You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
As the understanding of the subtleties of the caries process has advanced with clinical and scientific research, the recommendations for managing the disease have become increasingly multifaceted and convoluted. An understanding that caries is a complex interactive imbalance in the oral biofilm and that dentists need to work with natural processes, rather than against them, has led to a medical approach to managing a diseased biofilm and helping convert it back to a healthy state. Each new step in understanding has led to an addition to recommended treatment regimens. As these recommendations are added to current protocols, the protocols become very complex, confused, and difficult for the profession and staff to understand. The plethora of products on the market with subtle variations in indications and applications add an additional layer of complexity to managing a diseased biofilm. As a consequence, it has become difficult to educate patients and implement caries diagnosis and management strategies into a busy practice. If the diagnosis and management of a cariogenic biofilm can be distilled back to the core issues and kept simple, the ability to implement an effective caries management program into a busy private practice can become a reality.
Healthy mouths have a diverse range of bacteria within a healthy dental biofilm, potentially more than 400 species. A diseased biofilm is populated predominantly with a range of acidogenic, aciduric bacteria, leading to the signs and symptoms of caries—demineralization and cavitation of the teeth. The shift of a healthy biofilm to a diseased state can be caused by many risk factors, including diet, saliva, medications, absence of fluorides, and lack of homecare.1
Metabolism of carbohydrates by cariogenic bacteria results in more acid production. This production lowers the pH of the biofilm that inhibits many commensal organisms. When compounded with other risk factors, the acidic pH becomes the selection pressure that results in an overabundance of acidogenic organisms.2
Demineralization sufficient to cause cavitation is symptomatic of the underlying disease. Dental professionals must assess a patient’s risk profile and look for factors contributing to a cariogenic biofilm. This process is known as caries management by risk assessment (CAMBRA).1 By identifying the presence of a diseased biofilm and assessing a patient’s risk profile, the disease can be managed from a medical perspective rather than a surgical one. Any dental damage also has to be addressed, and minimally invasive surgical strategies used, along with remineralization concepts, to limit any damage that has been caused by a previously undiagnosed cariogenic biofilm. Primarily, the profession’s focus has been on repairing the cavities caused by a cariogenic biofilm. However, if a cavity has been diagnosed, then by definition, the patient has a cariogenic biofilm. Simply restoring the cavities will not change the diseased state of the biofilm. Dentists should be asking themselves how they can help patients avoid the formation of more cavities—the concepts of CAMBRA and minimal intervention (MI). Currently, these concepts are being implemented in educational institutions as the scientific validation continues.
Implementation Strategies
The primary reason for introducing CAMBRA into a practice is to benefit its patients. One of the primary stumbling blocks to CAMBRA implementation is fitting a new procedure into what is currently a smooth-running and efficient office. Both the dentist and staff often perceive that the time commitment is too complex to try to integrate new procedures into an already busy patient flow. Implementing CAMBRA will affect all of the systems in the practice, from scheduling, to fees, and everything in between. Implementation requires that all team members understand the philosophical change. The goal of CAMBRA is to help patients achieve dental health by educating them, motivating them to improve the behaviors that are contributing to their risk, and providing them with effective strategies and treatments to manage their disease and maintain oral health after it has been achieved. This paradigm shifts the team away from finding and filling cavities to asking questions first, asking more questions, determining why there is disease, figuring out how to prevent future disease, and drilling only what is necessary, followed by monitoring and measuring the treatment outcomes.
Preimplementation Planning There are three phases to setting up a practice for the implementation of CAMBRA:
- Planning
- Education
- Preimplementation practice
To effectively implement a CAMBRA program in the office, someone has to take the lead. Generally, this has to be the dentist, but a motivated assistant or hygienist also may be able to take the lead. To introduce new concepts requires some planning. The goals and objectives need to be written down, and an implementation plan needs to be formulated. A timeline also needs to be determined. This has to be done in consultation with staff to ensure implementation is going to be effective.
One of the observable stumbling blocks is a lack of “buy in” by some team members. Education is the best solution to this problem. There are many resources for scientific information: PubMed is an excellent resource for articles on caries risk assessment. Additional information can be gathered by attending local or state courses focused on CAMBRA. Taking the entire dental team to these courses is an excellent opportunity to get everybody on the same page. The primary educational source of the author’s change in philosophy and rationalization of MI and CAMBRA science and philosophies has come via the World Congress of Minimally Invasive Dentistry.
