensitivity to allergens in the environment.6 Examples of allergen stimuli that can generate asthmatic episodes include pollens, dust mites, cockroaches, pet fur, mold, and foods containing sulfites. Triggers for episodes of intrinsic asthma, which is not mediated by the immune system, are the air pollutants ozone and tobacco smoke, cold air, strenuous exercise, stress, and occupational exposure to dust generated by industrial activity.1 Medications, particularly aspirin and other nonsteroidal antiinflammatory agents, also have been associated with asthma attacks.2 Products that contain sulfites used as preservatives in wines, foods, and certain drugs, have been reported to initiate acute episodes of asthma.2,7 In addition, a viral or bacterial infection in the respiratory tract can increase the likelihood of an attack in patients with asthma.2 More recently, it has been determined that obesity, particularly in children but also in adults, contributes to the risk of developing asthma, increases its severity, and may make control more difficult.8
The impact of emotional stress on asthma is well recognized and has been documented extensively.9 Psychological stressors cannot only generate an asthma attack but also contribute to its severity. It has been shown when exposed to stress, patients with asthma develop more severe bronchoconstriction than those without the disease.9 Stress also may be a precursor to the development of asthma because it may increase the risk for developing hypersensitivity to environmental allergens that can result in asthma.9,10 The primary emotional stressors that have been associated with asthma are anxiety, panic, and depression.9
Stress as a potential mediator of asthma is particularly significant for the dental practitioner. Patient anxiety or phobia arising from the anticipation of pain or other discomfort during a dental visit is inherent to the practice of dentistry, and this perception may be further potentiated by patients’ concerns that an asthma attack may occur during dental treatment.
Materials used in conjunction with providing dental care may be another source of asthma in patients. These primarily include filling materials that contain methacrylate monomers, natural rubber latex, and glutaraldehyde disinfectants.11
As noted above, a variety of foods and wines may contain sulfites, which are used widely as preservatives and can cause asthma in sensitive individuals. Local anesthetics that contain epinephrine or levonordefrin require the addition of either sodium bisulfite or sodium metabisulfite, which act as antioxidant preservatives to prolong the shelf life of these anesthetic formulations.12,13 An estimated 5% of people with asthma are sensitive to sulfites thus increasing the likelihood of an attack after the administration of local anesthetic with a vasoconstrictor.7 Because the response to sulfites is apparently dose dependent, it rarely is reported in dentistry.12 However, case reports have described this occurrence, including a patient with a history of asthma who developed asthmatic symptoms and a skin rash after the administration of one carpule of lidocaine with 1:50,000 epinephrine.14 It has been suggested that patients with more severe asthma who require glucocorticoids for the control of their disease are at greater risk for this reaction.13 The reaction to sulfites may be an immunologically mediated sensitivity reaction or a direct effect of sulfur dioxide on nerve endings that results in bronchoconstriction.15
Medical Management for the Control of Asthma
The management of asthma is focused on preventing, controlling, or reversing the two major pathophysiologic components of the disease: contraction of the smooth muscles of the bronchioles and inflammation of the airway. Blocking the contraction of the smooth muscles and resultant bronchoconstriction is accomplished primarily by the sympathomimetic class of drugs (beta-adrenergic drugs or beta2-receptor agonists). The therapeutic intervention for the control of the bronchial inflammation usually entails the use of glucocorticoids. Drugs from either of these categories can be used to limit the frequency of acute episodes. These drugs can be administered orally or by inhalation, but inhalation with an aerosol device is preferred because of a more direct and rapid effect. Furthermore, inhalation devices can achieve therapeutic responses with smaller doses and adverse systemic reactions are less likely.16 In more severe cases, combinations of the bronchodilator and glucocorticoid may be necessary. A selective compilation of medications used for the treatment of asthma is shown in Table 1.
