You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Medical dictionaries describe asepsis as the process of removing pathogenic organisms or protecting against infection by such organisms. Aseptic techniques, therefore, are specific standards developed to reduce the potential for infection by microorganisms during invasive medical procedures. The modern history of surgical field asepsis dates back to Joseph Lister.1 After coming across Louie Pasteur’s work on the role of microorganisms in fermentation, Lister developed methods to wash wounds with carbolic acid (phenol) and observed a dramatic reduction in the incidence of gangrene postoperatively. His landmark paper in 1867, titled “Antiseptic Principle of the Practice of Surgery,” laid the foundation for aseptic techniques in modern surgery, including cleaning surgical instruments with phenol and even wearing gloves. Two decades earlier, however, Ignac Semmelweis, a Hungarian physician described as the “savior of mothers” had independently discovered that the simple act of a surgeon washing his hands with a chlorinated lime solution preoperatively reduced the incidence of puerperal fever in obstetric clinics.2 This discovery was particularly impressive because it preceded Robert Koch’s postulate and the Germ Theory of Disease.3 Too early for his time, Semmelweis was ridiculed by his contemporary colleagues for his seemingly bizarre presurgical ritual. With time, however, the role of microorganisms in the pathogenesis of disease and wound healing became clear, and aseptic techniques related to all stages of surgery and healing became an integral part of surgical protocol and clinical medicine.
Over time, various fields of medicine discovered the role of infectious microorganisms in pathogenesis of their corresponding tissues and organ systems. In endodontics, while the bacterial infection of the pulp was postulated as one of the causes of pulpal and periapical disease,4 it was not until the landmark Kakehashi and Stanley article in 1965 that the exclusive importance of microorganisms in the pathogenesis of pulpal disease became clear.5 This study proved that in the absence of bacteria, mechanical exposure of the dental pulp always leads to dentin bridge formation and repair instead of pulpal necrosis. Furthermore, later studies demonstrated that the failure of conventional endodontic therapy is also associated with the regrowth of bacteria in the root canals after endodontic therapy.6,7
As a result of Kakehashi’s landmark study, the endodontic triad of cleaning, shaping, and obturation can now be viewed in a different light. The triad can be reduced to those sets of techniques that deal with the process of root canal decontamination (cleaning and shaping procedures), and those that deal with the prevention of recontamination (obturation and coronal seal).
Why Do Vital Cases Fail?
Once the role of bacteria in endodontic infections and failure of conventional endodontic therapy became clear, a logical question surfaced. If bacteria were the cause of failure in failed conventional endodontic cases, why is there occasional treatment failure in completely vital endodontic cases—for example, after pre-prosthetic endodontic therapy? Clearly, such cases are devoid of bacterial content at the time of treatment. So, where does the bacteria come from to cause failure in such cases? Coronal leakage may be a mechanism of failure in long-term recontamination of root canals, but for those vital cases that fail soon after treatment with persistent postoperative symptoms, there must be another explanation. If the Kakehashi landmark study is true, it can be deduced that endodontic failure of vital cases that lead to periapical infection is caused by bacterial contamination of the operating field during the root canal procedure, introducing microbes into the previously sterile root canal system. Otherwise, if the root canal therapy was performed in completely aseptic environment, much like Kakehashi’s germ-free rats, there should, theoretically, be no harmful consequences to vital pulp capping, pulpotomies, or even missing an entire canal such as the second mesial–buccal canal in a completely vital (sterile) tooth undergoing root canal therapy procedure.
While providing a completely sterile field is not clinically possible, reducing the microbial burden and minimizing field contamination during conventional endodontic cases is mandatory for improving the success rate. This article attempts to review some potential sources of contamination and cross-contamination during conventional non-surgical root canal therapy. Dentists and assistants following the strictest aseptic protocol should logically improve case outcome by reducing canal contamination during root canal therapy.
