You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
As clinicians, we often need to accelerate the healing process, augment bone lost from periodontal diseases, trauma, or congenital abnormalities or for optimal implant placement, and improve the soft-tissue for better esthetics and patient comfort. Growth factors have long been viewed by the scientific community as having the potential to revolutionize our ability to positively influence the healing process and clinical outcomes in a multitude of procedures.1 Platelet-derived growth factor (PDGF) has received some of the greatest attention because of its broad wound-healing activities on both the bone and soft tissue.2-4 Over the past several years, purified recombinant human PDGF (rhPDGF) in combination with synthetic matrices such as beta tricalcium phosphate (-TCP) or allografts has been studied in a series of rigorous clinical and pre- clinical trials that have yielded exciting results.5-10 These evidence-based findings are summarized in this article, and the potential for this therapy to become the new standard of care for treatment of certain types of bone and soft tissue defects is discussed.
Platelet-Derived Growth Factor— The Body's Wound-Healing Catalyst
PDGF is one of the body's main initiators of healing, whether the injury is in soft tissue (eg, the gingiva) or hard tissue (eg, bone).1 It is a protein naturally found sequestered in blood platelets as well as in bone matrix in 3 different forms: PDGF-AA, PDGF-AB and PDGF-BB.11-13 It is secreted locally during clotting by blood platelets at the site of soft- or hard-tissue injury and stimulates a cascade of events that leads to the wound-healing response.1 The PDGF-BB form in particular is a potent stimulator of many types of connective-tissue cells including periodontal ligament, fibroblasts, cementoblasts and osteoblasts.1,14-18
Numerous studies have shown that PDGF-BB acts as a magnet to promote rapid cell migration (chemotaxis) into the injured region with subsequent proliferation (mitogenesis) of osteoblasts and periodontal ligament fibroblasts by binding to well-characterized cell surface receptors.1,14-18 Platelet-derived growth factor has also been shown to be important during embryonic development.19 Studies with transgenic mice lacking the PDGF α-receptor showed abnormal development and repair of the craniofacial bones and vertebrae, indicating that PDGF is essential for normal bone development and repair.19
Development of the PDGF-Based Therapies for Periodontal and Implant Related Use
The human gene for PDGF-BaB has been isolated and placed into yeast cells that could be grown under sterile conditions and used to produce large quantities of the recombinant (synthetic) human protein, commonly referred to as rhPDGF.20 The synthetic rhPDGF was then tested in many laboratory animal studies. These studies indicated that rhPDGF has the potential to be used therapeutically to direct and control skin, gingival, and bone regeneration.21
For example, in the general bone field a tibial osteotomy study using rhPDGF in rabbits demonstrated that this protein substantially increased the rate of fracture repair compared with control sites; the repaired bone demonstrated biomechanical strength that was not significantly different from unoperated normal intact bone.21 Also, when PDGF was injected subperiosteally in long bones it induced intramembranous bone formation.22 In a detailed study in osteoporotic animals involving dual-energy x-ray absorptiometry bone density scans, quantitative computed tomography scans, biomechanical testing, and histological analyses, the periodic injection of rhPDGF substantially increased bone density in the long bones and spine.23
To develop rhPDGF for routine clinical use in periodontal and oral surgical procedures, extensive in vitro and in vivo animal studies and human clinical trials have been performed using PDGF-BB alone, in combination with other growth factors such as insulin-like growth factor (IGF), or in combination with many currently avail- able grafting materials. These have been extensively re- viewed elsewhere,1,20,24 and it is not the authors' intention to provide a complete review in this article, but rather highlight a few studies that illustrate these findings.