After the whole team understands the concepts and science behind the MI and CAMBRA philosophies, the road blocks tend to disappear. Depending on the structure of the practice, it may be more effective to introduce the program in smaller steps to help establish the most efficient strategies. Commonly identified issues are:
- How will this interfere with current treatment flows?
- What are the costs involved?
- What is the potential income?
- Who is going to be in charge?
- Who will do the initial CAMBRA risk assessment?
- What products and materials are going to be required?
- What is the timeline required for staged or full implementation?
There needs to be constant review process during implementation so that everyone can contribute their thoughts. It is preferable to have team members volunteer for specific steps rather than assign them. By breaking the process into sections and asking who would like to do what, implementation is more likely to go smoothly. Success comes with commitment to what is a major philosophical change in how we manage caries.
Keeping It Simple
After the commitment has been made, the primary goal is to identify the assessment protocols and treatment materials that will be required. There are a plethora of assessment, diagnostic, and treatment techniques on the market, and this can complicate the decision process as to what is most effective from both a time and cost basis. The goal is to keep the process both time efficient and cost effective for the dentist and the patient, and we need to recognize that time is the primary cost to a practice.
Practice Makes Perfect
After the implementation plan has been designed and the products selected, it is very effective to run through the protocol with both the dentist and staff being the patient. This will help identify problem areas that are specific for a given practice situation, and will raise awareness of both the dentist and staff to the patient’s perceptions of the whole process. It also will ensure the whole team is on the same page so that the patients are receiving the same message, regardless of which team member is speaking with them. This preimplementation run-through will help everyone understand his or her part in the process.
In-Office Strategies
There are five basic steps to assessing and treating a cariogenic biofilm.
- Risk Assessment
- Biofilm Assessment
- Diagnosis
- Patient Education
- Treatment Implementation
Risk Assessment
The goal is to identify whether patients are at risk. There are several forms and systems that have been developed to assess a patient’s risk profile. The initial CAMBRA forms were developed by Featherstone and colleagues and can be downloaded from the CDA Foundation Web site (www.cdafoundation.org). In the author’s experience, these forms are inefficient in private practice because they fail to keep the protocols simple and time efficient. While these detailed forms leave nothing to chance, when in use as a private practice screening tool they can be complicated. The forms that have been adopted by the author are a distillation of current research, including recent research by Featherstone et al,3,4 that has identified four clinical disease indicators that are indicative of high risk:
- Visible cavitations
- Cavities in the past 3 years
- Radiographic interproximal lesions
- White spot lesions
The presence of any of these factors indicates high risk. The forms also contain a series of validated questions that will highlight patient risk behaviors. The adult form (Figure 1) takes approximately 2 minutes to complete with the patient, keeping the focus on gathering pertinent information in a time-effective manner. The results indicate if the biofilm requires further assessment, and then provide treatment recommendations for the patient based on the overall results. The child form has a different series of risk-assessment criteria based on the recognized clinical indicators of risk for children (Figure 2).
Biofilm Assessment
An assessment of the state of the biofilm also is helpful in the overall management of caries. The benefits are twofold. The results will give a reference point before commencing any treatment and confirm the results obtained from completion of the risk assessment form. After treatment of the biofilm and risk modification, the biofilm can be retested to assess treatment efficacy.
Several systems can be used to assess the biofilm. Tests, such as CRT® (Ivoclar Vivadent, Inc., www.ivoclarvivadent.com), GC Plaque Indicator Kit (GC America, Inc., www.gcamerica.com), and CariScreen™ (Essology Pty Ltd, www.essology.com) directly assess the biofilm. Other tests tend to look at the state of the saliva to assess whether there are any factors that may be contributing to a diseased biofilm. At this stage in the patient assessment, testing saliva is not time effective and should be reserved for patients where poor salivary flow rates, pH, and buffering might be a contributory factor.
Bacterial cultures can be used to assess the presence of salivary planktonic mutans streptococci and lactobacilli. These tests are clinically time consuming and culture results take 2 to 3 days to become available, which complicates the logistics of patient assessment. The results will provide a broad indication of the presence and levels of mutans streptococci and lactobacilli (Figure 3), but do not assess the overall cariogenic potential of the biofilm, which can have many acidogenic, aciduric species. To date more than 23 cariogenic species have been identified in plaque biofilm.
Another test for salivary planktonic mutans streptococci is the monoclonal antibody test (Saliva-Check Buffer Kit, GC America, Inc). This is more time effective: only 15 minutes to get a result. However, it only tests for mutans streptococci and ignores all other potential contributory cariogenic species (Figure 4).
