You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Professor Wilhelm Conrad Roentgen discovered x-rays, a form of ionizing radiation, on November 8, 1895, more than 120 years ago. Although Roentgen conducted numerous experiments to determine the properties and characteristics of x-rays, the deleterious effects of x-radiation were not discovered initially. Soon thereafter, it was observed that lengthy exposure to the x-ray beam resulted in burns, skin alterations and hair loss.
In 1898, Dr. William Rollins, a Harvard graduate and Boston dentist, received a severe burn on his arm while exposing his hand to the x-ray beam. This event stimulated his interest and research in radiation protection. In 1901, he published the results of his experiments on guinea pigs, which demonstrated the association between x-ray exposure and biological damage.1 As a result, he recommended several precautions: wear leaded glasses, enclose the x-ray tube in a leaded housing, record only the area of interest and shield the rest.1 In addition, he suggested filtration of the x-ray beam and developed rectangular collimation to restrict the size of the x-ray beam. He published over 200 articles on x-radiation hazards in Notes on X-Light from 1897-1908.1 Although his safety suggestions were ignored, his recommendations are elements of current radiation safety and protection measures. Rollins, like many other early pioneers, contributed to the understanding and prevention of the biologic effects of x-radiation.
Now there are a variety of international and national organizations and agencies, some with regulatory responsibilities, that analyze data, publish reports and disseminate information on radiation protection, risk estimates and units of measurement. On the international level are the International Commission of Radiological Protection (ICRP), the United Nations Scientific Committee on the Effects of Atomic Radiation (UNSCEAR) and the International Commission on Radiation Units and Measurements (ICRU).2 On the national level are the Biological Effects of Ionizing Radiations (BEIR) Committee, the National Council on Radiation Protection and Measurements (NCRP), the Nuclear Regulatory Commission (NRC), the Environmental Protection Agency (EPA), the Occupational Safety and Health Administration (OSHA) and the National Center for Devices and Radiological Health (NCDRH), the U. S. Food and Drug Administration (FDA) and the U. S. Department of Health and Human Services (HHS).2 A rather sophisticated network of organizations, agencies and bureaus deal with varying aspects of radiation and radiation protection. In addition, there are federal and state laws that regulate dental x-ray equipment, x-ray machine inspection and the credentialing of dental radiographers.
This brief summary demonstrates how the study of radiation biology began and how radiation safety and protection measures evolved from early research efforts to formally established organizations that examine leading scientific thought and provide guidance and recommendations on radiation protection and measurement. The subsequent information will provide an overview of radiation biology and the safety and protection measures that the dental assistant can implement to reduce radiation exposure to dental patients as well as minimize occupational exposure. This information applies to both film-based and digital radiography.
Radiation Biology
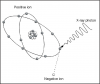
Radiation biology is defined as the study of the effects of ionizing radiation on biological systems. Understanding the effects is essential to the safe and effective use of radiation for diagnostic and therapeutic purposes. The human organism is configured in an increasingly complex manner from atoms, to molecules, cells, tissues, organs and systems. Interaction of x-radiation with human tissue occurs at the atomic level through excitation and, more commonly, ionization. (Figure 1) When an atom is ionized, its chemical binding properties are altered. If the atom is part of a large molecule, ionization may result in breakage of the molecule or a change in location of the atom within the molecule.3 These alterations may impair function and result in cell death. However, cells and tissues can repair, regenerate and recover. Early effects of radiation are injuries that occur within minutes, hours and days while late effects are those injuries that occur within months, years and decades after exposure.