Lynch and colleagues were the first to publish the regenerative potential of PDGF-BB when used to treat naturally occurring periodontal defects in Beagle dogs.14,25 The authors observed increased cellular activity after applying PDGF-BB, leading to significant bone, cementum, and periodontal ligament regeneration. When used around dental implants, direct application of rhPDGF-BB in combination with IGF produced 2 to 3 times more new bone at earlier points of time in dogs.26 Promising results also were seen in immediate extraction socket implants treated with PDGF-BB/IGF in combination with an ePTFE nonresorbable barrier membrane. Bone density and bone-to-implant contacts were increased twofold for sites treated with the growth factors compared with sites treated with the membrane alone or membranes combined with bone grafts.27
A human clinical trial for rhPDGF/IGF treatment applied to osseous periodontal defects was reported by Howell and colleagues.28 A statistically significant increase in alveolar bone formation was seen in the treatment sites that received rhPDGF/IGF in a methylcellulose carrier at 9 months postoperatively when compared with untreated control sites. New bone height for the treated group was 2.08 mm compared with 0.75 mm new bone height for sites treated with open flap debridement. In retrospect, this was a remarkable finding because it was subsequently demonstrated that the methylcellulose gel carrier used to deliver the growth factors obstructed bone formation in isolated defects treated with guided bone regeneration (GBR) in a rat model.29
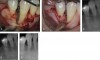
More recently, rhPDGF-BB has been combined with tissue-specific scaffolds such as bone grafts or bone substitutes to take advantage of the fundamental healing principles embodied in the concepts of tissue engineering (Figure 1).1 Using this approach dramatically enhanced bone formation, and gingival healing has been found in large, critical sized alveolar bone defects. Simion and colleagues demonstrated substantial vertical alveolar ridge augmentation using rhPDGF-BB and xenogenic scaffolds in an established canine model for alveolar ridge defects.30 A PDGF-infused block of deproteinized bovine cancellous bone was placed in the defect site and stabilized using 2 titanium dental implants. The effect of rhPDGF with and without a resorbable barrier membrane placed between the surgical flap and the graft block was examined and compared with untreated graft blocks covered with the collagen membrane. Radiographic and histological analysis indicated that the greatest bone regeneration occurred for the rhPDGF-treated graft block without the collagen membrane (Figure 2), while the traditional GBR procedure using the graft and membrane resulted in little or no bone formation.
This study demonstrated that when there is direct access to the periosteum and its rich supply of osteogenic and angiogenic cells, PDGF exerts a potent osteogenic, angiogenic, and chemotactic effect, leading to greatly improved bone formation. Bone formation occurred from both the coronal and apical surfaces of the PDGF-treated graft, indicating that osteoblasts and bone-forming pre- cursor cells were attracted into the graft from both the superior (coronal) periosteal surface and the inferior (apical) medullary spaces.30
Clinical Availability of Growth Factor Enhanced Matrices (GEM)
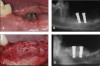
Building on the tissue engineering concept of combining rhPDGF with tissue-specific matrix materials, a clinical trial was completed to assess the effect of rhPDGF mixed with bone allograft for treatment of interproximal intrabony defects and Class II furcation lesions associated with advanced periodontitis.5,6 After full-thickness flap reflection and thorough defect debridement, sites were treated with demineralized freeze-dried bone allograft that had been soaked in a solution containing rhPDGF-BB (Figure 3).
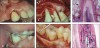
This was the first study reported providing clear histological evidence of periodontal regeneration for human Class II furcation defects.19 The regenerative effect of rhPDGF in combination with mineralized freeze-dried bone allograft has been further documented clinically in a separate case study reported by Nevins.7 Two patients with extremely severe bone loss requiring surgical bone grafting were treated with freeze-dried bone allograft saturated with rhPDGF-BB, and a resorbable barrier membrane was placed over the defect before flap closure. Patients were followed for 11 months, at which time surgical reentry was performed to evaluate bone fill at the defect site. The patients in this case series exhibited excellent soft-tissue healing after surgery despite the severity of the original defects. In addition, excellent radiographic fill was observed. To verify these radiographic results and re-treat any residual defect, surgical reentry was performed. Bone was observed to have nearly completely filled the original defect (Figure 4).