Oral Complications
If the treatment of asthma entails the use of sympathomimetic agents, these drugs have the potential side effect of a reduction in salivary output because of drug inhibition of neural stimuli to the salivary glands.17 Alternative therapy with glucocorticoids increases susceptibility to oral Candida infections (thrush). These side effects may be additive because a reduction in saliva can increase the risk of an overgrowth of Candida independently.18 Given the high prevalence of asthma in the population and the likelihood that affected patients may be using a combination of a sympathomimetic and a glucocorticoid drug, dentists would expect to encounter dry mouth as well as Candida infections more frequently. Reports from the 1980s have described oral and esophageal candidiasis, with prevalence estimates as high as 34% when patients were being treated with glucocorticoids.19 In a more recent review, however, the results of multiple studies that involved 3,741 patients who were using glucocorticoid inhalers found only 0% to 5% had manifestations of clinical candidiasis.20
Limited information is available regarding the effect of sympathomimetic drugs on salivary flow. One study, limited to 28 subjects, determined that salivary flow was not changed after the use of a beta2-receptor agonist inhaler.21 Other studies of children and adolescents with asthma have found no increase in the rate of dental caries that would have been expected if their salivary function had been affected by these inhalers.22,23
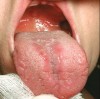
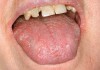
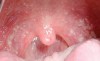
The apparent low incidence of these complications may be attributed to the small doses of drug that metered inhalers deliver (usually measured in micrograms [mcg]) and no greater than 2.5 mg (for albuterol).16 This, however, does not rule out the possibility of oral complications developing in people with severe asthma who may require higher doses via more frequent inhalations or the supplemental administration of systemic corticosteroids. Patients with asthma also may have coexisting medical conditions that entail the use of other classes of xerostomia-producing drugs, such as antidepressants, anxiolytics, diuretics, and antihistamines.17 Additional nondrug-related risk factors for oral candidiasis include smoking, use of dentures, and diabetes mellitus.18 Examples of several patients with Candida-related complications from their asthma inhalers but who had other risk factors are shown in Figure 1 through Figure 3. The frequent involvement of the soft palate may be related to the trajectory of the aerosol from the inhaler.
Another oral complication described as angina bullosa hemorrhagica has been attributed to the use of inhalers for asthma. This condition manifests as the sudden appearance of a blood-filled blister or bulla on the oral mucosa.24 Although some cases have been associated with inhalers, physical trauma appears to be the primary cause.
The availability and use of spacing devices between the inhaler and the mouth and advising patients to rinse after its use may help prevent candidiasis25 as well as other oral complications, particularly in children.16 Nevertheless, when dental clinicians encounter patients who have asthma or other diseases that entail the use of inhalers, dentists should always look for oral complications that may be associated with these devices.
Asthma as a Medical Emergency
Chronic asthma or a history of more severe asthma can lead to permanent airway remodeling, which has the potential to exacerbate all facets of the disease.1 As noted earlier, anxiety and/or stress associated with dental treatment may precipitate an asthma attack.9 A combination of these factors can result in an acute and possibly life-threatening office emergency. Preparing for and reducing the potential of an acute episode must include a complete history of the disease, including medications, frequency of episodes, hospitalizations, and known stimulating factors. Patients who use inhalers for asthma management should be instructed to bring these devices to their appointments.
Both inspiratory and expiratory wheezing are the hallmark symptoms. As the acute episode progresses, patients often become anxious and agitated. Dental treatment should be terminated immediately and instruments, rubber dam, and gauze removed from the patient’s mouth. The office emergency plan should be activated. The patient should be placed in a more upright but comfortable position and receive oxygen by full-face mask. A bronchodilator, such as the beta2-receptor agonist (albuterol), should be provided for inhalation and repeated every 15 minutes if necessary. Beta2-receptor agonists are preferred because an immediate bronchodilating effect is required. Corticosteroid inhalers are not appropriate for the management of an acute asthma attack because their benefit is derived from their antiinflammatory action, which has slower onset. Corticosteroids, however, can be used to supplement the beta2-bronchodilator but may have to be administered orally or intravenously for optimal efficacy.2
Clinical Recommendations
Foremost, the dental practitioner must be vigilant and prepared to cope with an acute episode of asthma while providing dental care (Table 2). The likelihood of this occurrence is magnified by the number of patients in the population who have asthma, in conjunction with stress and other factors in the dental environment that can precipitate an asthma attack. Any patient with a history of asthma should provide the dentist with a description of disease severity, which includes the frequency of episodes, precipitating factors, and degree of control such as rate of inhaler use, and episodes that required hospitalization.
Patients should be reminded to bring their inhaler or other medication to the office and to take their medications before the appointment, as directed. Additional measures include avoiding medications known to have induced their episodes, such as aspirin, nonsteroidal antiinflammatory drugs, and sulfites.
Control of fear and anxiety is another preventive strategy that requires input from the patient as to how it was managed in previous experiences. The use of respiratory depressant agents such as sedatives or opioids in patients with severe asthma should be administered cautiously.2
The practitioner also should be aware that medications used for the treatment of asthma increase the risk of oral infection with Candida. The sympathomimetic agents may cause a reduction in saliva, particularly if taken in conjunction with other xerostomia-producing drugs. This may increase susceptibility to the development of dental caries.