Radiation acts on biologic systems indirectly and directly through the processes of ionization and free radical production. An indirect effect occurs as a result of the radiolysis (breakdown) of water and the production of free radicals while a direct effect occurs when the ionizing radiation interacts directly with a particularly radiosensitive molecule like deoxyribonucleic acid (DNA).3
Indirect effect of radiation
Water is the predominant molecule in a biologic system, approximately 80% by weight. As a result, irradiation of water represents the primary radiation interaction in the body and is the most likely pathway for biologic damage to occur.3 Ionization or radiolysis of H2O results in dissociation or separation into two ions or an ion pair (positively charged water molecule and displaced electron). A number of complex reactions can occur after the initial ionization event. The ion pair may reform into a stable water molecule without any damage. On the other hand, if the ions do not rejoin, the negative ion can attach to another water molecule to produce a third type of ion, a negatively charged water molecule.3 As a result, a series of unstable molecules are formed which rapidly disjoin to form reactive products called free radicals, largely hydrogen (H*) and hydroxyl (OH*) free radicals. Free radicals are uncharged or neutral atoms with a single unpaired electron in the outer electron shell and, thus, are very reactive. Free radicals contain excess energy that can be transferred to other molecules to disrupt bonds, produce point lesions and form products poisonous to the cell.4 A hydroxyl (OH*) free radical can join with another to form hydrogen peroxide (H2O2) and the hydrogen (H*) free radical can interact with molecular oxygen (O2) to form the hydroperoxyl radical (HO2*). These two products are considered to be the primary damaging agents resulting from the radiolysis of water.4 Other free radicals can be produced by the interaction of hydrogen and hydroxyl free radicals with organic molecules. Organic free radicals are unstable and transform into stable altered molecules that have different chemical and biological properties than the original molecules.6 Since free radicals and altered biomolecules can migrate freely through the tissues, the indirect effects of radiation can manifest at sites distant from the original exposure. In summary, the ionization of the water molecule can change the original configuration, function, properties and products of the molecule. The indirect effect accounts for approximately 2/3 of radiation induced biologic damage.
Direct effect of radiation
The direct effect occurs when the energy of the photon or secondary electron ionizes a biologic macromolecule such as a protein, lipid, carbohydrate, or nucleic acid.3 Protein and nucleic acid synthesis is essential to cell reproduction and survival. Radiation damage to these macromolecules may result in late effects or cell death. Cells that produce more macromolecules are less radiosensitive. For instance, proteins are more abundant and less radiosensitive while DNA is not very abundant and is the most radiosensitive.3 Because it contains the genetic information for each cell, DNA is the most important macromolecule. The cell nucleus holds the DNA and chromosomes that control growth and development of the cell. Radiation may break DNA strands, alter base sequence, disrupt molecular bonds and cause cross-linking of DNA strands.4
When macromolecules are irradiated, three major effects occur; main-chain scission, cross-linking and point lesions.3 Main-chain scission is a cutting or breakage in the long-chain molecule that divides a long, single molecule into many molecules. This breakage reduces the size of the molecule and changes the viscosity of the macromolecule solution. In cross-linking, irradiation of a macromolecule can produce side spurs that will attach to another segment of the molecule or a neighboring molecule. Although some macromolecules have side spurs, those created by irradiation increase the viscosity of the macromolecule solution. In addition, irradiation can produce point lesions. Point lesions occur at the point of a single chemical bond and can cause impairment or loss of cell function. These effects are reversible through repair and recovery. Altered macromolecules differ structurally and functionally from the original macromolecules. Similar to the radiolysis of water, irradiation of macromolecules can produce changes that alter their original configuration and function. The direct effect accounts for 1/3 of radiation induced biologic damage.
Short and long-term effects
The effects of radiation are not evident immediately. There is a delay between irradiation and the appearance of biologic damage. This delay or time interval is known as the latent period. The actual length of the latent period depends on the total dose and delivery rate. Generally speaking, a high dose delivered over a short period of time will result in a brief latent period. Short-term, early or acute effects may occur minutes, hours, or weeks following exposure. Usually short-term effects are the result of high doses of radiation to the whole body. The symptoms include nausea, vomiting, diarrhea, fever, hair loss, hemorrhage and total collapse. The ultimate early effect is death. Long-term, late or chronic effects may occur months, years, or decades following exposure. Long-term or chronic effects are usually the result of low doses of radiation received over a long period of time. These effects may not be observable for months, years and decades and may result in cancers later in life.