The excellent clinical, radiographic, and histologic results obtained using rhPDGF-BB in combination with a bone matrix provided evidence to support the development of a totally synthetic engineered growth-factor enhanced matrix, combining rhPDGF-BB with ß-TCP, a well known synthetic bone substitute. The results of a large multicenter randomized blinded human clinical trial evaluating the effective- ness of rhPDGF-BB with a porous ß- TCP matrix were recently reported by Nevins and colleagues.8 The study included 180 participants with at least 1 interproximal periodontal defect 4 mm or greater in depth after debridement. Three treatment groups were evaluated: Group 1— ß-TCP plus 0.3 mg/mL rhPDGF-BB (GEM 21S Growth-factor Enhanced Matrix, BioMimetic Therapeutics, Inc., ); Group 2—ß-TCP plus 1.0 mg/mL rhPDGF-BB; and Group 3—ß-TCP plus buffer alone. ß-TCP granules were saturated with the treatment solution before the graft was placed in the defect site. Patients were followed for a period of at least 6 months, with periodic clinical and radiographic evaluations to monitor the safety and effectiveness of the treatment.
Outcome measures included evaluation of soft-tissue changes and radiographic assessment of bone growth. Probing depth reduction, gingival recession, and clinical attachment level (CAL) were measured, and differences from baseline measurements for each treatment group were determined. Comparisons between the rhPDGF-BB treatment groups (Groups 1 and 2) and the positive control group (Group 3) were made and evaluated for statistical significance. Safety was monitored throughout the trial by assessing the frequency and severity of clinical or radiographic adverse events.
Excellent postsurgical healing was observed for all sites treated with rhPDGF-BB (Groups 1 and 2). The study results indicated that there was significantly greater CAL gain at 3 months for Group 1 compared with Group 3 (ß-TCP alone), indicating an early benefit of rhPDGF-BB treatment. At 6 months, the Group 1 CAL gain continued to be greater than Group 3, but this difference was not statistically significant. Also, there was significantly less gingival recession at 3 months for Group 1 compared with Group 3. Group 2 (1.0 mg/ml rhPDGF-BB + ß-TCP) demonstrated similar trends to Group 1 for CAL gains and gingival recession when compared with Group 3, but the differences were not significant. Radiographic assessment revealed that the percent bone fill was significantly increased at 6 months for Group 1 compared with both Groups 2 and 3 (Figure 5A). A subgroup analysis further indicated that rhPDGF-BB treatment (Groups 1 and 2) improved bone fill in smokers and for all defect types (1, 2, 3 wall and circumferential). Similarly, lin- ear bone growth was also significantly greater for Group 1 compared with Groups 2 and 3 (Figure 5B). No significant differences were seen in the number or severity of adverse events among the groups, indicating that both rhPDGF-BB and the ß-TCP matrix were well tolerated in the defect site. A clinical case from the clinical trial treated with rhPDGF is shown in Figure 6.
The results of this clinical trial clearly demonstrates the clinical benefit of using rhPDGF-BB in combination with a synthetic ß-TCP matrix (GEM 21S) for bone and gingival tissue regeneration. Additionally, evaluation of representative cases from the pivotal clinical trial has demonstrated that these results remain stable, with CAL, percent bone fill, and linear bone growth increasing or remaining stable for at least 24 months after surgery. A 24- month follow-up of patients from this study showed that radiographic bone fill continued to increase in treated defects, with increasing levels of radiopacity and patterns of trabeculation observed during this extended period.9
To put these results into perspective, a review of the historical literature for other approved regenerative therapeutics further suggests that rhPDGF compares favorably to other existing treatments approved by the Food and Drug Administration in terms of CAL gain and radiographic percent bone fill (Table 3). Preliminary clinical results indicate that recession defects also may be effectively treated with rhPDGF and a collagen pad or membrane and a coronally advanced flap as an alternative to subepithelial connective tissue grafting. In a pilot clinical trial reported by McGuire and Scheyer, rhPDGF was applied to recession defects > 3 mm.10 A collagen membrane, saturated with PDGF-BB, was placed over the growth factor enhanced matrix in accordance with standard guided tissue regeneration practices. Results of the study suggest that rhPDGF plus a collagen membrane was as effective in covering recession defects as connective tissue grafting, without the need for a second surgical site. A larger study is currently underway to further document this use of rhPDGF.
