You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Dental professionals are becoming more involved in interdisciplinary care, identifying symptoms in the oral cavity that may be indications of systemic disease. A thorough patient assessment, examining both intraorally and extraorally, should be part of routine examination during dental treatment. It is also vitally important to inform the patient of all procedures completed during the examination and why they are done. Not only is it part of good patient care, this level of education pertaining to all procedures builds credibility with patients, as they may then recognize the broad spectrum of oral healthcare; it involves more than preventive and restorative care.
HPV-Induced Oropharyngeal Cancer
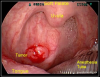
As we begin examining some conditions affecting the GI tract, the first structures beyond the oral cavity are encompassed by the oropharynx. One of the most significant conditions affecting the oropharynx (OP)-which includes the posterior soft palate, the tonsils (including the anterior and posterior tonsillar pillars), the base of tongue, and beyond-is oropharyngeal cancer (OPC) (Figure 1). It is reported that the etiologic agent for most oropharyngeal cancers is human papillomavirus (HPV), primarily the high-risk subtypes of either HPV 16 or 18. These subtypes are referred to as high risk because of their oncogenic potential and are believed to remain in a latent phase in oropharyngeal epithelium after infection years prior. The prevalence of high-risk oral HPV is much more common in men (7.3%) than in women (1.4%).1 It has long been recognized that HPV is the etiologic agent in cancers of the cervix and other genital structures. HPV-induced oropharyngeal cancers have surpassed all other HPV-induced cancers and, as expected based on the prevalence of high-risk HPV, are far more common in men than women.
Like most cancers of the oral cavity, most oropharyngeal cancers are squamous cell carcinomas (SCCs). They are different, however, from oral cavity SSCs. Most SCCs of the oral cavity are believed to be multifactorial in etiology, the most significant etiologic agents being tobacco and alcohol use. While HPV may play a role in some oral cancers of the oral cavity, it is considered to be a much less significant etiologic agent than in those of the oropharynx; most SCCs of the oral cavity are HPV-negative tumors.
When dental professionals refer to head and neck cancers, there must be specific identification as to the type, rather than classifying all as "oral cancer." For example, a squamous cell carcinoma is how most oral cavity and oropharyngeal cancers are classified, even though they have different etiologies and should be delineated as SCC of the oral cavity (OCSCC) or SCC of the oropharynx (OPSCC). Head and neck cancers can include salivary gland tumors, lymphoid tumors, and others with obscure etiologies. These head and neck cancers do not have any correlation to HPV2; it is imperative to be specific to avoid confusion among dental professionals.
There are several additional differences to be noted between OCSCC and OPSCC. Epidemiologically, OCSCC is most prevalent in African-American males. This is not the case for HPV-induced oropharyngeal cancers. It is reported that only 4% of African-American males versus 34% of middle-aged white males are affected with HPV-induced oropharyngeal cancers.3The parameters of sexual behavior also play a major etiologic role in OPSCC, unlike OCSCC. This is not only oral sexual behavior, behavior, however. The lifetime number of sexual and oral sexual partners, and an earlier age at sexual debut, also contribute to the profile. While a majority of OCSCC cases are in an older adult population, the average age for OPSCC is approximately 10 years younger than HPV-negative tumors of the oral cavity.4 Symptoms are quite different as well. The oral cavity lesions observed in OCSCC are virtually non-existent in OPSCC. Many of these tumors are not easily seen by the dental professional during intraoral exams, unless they are present in the tonsillar areas, and even then, they may be difficult to detect.5 The extraoral exam is the most important step in discovering these pathologies. Palpating is essential, as the first symptoms are often from the migration of the pathology into the regional lymph nodes resulting in a neck mass. Other symptoms may include throat and/or ear pain, hoarseness, and pain or difficulty during swallowing.