Contamination vs Cross-Contamination
For the purposes of this article, contamination is defined as the introduction of bacteria from the patient’s own mouth into the sterile operatory field and cross-contamination is the introduction of bacteria from a different source other than the patient into the surgical field. Because all biological surfaces—including skin, mucosa, the lining of the respiratory and digestive tracts—as well as many biological secretions, such as saliva and crevicular fluid, are full of an individual’s own bacterial flora, surgical field contamination or cross-contamination may easily occur as a result of contact with such surfaces and fluids during the various stages of the surgical procedure.
Preventing Cross-Contamination
The first step in understanding how to manage dental The required protocol for prevention of cross-contamination in the dental operatory has been adequately explained through the practice of universal precautions and adherence to standard sterilization and infection control methods recommended by the Center for Disease Control (CDC), American Dental Association (ADA), and Occupational Safety and Health Administration (OSHA).8-10 However, it is important to realize that the level of asepsis required to prevent the level of cross-contamination that can cause failure of endodontic therapy is somewhat higher than that, practiced to prevent the transfer of infectious systemic disease. In the former case, even normal microbial flora from the operator’s or the dental nurse’s skin or bodily fluids that would otherwise be completely innocuous to the patient’s systemic health can potentially become a source of antigenic activity inside the patient’s root canals. If such normal constituents of the skin flora end up inside the root canal, they may potentially cause a periapical infection if they find access to those tissues. This has even been observed by the presence of Candida albicans and mold inside failed root canal treated teeth.11
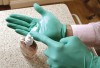
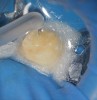
Therefore, adherence to full aseptic techniques during surgical set-up and avoiding bare skin contact with any of the surfaces that will later come in contact with the surgical field—either directly or indirectly through gloved hands or instrument handles—is very important to avoid a potential source of cross-contamination (Figure 1 and Figure 2). Even pulling gloves out of the glove box with bare, unwashed hands will immediately contaminate the glove with the wearer’s skin bacteria. Gloves should be worn by prewashed hands and only by grabbing them from the sleeve cuff portion while avoiding contact with the rest of the gloves (Figure 3). Alternatively, alcohol disinfection of gloves immediately after wearing them is a good practice because the standard examination gloves are not sterile (Figure 4). The practice of disinfecting gloves throughout the root canal procedure by both the clinician and the assistant—particularly just before root canal obturation—ensures that these surfaces remain clean. These are only a few of the many possible means of cross-contamination. The dental team should actively think about other possible means of cross-contamination in the workflow and find means to eliminate their occurrence.
Preventing Contamination
Because the patient’s own bacteria are closest in proximity to the operating field, contamination is generally the most common method of root canal infection during root canal therapy. Therefore, such potential routes for contamination and how they can be avoided should be the primary concern of astute practitioners. The most significant method to reduce the incidence of contamination is adherence to complete isolation of the surgical field with a rubber dam and implementation of methods to reduce exposure to the patient’s own crevicular fluid, saliva, breath, and skin flora.
Rubber dam isolation is the standard of care and performing endodontic therapy without a dam is considered negligence.12,13 It is important to understand that the main function of the rubber dam in endodontic therapy is not to prevent inhalation of endodontic files or simplify access to the field; but rather to provide the operator with a completely aseptic field devoid of the microbial contents of saliva, crevicular fluid, and even the patient’s own breath. As a result, isolation substitutes that keep the patient’s tongue away, such as cotton or gauze, may provide a seemingly accessible field, but are not adequately aseptic to eliminate all potential endodontic pathogens from the field. Touching adjacent teeth for fulcrum contaminates gloves, which indirectly contaminates files during file pre-bending and manipulations of other instruments during the procedure.