Conclusion
Combining rhPDGF with a tissue-specific matrix such as a bone graft or synthetic bone substitute for osseous defects or a collagen pad for gingival recession has the potential to fulfill the clinical need for a commercially available material that predictably results in regeneration of bone and soft tissue. The results of human histologic trials demonstrate that rhPDGF combined with bone allograft results in robust periodontal regeneration. A large, multicenter clinical trial for GEM21S demonstrates that this product is safe and effective for wide- spread clinical use. Clinical and radiographic results show that it effectively and reproducibly accelerates significant gains in soft-tissue attachment level gain and bone fill in moderate to severe periodontal defect sites. This treatment approach is supported by extensive and rigorous in vitro, preclinical, and clinical studies that provide strong evidence for the mechanism of action for rhPDGF in healing bone, ligament, and gingiva.
These extensive data indicate that an off-the-shelf therapy such as rhPDGF-BB with allograft or ß-TCP pro- vides substantial benefits for clinical practitioners and patients undergoing periodontal or peri-implant surgery. With the promise of accelerated healing, more abundant and higher quality bone formation without the need to harvest autogenous bone, rhPDGF-BB in combination with osteoconductive matrices appears to have the potential to become the new standard of care.
References
1. Lynch SE, Genco RJ, Marx RE, eds. Tissue Engineering: Applications in Maxillofacial Surgery and Periodontics. Chicago, Ill: Quintessence Publishing Co;1999: 1-297.
2. Deuel TF, Kawahara RS, Mustoe TA, et al. Growth factors and wound healing: platelet-derived growth factor as a model cytokine. Annu Rev Med. 1991;42:567-584.
3. Whitby DJ, Ferguson MW. Immunohistochemical localization of growth factors in fetal wound healing. Dev Biol. 1991;147:207- 215.
4. Lynch SE, Nixon JC, Colvin RB et al. Role of platelet-derived growth factor in wound healing: synergistic effects with other growth factors. Proc Natl Acad Sci USA. 1987;84:7696-7700.
5. Camelo M, Nevins ML, Schenk RK, et al. Periodontal regeneration in human Class II furcations using purified recombinant human platelet-derived growth factor-BB (rhPDGF-BB) with bone allograft. Int J Periodontics Restorative Dent. 2003;23:213- 225.
6. Nevins M, Camelo M, Nevins ML, et al. Periodontal regeneration in humans using recombinant human platelet-derived growth factor-BB (rhPDGF-BB) and allogenic bone. J Peridontol. 2003;74:1282-1292.
7. Nevins M, Lynch SE: Regeneration of periodontal defects using recombinant human platelet-derived growth factor (rhPDGF-BB) and mineralized freeze-dried bone allograft (FDBA). Int J Perio Rest Dent. 2006; in press.
8. Nevins M, Giannobile WV, McGuire MK, et al. Platelet-derived growth factor stimulates bone fill and rate of attachment level gain: results of a large multicenter randomized controlled trial. J Periodontol. 2005;76:2205-2215.
9. McGuire MK, Kao RT, Nevins M, et al. rhPDGF-BB promotes healing of periodontal defects: 24-month clinical and radiographic observations. Int J Periodontics Restorative Dent. 2006;26:223-231.
10. McGuire MK, Scheyer ET. Comparison of recombinant human platelet-derived growth factor-BB plus beta tricaclium phospahte and a collagen membrane to subepithelial connective tissue grafting for the treatment of recession defects: A case series. Intl J Periodontics Restorative Dent. 2006;26:127-133.
11. Antoniades HN. Human platelet-derived growth factor (PDGF): purification of PDGF-I and PDGF-II and separation of their reduced subunits. Proc Natl Acad Sci USA. 1981;78:7314-7317.
12. Ross R, Raines EW, Bowen-Pope DF. The biology of platelet- derived growth factor. Cell. 1986;46:155-169.
13. Johnsson A, Heldon CH, Wasteson A, et al. The c-sis gene encodes a precursor of the B chain of platelet-derived growth factor. EMB J. 1984;3:921-928.