HPV-positive tumors of the OP are known to have a better prognosis than HPV-negative tumors of the oral cavity. HPV-positive tumors exhibit a better response to chemotherapy and/or radiation treatments. It is believed that the unique biology of HPV-positive tumors and perhaps a low co-morbidity in younger age groups are contributors. There is also no field cancerization associated with HPV malignancies, as opposed to what is observed with HPV-negative tumors in the oral cavity. The spread to regional lymph nodes in OPSCC occurs more quickly than in the OCSCC, even though the disease is more treatable.6
As a result of the vast differences between HPV-positive tumors and HPV-negative tumors, there is now a separate clinical and pathological tumor, node, and metastasis (TNM) grading and staging classification for HPV-positive oropharyngeal cancer in order to create a better understanding of the disease behavior.7 HPV status identifies a biologically distinct disease entity underpinned by a definable and different molecular etiologic process.6 Research at the molecular level of carcinogenesis is showing potential for enhanced diagnosis and treatment possibilities for oral cavity and oropharyngeal cancers.8
Treatments, whether in the form of radiation, chemotherapy, immunotherapy, excisions, or others, can result in many side effects both intraorally and extraorally. Some of the side effects may include trismus, tongue immobility, mucosal sensitivity, and xerostomia, many of which may be long-term. During treatments, a number of products in the form of rinses, pastes, and more may help with oral mucositis.9The dental professional should be aware of the patient's experiences with treatments and offer options to ease the discomfort for the patient.
There appears to be confusion among the general population concerning HPV-induced disease, compounding the necessity for dental professionals to be knowledgeable about the other and far more common HPV lesions that manifest in the oral cavity. Compared with HPV oral lesions in general, HPV-induced malignant transformation is rare.
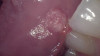
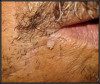
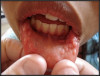
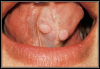
HPV lesions that manifest themselves in the mouth or on the skin do not present major consequences. Although there are many subtypes noted for HPV lesions, the most common lesions that are present in the oral cavity are of the low-risk subtypes which do not become carcinogenic.2 The squamous papilloma is the most common, followed by verruca vulgaris, multifocal epithelial hyperplasia, and condyloma acuminatum (Figure 2 through Figure 5).10
The lesions could look very similar; however, they are slightly different versions, often caused by the same subtype. A complete description of these lesions can be accessed elsewhere.10,11
Gastroesophageal Reflux Disease
Beyond the oral cavity and the oropharynx is the esophagus. The primary condition affecting the esophagus is gastroesophageal reflux disease (GERD).12 GERD is described as the unobstructed, free movement of stomach contents into the esophagus or mouth causing troublesome symptoms or complications.12
GERD is ranked as the most frequent GI diagnosis associated with outpatient clinics in the United States. Symptoms include heartburn and regurgitation and it may be responsible for causing inflammation of the esophagus.13
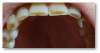
The most common oral manifestation of GERD (and other causes of stomach contents reaching the mouth) is dental erosion. Tooth erosion, also known as erosive tooth wear, is usually a slow process that occurs over many years and is actually a combination of processes both chemical and mechanical (Figure 6).13
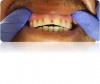
Tooth erosion from GERD occurs as a result of exposure of the teeth to hydrochloric acid from the stomach. This erosion, in conjunction with abrasive factors such as tooth wear, mastication, toothbrushing, etc, typically manifests as ditching or cupping of teeth on the incisal edges and lingual surfaces of the maxillary anterior teeth and the cusps of the occlusal surfaces of both maxillary and mandibular posterior teeth. This "ditching" effect is different than a patient ingesting large quantities of carbonated beverages, which typically results as erosion of the labial or facial surfaces of the anterior teeth (Figure 7). The lesions caused by GERD are hard to the touch with an explorer instrument, as opposed to decay.14
Several interventions may be implemented to treat or minimize these oral lesions caused by GERD. Salivary stimulants and/or substitutions of salivary secretions may help with reducing the effects on the teeth. Diet modifications can lessen the acidity, as can the simple act of rinsing the mouth with water after meals and/or regurgitation to remove residual acids. Sodium bicarbonate can be added to the water making a more alkaline rinse for additional neutralization of the oral cavity. Patients should refrain from brushing immediately following a reflux episode, to allow for the re-establishment of salivary pellicle and subsequent tooth surface remineralization.13 Also, the use of stannous fluoride provides a barrier on the tooth surfaces to combat acid attack.12 There is research that also shows that the use of amorphous calcium phosphate (ACP), as well as casein phosphopeptide (CCP), is beneficial in assisting with relief from the detrimental effects of and/or the reduction in lesions caused by GERD.15,16Physical barriers such as sealants and minimally invasive restorations, as well as products to dissipate hypersensitivity of the eroded areas, assist with reducing the effects of erosion.
Further oral manifestations caused by GERD may include aphthous type ulcerations, burning mouth syndrome, palatal erythema, epithelial atrophy, and xerostomia. Xerostomia may be caused not only by the disease but also by the medications used to treat GERD.17 Treatment for these conditions can include various mouth rinses and medicaments; however, management of the reflux disorder is the primary course of treatment to alleviate these oral manifestations.