If the primary goal is to keep the focus on the clinical time efficiency and applicability of a test, a simpler and more efficient technique is to perform real-time assessment of the cariogenic potential of the biofilm. There are two tests that are clinically time efficient and effective. The GC Plaque Indicator Kit assesses the acidogenic potential of the biofilm by taking a direct sample of the plaque and applying a sucrose/litmus solution to it.5 If high levels of acidogenic bacteria are present, they produce a rapid increase in acid from digestion of the sugar in 5 minutes, leading to a drop in pH, which is indicated by the litmus solution (Figure 5).
The CariScreen test uses a different approach to assess the cariogenic potential of a biofilm. This test, based on the ability of aciduric bacteria to survive in a low pH environment, checks for total cariogenic potential of the biofilm rather than the presence of 1 or 2 bacterial species. This ability to survive in a low pH environment is based on the presence of a cellular wall hydrogen ion pump that maintains internal cellular homeostasis at the more neutral pH required for the cellular biochemistry to function correctly. This pump requires significant energy, which is supplied by mitochondrial adenosine triphosphate (ATP). The CariScreen test uses a bioluminescence assay to measure biofilm ATP levels.6 This test occupies about 10 seconds of the clinician’s time and takes about 30 seconds to provide a real-time chairside result (Figure 6).
How biofilm screening tests are integrated into a practice depends on the staffing structure. As a rule, the more expanded the practice structure, with hygienists and expanded functions auxiliaries, the easier the tests are to implement. A solo practice without auxiliary personnel is probably the most difficult practice in which to integrate new procedures. From this perspective, the GC Plaque Indicator Kit and CariScreen system are more time effective because direct clinical patient contact is limited to less than 15 seconds for each test.
Diagnosis
Is the patient at risk? It should take no more than 5 clinical minutes to gather the information required to make an initial diagnosis. Up to this point, the data gathering can be assigned to any trained staff member. The gathered information allows the dentist to make one of three basic diagnoses: the patient is low risk, at risk of developing a cariogenic biofilm, or definitely has a cariogenic biofilm. If a patient has a diagnosed cavity, the patient has a cariogenic biofilm, and simply restoring the cavity will do nothing to change the state of the biofilm, which leads to the final step of treating the disease: treatment of the diseased biofilm.
Patient Education
The effective use of clinical time will have a bearing on the perceived costs of delivering a caries management program. The critical breakdown point is the failure to help patients understand why this service is being offered, and the financial and biological benefits when they succeed in controlling the disease. Preeducation of patients can significantly reduce the time needed to explain concepts to patients. An effective mechanism is to take the time to write a practice newsletter, introducing the concepts and the reasons this new service is being offered. By incorporating the newsletter as part of the patient’s recall reminder, the information is delivered just before contact with the practice, ensuring the information is foremost in the patient’s mind. Another option is to place the information on the practice Web site and send an e-mail to the patient database to direct patients to the new page. The more basic information a practice can give patients in these formats, the less chair time that will be needed to explain CAMBRA. The information staff members give patients in the operatory will reinforce what they read earlier.
Generally, the first contact will be with the hygiene department, and it may be necessary to schedule a few extra minutes to allow for a discussion on the subject of CAMBRA. The most effective message will come from the dentist, but it is imperative that all team members are supplying similar information to the patient. Primarily, the patient has to understand the changes that are happening and why they are being implemented. A logical goal for the CAMBRA conversation is for the patient to understand that just treating cavities will not treat his or her disease and that cavities are only underlying signs and symptoms of a bacterial infection. The patient needs to understand that this biofilm infection must be diagnosed and treated as a disease process. The patient also needs to understand the concept of the balance between health and disease and the pathologic factors vs the protective factors. With proper background, the patient should be able to help identify any changes in risk factors during future visits.
In the author’s practice, any patient that is diagnosed as being at risk during the first appointment is given a short 10-minute, narrated PowerPoint® CD to take home to view. This CD covers the basics of a biofilm disease and presents the most common risk factors and how to manage the disease. A simple analogy is used, a weed-infested lawn equating to a cariogenic biofilm, and how simply mowing the lawn (brushing teeth) does not change the fact that the lawn is full of weeds. It is now simply a short weedy lawn. Using nontechnical analogies helps patients grasp these new concepts.
If, by the second visit, the patient has not reviewed the information, then the previous communication effort has failed, and they have not yet grasped the significance of the information given to them at their first visit. Sadly, some patients do not want to be helped. That is their prerogative.