14. Lynch SE, Williams RC, Polson AM, et al. A combination of platelet-derived and insulin-like growth factors enhances periodontal regeneration. J Clin Periodontol. 1989;16:545-558.
15. Centrella M, McCarthy TL, Kusmik WF, et al. Relative binding and biochemical effects of heterodimeric and homodimeric isoforms of platelet-derived growth factor in osteoblast-enriched cultures from fetal rat bone. J Cell Physiol. 1992;147:420-426.
16. Piche JE, Graves DT. Study of the growth factor requirements of human bone-derived cells: a comparison with human fibroblasts. Bone. 1989;10:131-138.
17. Bowen-Pope DF, van Koppen A, Schatterman G. Is PDGF really important? Testing the hypotheses. Trends Genet. 1991;7:413-418.
18. Soriano P. Abnormal kidney development and hematological disorders in PDGF beta-receptor mutant mice. Genes Dev.1994;8:1888-1896.
19. Schatterman GC, Morrison-Graham K, van Koppen A, et al. Regulation and role of PDGF receptor alpha-subunit expression during embryogenesis. Development. 1992;115:123-131.
20. Lynch SE: Bone regeneration techniques in the orofacial region. In Bone Regeneration and Repair: Biology and Clinical Applications. Eds. Lieberman JR and Friedlaender GE. Humana Press, 2005;359-390.
21. Nash TJ, Howlett CR, Martin C, et al.: Effect of platelet-derived growth factor on tibial osteotomies in rabbits. Bone. 1994;15:203-208.
22. Joyce ME, Jingushi S, Scully SP, et al.: Role of growth factors in fracture healing. Prog Clin Biol Res. 1991;365:391-416.
23. Mitlak BH, Finkelman RD, Hill E et al.: Effect of systemically administered PDGF-BB on the rodent skeleton. J Bone Miner Res. 1996;11:238-247.
24. Giannobile WV and Somerman MJ: Growth and amelogenin-like factors in periodontal wound healing. A systematic review. Ann Periodontol. 2003;8:193-204.
25. Lynch SE, de Castilla R, Williams RC, et al. The effects of short-term application of a combination of platelet-derived and insulin-like growth factors on periodontal wound healing. J Periodontol. 1991;62:458-467.
26. Lynch SE, Buser D, Hernandez RA, et al. Effects of the platelet- derived growth factor/insulin-like growth factor-I combination on bone regeneration around titanium dental implants. Results of a pilot study in beagle dogs. J Periodontol. 1991;62:710-716.
27. Becker W, Lynch SE, Lekholm U, et al. A comparison of ePTFE membranes alone or in combination with platelet-derived growth factor and insulin-like growth factor-I or demineralized freeze-dried bone in promoting bone formation around immediate extraction socket implants. J Periodontol. 1992;63:929-940.
28. Howell TH, Fiorellini JP, Paquette DW, et al. A phase I/II clinical trial to evaluate a combination of recombinant human platelet-derived growth factor-BB and recombinant human insulin-like growth factor-I in patients with periodontal dis- ease. J Periodontol. 1997;68:1186-1193.
29. Lioubavina-Hack N, Karring T, Lynch SE, et al. Methyl cellulose gel obstructed bone formation by GBR: an experimental study in rats. J Clin Periodontol. 2005:32:1247-1253.
30. Simion M, Rocchietta I, Kim D, et al. Vertical ridge augmentation by means of deproteinized bovine bone block and recombinant human platelet-derived growth factor-BB: a histologic study in a dog model. Int J Periodontics Restorative Dent. 2006;26:415-423.






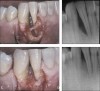
![Figure 5 rhPDGF-BB combined with ß-TCP significantly improved linear bone growth [5B] compared with ß-TCP alone in a large multicenter prospective randomized controlled fully blinded clinical trial.](/media/thumbnail/9743)