Ideally, once the rubber dam is placed and a primary isolation has been obtained, all potential areas of salivary seepage around the dam should then be caulked using a caulking material.14,15 Seepage of crevicular fluid into the field is also a potential source of contamination. It has been shown that the intracanal bacterial composition during root canal infections mimics the composition of the bacteria found in the crevicular fluids.16 As a result, all access to fluids from this area should be blocked. The rubber dam should ideally be inverted so that seepage is reduced. Sodium hypochlorite disinfection of the tooth surface at this phase is recommended, as it adds to the overall efficacy and cleansing of the root canal instrumentation.17-20
It is important to realize that touching the patient’s face and skin to reposition them for an x-ray or better visualization will immediately contaminate the dental assistant’s gloves. Double-gloving during radiography and touching the patient’s face, or changing gloves after such incidences are both recommended.
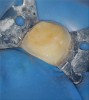
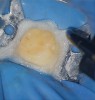
After caries removal and just before the pulpal exposure phase of root canal therapy, final isolation of the field should be performed. At this time, a well-inverted rubber dam without any tears should be in place and all microscopic areas of seepage between the rubber dam clamp and the tooth should be sealed using a caulking material (Figure 5). The surgical field and the tooth may then be flooded with sodium hypochlorite and the patient should not experience any bleach-like taste. This simple test ensures that a fluid tight seal is present around the tooth and final isolation has been achieved. At this point, pulpectomy may be performed knowing that an aseptic field is present. Checking this seal throughout the procedure by flooding the tooth with hypochlorite has both the benefits of disinfecting all internal and external surfaces of the tooth while checking for this important fluid tight seal (Figure 6 and Figure 7). Should the seal be compromised, an attempt to repair or replace the dam should be done first before continuing to perform the root canal procedure.
Complete removal of decay and sealing the area with a core or caulking material during root canal therapy should always be a priority before the pulpectomy procedure is begun. Under no circumstances should decay be present in the tooth before instrumentation of the pulp. All traces of decay should be removed first, including faulty restorations with potential decay underneath, and those areas must be decontaminated and potentially rebuilt before the pulpectomy procedure. Otherwise, the risk of carrying field bacteria deep into the root canal is present. Ideally, a caries-indicating solution should be used to confirm complete removal of all decay. Performing root canal therapy in the presence of decay is against all endodontic principles and defies the concept of asepsis.
Three Phases of Field Disinfection
Because the potential for field contamination and cross-contamination with negative consequences on the outcome of non-surgical root canal therapy is a real possibility, the author has classified three distinct phases of field disinfection with corresponding techniques for aseptic control that can help reduce the bacterial burden in the operating field and potentially enhance success. These three phases precede the three clinical endodontic milestones of pulpal access, instrumentation, and obturation, which is the clinical term for filling and sealing. It is important to understand that if endodontic therapy is to be performed preprosthetically in a vital pulp, maximum asepsis is required throughout all three phases, as such cases are generally sterile and their inherent sterility should be maintained throughout the procedure. However, if decay is present or if root canal therapy is being performed in a non-vital/necrotic pulp, incrementally higher levels of asepsis are required toward the end of the procedure as canal decontamination potentially recontaminates the surgical field throughout the cleaning and shaping process.
The first phase of aseptic control are those techniques used just before pulpal exposure and before instrumentation of the root canals. During this phase, standard aseptic techniques consist of using a rubber dam and sterile instruments. However, because anesthesia is tentative and proper orientation for pulpal access may require the occasional removal of the rubber dam at various times, and because removal of old fillings and decay is constantly contaminating the operating field, this phase of asepsis is less crucial than the later phases that begin after pulpal exposure.
Because all decay should be removed during the access preparation, the resulting field is once again contaminated after caries removal with a high-speed or low-speed handpiece at the end of the first phase of aseptic control. The second phase of aseptic control is marked by renewed decontamination of the field and instruments (including the hand mirror) before the pulpectomy procedure. This kind of surface disinfection is best achieved through wiping the operating surfaces (both the tooth and the rubber dam,) with a solution of hypochlorite or iodine. The dental mirror, the clinician’s and assistant’s gloved hands, and other examination tools that come in contact with the patient’s mucosa during examination and anesthesia before placement of the rubber dam and during caries removal are also contaminated with saliva and carious dentin. These items should either be wiped clean again or, ideally, a different set of instruments and gloves (an access set-up) should be used before the pulpal exposure rather than afterward (Figure 8).