References
1. Schreck DM. Asthma pathophysiology and evidence-based treatment of severe exacerbations. Am J Health Syst Pharm. 2006;63(10 suppl 3):S5-S13.
2. McFadden ER Jr. Asthma. In: Kasper DL, Braunwald E, Fauci AS, et al, eds. Harrison’s Principles of Internal Medicine. 16th ed. New York, NY: McGraw-Hill; 2005:1508-1516.
3. Sly RM. Continuing decreases in asthma mortality in the United States. Ann Allergy Asthma Immunol. 2004;92(3):313-318.
4. Moorman JE, Rudd RA, Johnson CA, et al. National surveillance for asthma—United States, 1980-2004. MMWR Surveill Summ. 2007;56(8):1-54.
5. Colice G, Wu EQ, Birnbaum H, et al. Healthcare and workloss costs associated with patients with persistent asthma in a privately insured population. J Occup Environ Med. 2006;48(8):794-802.
6. Arruda LK, Solé D, Baena-Cagnani CE, et al. Risk factors for asthma and atopy. Curr Opin Allergy Clin Immunol. 2005; 5(2)153-159.
7. Seng GF, Gay BJ. Dangers of sulfites in dental local anesthetic solutions: warning and recommendations. J Am Dent Assoc. 1986;113(5):769-770.
8. Shore SA. Obesity and asthma: implications for treatment. Curr Opin Pulm Med. 2007;13(1):56-62.
9. Lehrer P, Feldman J, Giardino N, et al. Psychological aspects of asthma. J Consult Clin Psychol. 2002;70(3):691-711.
10. Wright RJ, Cohen RT, Cohen S. The impact of stress on the development and expression of atopy. Curr Opin Allergy Clin Immunol. 2005;5(1):23-29.
11. Hamann CP, Rodgers PA, Sullivan KM. Occupational allergens in dentistry. Curr Opin Allergy Clin Immunol. 2004;4(5): 403-409.
12. Finder RL, Moore PA. Adverse drug reactions to local anesthesia. Dent Clin N Am. 2002;46(4):747-757.
13. Pérusse R, Goulet J, Turcotte J. Contraindications to vasoconstrictors in dentistry. II. Hyperthyroidism, diabetes, sulfite-sensitivity, corticodependent asthma, and pheochromocytoma. Oral Surg Oral Med Oral Pathol. 1992;74(5):687-691.
14. Schwartz HJ, Gilbert IA, Lenner KA, et al. Metabisulfite sensitivity and local anesthesia. Ann Allergy. 1989;62(2):83-86.
15. Phillips JF, Yates AB, Deshazo RD. Approach to patients with suspected hypersensitivity to local anesthetics. Am J Med Sci. 2007;334(3):190-196.
16. Drugs for asthma. Treat Guidel Med Lett. 2005;3(33):33-38.
17. Moore PA, Guggenheimer J. Medication-induced hyposalivation; etiology, diagnosis, and treatment. Compend Contin Educ Dent. 2008;29(1):50-55.
18. Guggenheimer J, Moore PA. Xerostomia: etiology, recognition and treatment. J Am Dent Assoc. 2003;134(1):61-69.
19. Spector SL, Wangaard C, Bardana EJ Jr. The use of cultures and immunologic procedures to predict oropharyngeal candidiasis in patients on steroid aerosols. Clin Allergy. 1982;12(3):269-
278.20. Ellepola ANB, Samaranayake LP. Inhalational and topical steroids, and oral candidosis: a mini review. Oral Dis. 2001;7(4):211-216.
21. del-Río-Navarro BE, Corona-Hernández L, Fragoso-Ríos R, et al. Effect of salmeterol and salmeterol plus beclomethasone on saliva flow and IgA in patients with moderate-persistent chronic asthma. Ann Allergy Asthma Immunol. 2001;87(5):420-423.
22. Meldrum AM, Thomson WM, Drummond BK, et al. Is asthma a risk factor for dental caries? Finding from a cohort study. Caries Res. 2001;35(4):235-239.
23. Schulman JD, Taylor SE, Nunn ME. The association between asthma and dental caries in children and adolescents: a population-based case-control study. Caries Res. 2001;35(4):240-246.
24. Yamamoto KY, Fujimoto M, Inoue M, et al. Angina bullosa hemorrhagica of the soft palate: report of 11 cases and literature review. J Oral Maxillofac Surg. 2006;64(9):1433-1436.
25. Salzman GA, Pyszczynski DR. Oropharyngeal candidiasis in patients treated with beclomethasone dipropionate delivered by a metered-dose inhaler alone and with Aerochamber. J Allergy Clin Immunol. 1988;81(2):424-428.