Inflammatory Bowel DiseasE
Inflammatory bowel diseases (IBD) have caused significant oral manifestations. IBD is separated into two classifications: Crohn's disease and ulcerative colitis.18 Both diseases may share similar symptoms; however, they are not the same illnesses. Crohn's and colitis affect different areas of the GI tract. Crohn's disease inflammation may occur at any part of the GI tract (mouth to anus), whereas ulcerative colitis is typically confined to the colon.
Crohn's disease presents as patchy inflammation throughout the small and large bowels. It has an age onset from 15 to 35 years and 55 to 70 years. Depending on the location of the disease, its symptoms may include abdominal pain, diarrhea, weight loss, and fatigue. In addition, bloody stool could be present. Crohn's disease also shows the presence of granulomas and affects all layers of the bowels, whereas ulcerative colitis only affects the mucosal lining.18,19
Crohn's causes narrowing of the bowel, which has a cobblestone appearance due to polypoid lesions occurring in the bowel. It also demonstrates skip lesions. Treatment for Crohn's includes anti-inflammatory medications, steroids, and immunosuppressants.
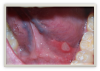
Oral lesions resulting from Crohn's include aphthous-type ulcerations (Figure 8), cobblestone mucosa, lip/facial swelling, orofacial granulomatosis, mucosal tags, xerostomia, angular cheilitis, and glossitis.18,19Treatment of the lesions can range from prescription drugs to homeopathic types of treatments, such as edible oils, acupuncture, and herbal remedies like ginseng. Dental professionals should work in tandem with the patient's primary care physician in order to identify the most effective treatment plan for that individual.
There are several oral lesions associated with ulcerative colitis. The most common lesion with either undiagnosed or active ulcerative colitis is pyostomatitis vegetans. This lesion is rarely seen in patients with Crohn's. Other lesions include aphthous-type ulcerations, glossitis, cheilitis, and gingival inflammation and pyostomatitis.20,21
Treatment for these lesions requires management of the systemic conditions, along with using topical steroid gels and over-the-counter rinses and medications.
Peutz Jegher's Syndrome
There are many conditions that are known to produce intraoral pigmentations, such as developmental racial pigmentation and oral melanotic macule, drug therapy, metal impregnation, smoker's melanosis, and several systemic diseases. Among the various systemic diseases that manifest intraoral pigmentations are Addison's disease, Albright's syndrome, Gardner's syndrome, Kaposi's sarcoma, oral melanoma, and Peutz Jegher's Syndrome (PJS).11
Also known as hereditary intestinal polyposis, Peutz Jeger's Syndrome is a hereditary condition characterized by the development of intestinal polyps. Prevalence in the US ranges from 1 in 50,000 to 1 in 200,000.11,22 The polyps associated with PJS are histologically distinct, typically begin developing in childhood and are most common in the small bowel and colon. The GI polyps may cause GI bleeding, anemia and abdominal pain due to intussusception, obstruction or infaction.22PJS is known to produce perioral and intraoral pigmentations as well as pigmented areas on the hands and feet (Figure 8).11,23 Oral melanotic macules are common and may be the first clinical sign of the disease, placing dental professionals on the front line of diagnosis of this condition. Diagnosis is based on family history, detection of polyps through endoscopy and/or colonoscopy, and mucocutaneous pigmentation.
The magnitude of the condition lies with the propensity for carcinomatous change of the intestinal polyps over time. Persons with PJS have a significantly increased risk of developing intestinal cancer as well as cancers of other organs.11,22,23 Treatment requires monitoring polyps for carcinomatous change, with no routinely recommended treatment for the pigmentations. If objectionable esthetically, however, laser therapy can be an option.23
The Role of the Dental Professional
The dental practitioner's role in detecting oral manifestations is significant. They can become more involved in whole body wellness, as they possess the capability to recognize oral manifestations that may be signs that a systemic issue is present. The clinician should be a primary investigator, involved in collaborative interdisciplinary care for patients. The oral abnormality screening should take precedence within the patient assessment procedure. Checking both extraorally and intraorally, as well as cross-checking and referencing the patient's medical history, is mandatory.
Finally, beyond detecting and identifying oral symptoms, the dental professional also plays an important role by understanding when and how to make recommendations that will benefit the patient's oral health and mitigate oral effects of systemic issues, with treatments such as sealants, rinses, fluorides, varnishes, and more.