Instrumentation begins only after the second phase of disinfection has been performed. Throughout the instrumentation phase, a copious volume of irrigation is used to disinfect and decontaminate the root canal. Because many endodontically treated teeth are necrotic, addressing the removed necrotic debris is a significant factor in controlling bacteria. An alcohol-soaked piece of gauze may be used to remove gross debris from the endodontic file flutes. The use of bead sterilizers to disinfect files during root canal treatment has fallen out of favor; however, wiping the file flutes frequently cleans the instrument’s chip space, which consequently improves its cutting efficiency and avoids reintroduction of removed infected dentin back into the root canal. This second phase of asepsis begins with a change of gloves and disinfecting the operating field surface, and is maintained by wiping the file flutes frequently and using generous amounts of sodium hypochlorite to irrigate the root canals.
The third phase of asepsis is the most crucial phase and its onset is marked by the end of instrumentation and the preparation for canal obturation. Here, the author recommends a final surface decontamination of the operating field with either hypochlorite, iodine, or alcohol, combined with a final change of gloves and the use of sterile obturation instruments and clean filling material. First and foremost, the integrity of the rubber dam seal should be reevaluated. Should any leakage be present around the sulcus from a loss of seal throughout manipulation of the area during the instrumentation phase of treatment, the leakage should be addressed thoroughly and the area resealed and caulked. The presence of a fluid-tight seal must be confirmed during the third phase of aseptic control and before the obturation. Salivary leakage into the root canal during the obturation phase is complete failure of the required aseptic technique for proper root canal therapy.
Once the rubber dam seal is reconfirmed, a final irrigation of all canals with hypochlorite or a disinfecting irrigant is required before the obturation. The use of sterile or non-contaminated paper points and gutta-percha points is critical. Handling the gutta-percha or paper points with contaminated gloves is one of the most likely ways of inoculating the root canal just before obturation in a well-sealed, rubber-dam–isolated field.
The author recommends submerging the collected master-cones in a solution of full-strength sodium hypochlorite for a period of 30 seconds before use. This surface disinfection just before their introduction into the canals helps avoid the presence of any bacteria or mold on their surfaces. The author also recommends the use of sterile paper points to dry root canals. Stale, old paper points and gutta-percha may look clean, but they can easily be contaminated with bacteria or mold that can potentially cause root canal failure. Some paper point brands are still usable after autoclaving. This may be a consideration before the root canal procedure, or sterile single-use packs of paper points should be used.
Once the operating surfaces, the tooth, and all root canals are fully disinfected in the presence of a fluid-tight seal, and when clean gutta-percha and paper points are combined with the use of clean gloves and instruments, the last piece of the puzzle is the use of an antimicrobial sealer to address the likelihood that some contamination or cross-contamination is still present and that despite best efforts, a small amount of bacteria still remains in the root canal. Here, dentists and dental assistants should choose from a long list of potential sealers where their antimicrobial properties are taken into context against their safety, efficacy, and clinical ease of use.
Lastly, root canal decontamination does not end at the obturation stage of root canal therapy. The prevention of coronal microleakage after root canal therapy is a significant factor in long-term success and case outcome, and may be considered the fourth phase of asepsis. This phase spans from the placement of a well-sealing provisional restoration in the access cavity all the way to the integrity of the coronal seal of the final restoration. Astute clinicians should know that this last phase of asepsis is a continuum and its ultimate success is dependent on the quality and integrity of the restorative work and patient compliance to prevent root canal recontamination through coronal leakage.