If a patient desires additional information, the cariogram from the Malmö University (www.mah.se/english) is an interactive Windows®-based system that provides real-time changes in risk as various patient risk parameters are changed. This can be a valuable tool to help educate patients as to how their behaviors can modify their current and future risk. However, this can be time consuming, and in recognition of the cost associated with clinical time, it is best reserved for focused education for patients who need more help. Patients can access the site and use the cariogram to observe behaviors that will improve their risk profile by altering various parameters in the program.
Treatment Implementation
The results of the risk assessment will provide information that allows the treatment protocol to be tailored to a patient’s specific requirement. There are many protocols that have varying degrees of compliance and success.
Research has shown that low pH is the primary mediator in a biofilm becoming cariogenic.7-9 Logically, reversal of negative pH selection pressures should help a biofilm revert to health. Current brushing, flossing, and antibacterial rinse routines have led to varied success rates. Implementation of positive pH strategies and the development of a healthy biofilm may be the way of the future, rather than simply trying to eliminate the biofilm on a daily basis. Each practice needs to assess the current products on the market and decide on those that will help their patients most effectively.
Overtreament and Undertreatment Issues
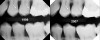
Some may argue that this treatment is supervised neglect or undertreatment. In the past, without good risk assessment and effective management of the biofilm disease, that may have been the case. With the current (and improving) understanding about the management of a biofilm imbalance and the use of risk assessment, this is no longer the case. As an example, a patient presented in 1999 with 2 interproximal radiographic lesions on teeth Nos. 12 and 13 (Figure 7). The radiographs are only a snapshot in time. By understanding the patient’s risk profile, immediate treatment was not indicated. This decision has been validated by examination of her radiographs from 2007, which show these lesions were from a historic cariogenic episode and were no longer progressing.
Many incipient lesions that do not penetrate the tooth’s enamel are candidates for more conservative, noninvasive therapy, including glass ionomer cement sealants, for control of the biofilm disease in combination with remineralization protocols.10-15
In the past, the rate of progression of dental caries made conservative decisions highly questionable. Today, with lower caries incident rates and the ability to reduce the rate of caries progression, surgical interventions need to be minimal in all but the most aggressive caries situations. As part of their risk assessment protocol, dentists need to evaluate the frequency of recall for each patient. If the dental team has evaluated the patient as a high caries probability patient, then prophylactic preventive therapies and other principles identified in this article should be implemented. This reduces the risks of either overtreatment or undertreatment.
Overtreatment occurs when interventions are unjustified or too aggressive for the clinical situation. The goal of MI dentistry is to preserve the maximum amount of healthy dental tissues. An example of this situation is the use of air-abrasion instruments to open questionable fissures to “see what’s in there” based on a sign that is not significantly correlated with decay penetration to the dentin, such as fissure staining. The consequences of overtreatment are well-characterized as the “restoration/rerestoration cycle.” Any cutting of tooth structure weakens the tooth and should be avoided if possible. On the other hand, undertreatment occurs when a clinician systematically provides nontreatment or less-than-optimal treatment where real pathology exists. The consequence of undertreatment includes advancing disease and potential loss of more tooth structure.
The Economics Of A Minimal Intervention Philosophy
Dentistry historically has focused on restoration of the damage caused by a cariogenic biofilm and ongoing retreatment of the initial lesion as the restorative materials or tooth structure continues to fail. Ongoing failure can result from the initial restorative treatment and/or ineffective management and treatment of the initial diseased biofilm, which leads to failure of the initial restorations.
Historically, third-party systems and patients placed a priority on restorative procedures because dental caries was pandemic and validated risk assessment tools were not available. Further, because sealants and fluoride treatments are covered financially, the focus is not on preventive treatments. Insurance companies and patients are willing to pay for a filling, but not fluoride or remineralization therapy (casein phosphopeptide-amorphous calcium phosphate) to remineralize and repair white spot lesions. Within this environment, there is a new current dental terminology (CDT) code for fluoride varnish as a therapeutic treatment for the moderate-to high-risk caries patient. As the positive effects and financial advantages of intervention in the disease process become more apparent, there is promising progress with new fee codes being added by third-party payers.