About the Author
Sherri Lukes, RDH, MS, FAADH
Independent speaker/consultant
References
1. Sonawane K, Suk R, Chiao EY, et al. Oral human papillomavirus infection: Differences in prevalence between sexes and concordance with genital human papillomavirus infection, NHANES 2011 to 2014. Annals of Internal Medicine.2017;167(10)714-725.
2. Mannarini L, Kratochvil V, Calabrese L,et al. Human papilloma virus (HPV) in head and neck region: review of literature. Acta Otorhinolaryngol Ital. 2009;29(3):119-126.
3. Centers for Disease Control and Prevention. HPV and Cancer. Updated August 2, 2019. CDC.gov/cancer/hpv/statistics/cases.htm. Accessed August 4, 2020.
4. Gillison ML, Broutian T, Pickard RKL, et al. Prevalence of oral HPV infection in the United States, 2009-2010. JAMA. 2012;307(7):693-703.
5. Burkhart NW. Squamous cell carcinoma. RDH. March 1, 2009. https://www.rdhmag.com/career-profession/personal-wellness/article/16405085/squamous-cell-carcinoma. Accessed August 4, 2020.
6. Huang SH, O'Sullivan B, Waldron J. The current state of biological and clinical implications of human papillomavirus-related oropharyngeal cancer. Semin Radiat Oncol. 2018;28(1):17-26.
7. Huang SH, O'Sullivan B. Overview of the 8th Edition TNM Classification for Head and Neck Cancer. Curr Treat Options Oncol. 2017;18(7):40.
8. Nikitakis NG, Pentenero M, Georgaki M, et al. Molecular markers associated with development and progression of potentially premalignant oral epithelial lesions: Current knowledge and future implications. Oral Surg Oral Med Oral Pathol Oral Radiol. 2018;125(6):650-669. doi: 10.1016/j.oooo.2018.03.012. Epub 2018 Apr 4. PMID: 29709496.
9. The Joanna Briggs Institute. Best Practices. Prevention and Treatment of Oral Mucositis in Cancer Patients. 1998. https://oralcancerfoundation.org/wp-content/uploads/2016/09/mucositis.pdf. Accessed August 5, 2020.
10. Prabhu SR, Wilson DF. Human papillomavirus and oral disease - emerging evidence: a review. Australian Dental Journal. 2013;58(1):2-10.
11. Neville BW, Damm DD, Allen CM, Chi AC. Oral and Maxillofacial Pathology. 4th ed. Philadelphia: Saunders; 2015.
12. Richter JE, Rubenstein JH. Presentation and epidemiology of gastroesophageal reflux disease. Gastroenterology. 2018;154(2):267-276.
13. Ranjitkar S, Smales RJ, Kaidonis JA. Oral manifestations of gastroesophageal reflux disease. J Gastroenterol Hepatol. 2012;27(1):21-27.
14. Fumagalli LA, Gatti H, Armano C, et al. Oral pathology unmasking gastrointestinal disease. Dent Health Oral Disord Ther. 2016;5(5):335-339.
15. Lussi A, Buzalaf MAR, Duangthip D, et al. The use of fluoride for the prevention of dental erosion and erosive tooth wear in children and adolescents. Eur Arch Paediatr Dent. 2019;20(6):517-527.
16. Afonso M, Buzalaf R, Magalhães AC, Wiegand A. Alternatives to fluoride in the prevention and treatment of dental erosion. Monogr Oral Sci. 2014;25:244-252.
17. Campisi G, Russo LL, Di Liberto C, et al. Saliva variations in gastro-oesophageal reflux disease. J Dentistry. 2008;36(4):268-271.
18. Crohn's & Colitis Foundation website. https://www.crohnscolitisfoundation.org. Accessed August 5, 2020.
19. Pedersen AML. Diseases Causing Oral Dryness. In: Carpenter G, ed. Dry Mouth. Springer-Verlag Berlin Heidelberg; 2015:7-31.
20. Jajam M, Bozzolo P, Niklander S. Oral manifestations of gastrointestinal disorders. J Clin Exp Dent. 2017;9(10):e1242-e1248.
21. Pocket Dentistry. Mucosal Manifestations of Gastrointestinal Disease. https://pocketdentistry.com/mucosal-manifestations-of-gastrointestinal-disease. Accessed August 5, 2020.
22. Beggs AD, Latchford AR, Vasen HFA, et al. Peutz-Jeghers syndrome: a systematic review and recommendations for management. Gut. 2010;59:975-986.
23. Langlais RP, Miller CS. Color Atlas Common Oral Diseases. 3rd ed. Michigan: Williams & Wilkins; 1998.