Conclusion
Endodontic therapy is a field that most resembles microbiology. Because complete removal and decontamination of the root canal system is the only recipe for a successful outcome, all potential sources of contamination and cross-contamination of the surgical field should be addressed with the same level of priority as the mechanical and technical skills to decontaminate already-infected teeth. The highest success rates are not achieved by mere removal of the bacteria from infected root canals; but also, long-term success hinges on the ability to prevent reinfection of the root canal not only during the treatment but also well after with the presence of a well-sealing restoration. Small steps in infection control and asepsis can dramatically reduce the chance of root canal failure in vital endodontic cases. Astute dentists and dental assistants will focus on the concept of asepsis through all phases of endodontic therapy. Adherence to good aseptic protocol will not only improve the success rate of endodontic therapy, it can also lead to successful outcomes in all facets of endodontic therapy, including those of vital pulp therapy.
References
1. Rhoda T, Joseph Lister—Father of Modern Surgery. New York: Bobbs Merrill; 1996.
2. Hanninen O, Farago M, Monos E. Ignaz Philipp Semmelweis, the prophet of bacteriology. Infection Control. 1983;4(5):367-370.
3. Madigan M, Martinko J. Brock Biology of Microorganisms. 11th ed. New York: Prentice Hall; 2005.
4. Miller W. An introduction in the study of the bacteriopathology of the dental pulp. Dent Cosmos. 1894;36:505.
5. Kakehashi S, Stanley HR, Fitzgerald RJ. The effects of surgical exposures of dental pulps in germ-free and conventional laboratory rats. Oral Surg Oral Med Oral Pathol. 1965;20:340.
6. Nair PNR , Sjogren U, Krey G, et al. Intraradicular bacteria and fungi in root filled, asymptomatic human teeth with therapy-resistant periapical lesions: a long-term light and electron microscopic follow-up study. J Endodon. 1990;16:580.
7. Sjogren U, Figdo D, Persson S, Sundqvist G. Influence of infection at the time of root filling on the outcome of endodontic treatment of teeth with apical periodontitis. Int Endodon J. 1997;30:297.
8. American Dental Association: OSHA: What You Must Know. Chicago, Ill: 1992.
9. Centers for Disease Control and Prevention: Guidelines for Infection Control in Dental Health-Care Settings—2003. MMWR. 2003;52:1.
10. Centers for Disease Control and Prevention. Recommendations for Prevention of HIV Transmission in Health-Care Settings. MMWR. 1987;36(Suppl 2S):1.
11. Baumgartner JC, Khemaleelkul S, Xia T. Occcurance of Candida albicans in infections of endodontic origin. J Endodon. 2000;26:695.
12. Cochran MA, Miller CH, Shedrake MA. The efficacy of the rubber dam as a barrier to the spread of microorganisms during dental treatment. J Am Dent Assoc. 1989;119:141.
13. Cohen S, Schwarts SF. Endodontic complications and the law. J Endodon. 1987;13:191.
14. Bramwell JD, Hicks ML. Solving isolations problems with rubber base adhesives. J Endodon. 1986;12:363.
15. Greene RR , Sikora FA, House JE. Rubber dam application to crownless and cone-shaped teeth. J Endodon. 1984;10:82.
16. Kobayashi T, Hayashi A, Yoshikawa R, et al. The microbial flora form root canals and periodontal pockets of non-vital teeth associated with advanced periodontitis. Int Endodon J. 1990;23:100.
17. Bystrom A, Sundqvist G. The antibacterial action of sodium hypochlorite and EDT A in 60 cases of endodontic therapy. Int Endodon J. 1985;18:35.
18. Bystrom A, Sundqvist G. Bacteriologic evaluation of the effect of 0.5 percent sodium hypochlorite in endodontic therapy. Oral Surg Oral Med Oral Pathol. 1983;55:307.
19. Bystrom A, Sundqvist G. Bacteriologic evaluation of the efficacy of mechanical root canal instrumentation in endodontic therapy. Scand J Dent Res. 1981;89:321.
20. Goldman LB, Goldman M, Kronman JH, Lin PS. The efficacy of several irrigating solutions for end
About the Author
Allen Ali Nasseh, DDS, MMSc
Clinical Instructor
Harvard School of Dental Medicine
Boston, Massachusetts
Private Practice
Boston, Massachusetts