CAMBRA has a number of procedures associated with it that have direct related fees and fee codes already in place. In the CDT 7, in addition to the normal prevention codes for prophylaxis and fluoride applications, there are codes for:
Radiographic interproximal lesions
- D0415: Bacteriology Studies
- D0145: Oral Evaluation Patient < 3 Years, Counseling Primary Caregiver
- D1206: Topical Fluoride Application for Therapeutic Measures Moderate-to High-Risk Caries Patient
It is likely that medical insurance may cover some of the diagnostic tests, such as salivary flow and buffering capacity measurements, in the near future. In the case in which a patient has no effective insurance coverage for CAMBRA, a good explanation of the positive benefits of accurately managing the disease most often will have the patient accepting the fees associated for the service. This is when preeducation of patients using newsletters and practice brochures becomes effective.
Conclusion
The implementation of MI concepts and CAMBRA began several years ago. It was a piecemeal, disjointed approach that had good scientific support but little practical advice for general practitioners, no validation forms, and disjointed treatment concepts for a bacterial biofilm disease.
Since the advent of modern dentistry, the profession has been focused on the surgical management of the damage caused by this disease. Dentistry now has reached a point, with research and the clinical implementation of MI concepts, where primary surgical intervention to treat a bacterial infection as a first option becomes an untenable treatment concept.
Understanding this concept leads to a decision when patients need to have their risk evaluated individually. Every patient will be unique, and the treatment will need to be custom-fit for that individual patient at that point in time. Then, dentists must continue to monitor each patient, because a low-risk patient today may become a high-risk patient tomorrow.
Dentists need to accept that this revolution, in all reality, will be an evolution in the way they practice. To implement CAMBRA effectively will require time and effort, and the steps need to be kept small and manageable, with the whole team remaining involved. As the science and concurrent clinical applications evolve, effective management of caries will continue to improve. CAMBRA is the current standard of care, but future advances will lead to further improvements in this standard.
Between the direct economic benefit and the new patient referrals, CAMBRA more than supports itself from a business model. However, money aside, the real reason to implement CAMBRA is for the patient. There is no greater reward than making a significant difference in a patient’s life.
Disclosure
The author is a shareholder in Oral BioTech.
References
1. Featherstone JD. Caries prevention and reversal based on the caries balance. Pediatr Dent. 2006;28(2):128-132.
2. Marsh P. Are dental diseases examples of ecological catastrophes? Microbiology. 2003;149(Pt 2):279-294.
3. Doméjean-Orliaguet S, Gansky SA, Featherstone JD. Caries risk assessment in an educational environment. J Dent Educ. 2006; 70(12):1346-1354.
4. Featherstone JD, Doméjean-Orliaguet S, Jenson L, et al. Caries risk assessment in practice for age 6 through adult. J Calif Dent Assoc. 2007;35(10):703-713.
5. Walsh LJ. Dental plaque fermentation and its role in caries risk assessment. International Dentistry SA Australasian Edition. 2006; 1(3):4-13.
6. Sauerwein R, Pellegrini P, Finlayson T, et al. ATP bioluminescence: quantitative assessment of plaque bacteria surrounding orthodontic appliances [abstract]. J Dent Res. 2008;87(Spec Iss B):1288.
7. Kleinberg I. A mixed-bacteria ecological approach to understanding the role of the oral bacteria in dental caries causation: an alternative to Streptococcus mutans and the specific-plaque hypothesis. Crit Rev Oral Biol Med. 2002;13(2):108-125.
8. Kleinberg I, Marsh PD. Dental plaque as a biofilm and a microbial community—implications for health and disease. BMC Oral Health. 2006;6(Suppl 1):S14.
9. Sissons CH, Anderson SA, Wong L et al. Microbiota of plaque microcosm biofilms: effect of three times daily sucrose pulses in different simulated oral environments. Caries Res. 2007;41(5):413-422.
10. Walsh LJ. Application of the system for total environmental management (STEM) to demineralization, dental erosion and wear. Australian Dental Practice. Jan/Feb 2008;52-58.
11. Malcmacher L. Enamel remineralization: the medical model of practicing dentistry. Dent Today. 2006;25(11):68-69.
12. Mount GJ. A new paradigm for operative dentistry. Aust Dent J. 2007;52(4):264-270.
13. Reynolds EC, Cai F, Cochrane NJ, et al. Fluoride and casein phosphopeptide-amorphous calcium phosphate. J Dent Res. 2008; 87(4):344-348.
14. Cochrane NJ, Saranathan S, Cai F. Enamel subsurface lesion remineralisation with casein phosphopeptide stabilised solutions of calcium, phosphate and fluoride. Caries Res. 2008;42(2):88-97.
15. Diefenderfer KE, Stahl J. Caries remineralization therapy: implications for dental readiness. Mil Med. 2008;173(1 Suppl):48-50.