The effects of radiation are cumulative or additive. Although tissues have the capacity to repair damage, some damage is not repairable and accumulates in the tissues. The cumulative effect is residual injury without repair from repeated radiation exposure. Low doses received by patients from dental radiography produce very little damage yet some damage does occur. This unrepaired damage may lead to health problems later in life such as cataracts, cancer, leukemia, genetic abnormalities and congenital defects. It is generally accepted that the benefits of diagnostic imaging outweigh the risks and that the risks are small.5 However, the actual risk associated with low dose diagnostic exposures is not currently known.6,7 Therefore it is prudent for the clinician to keep radiation exposure to patients to a minimum.
Factors that influence radiation effects
There are a variety of other factors that influence the biologic effects of ionizing radiation. The most significant factors will be presented and include radiosensitivity, linear energy transfer, dose factors, area of exposure, somatic and genetic effects and stochastic and non-stochastic effects.
Radiosensitivity
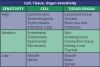
Approximately ten years following the discovery of x-rays, French radiobiologists Jean Bergonié and Louis Tribondeau described the types of cells most sensitive to the effects of radiation. They observed that the most susceptible cells demonstrate a high mitotic rate, long history of cell division, are immature and undifferentiated (not highly specialized) and have a large nucleus to cytoplasm ratio. As a result, some cells, tissues and organs are more sensitive to the effects of radiation than others. Table 1 outlines those tissues that are highly sensitive, moderately sensitive and those that have low sensitivity to ionizing radiation. These observations have become to be known as the Law of Bergonié and Tribondeau.
Linear Energy Transfer
Linear energy transfer (LET) is the measure of the rate at which energy is transferred from the incident radiation to tissue along the path the radiation is traveling. As the LET increases, so does the biologic damage. This is known as the relative biologic effectiveness (RBE). For example, protons and alpha particles have high LET and are more damaging than x-rays. X-rays have a relatively low LET and are sparsely ionizing. Therefore, they ionize relatively few atoms and/or biomolecules and are less likely to cause a direct biologic effect.
Dose Factors
Dose factors have a bearing on the biologic effects of radiation. Dose is defined as the amount of radiation absorbed per unit mass of tissue. Generally speaking, the severity of damage is dependent upon the amount of radiation received. The dose rate is the radiation dose delivered per unit time. The higher the dose rate, the greater the damage done. Lower dose rates allow greater opportunity for repair and less net damage. The total radiation dose can be fractionated or divided into multiple smaller doses delivered over a period of time. This approach is used in radiotherapy as a means of allowing cellular repair and enabling the patient to survive high doses of radiation that are needed to kill tumor cells.
Area of Exposure
The volume of tissues exposed to radiation is an important factor as well. The body can tolerate rather high doses (fractionated, 4000-5000 rad/40-50 Gy) to a localized or restricted area of the body. This is one of the principles involved in radiotherapy. One relevant example of a localized area of exposure is therapeutic radiation for head and neck cancer. A variety of tissues would be in the field of radiation such as skin, mucous membranes, taste buds, salivary glands, teeth, muscle and bone. Localized biologic effects could include erythema, mucositis, changes in taste acuity, xerostomia, radiation caries, muscle atrophy and osteoradionecrosis. On the other hand, high doses of radiation to the whole body are usually fatal. A whole body dose of 2 Gy (200 rad) is usually enough to cause death within several days to weeks.3 Diagnostic x-ray beams are not intense or large enough to cause death but accidental exposure from a nuclear reactor meltdown could produce a lethal whole body dose.4 There are three different syndromes associated with high-level radiation exposure to the whole body, hematological (2-10 Gy/200-1000 rad), gastrointestinal (10-50 Gy/1000-5000 rad) and central nervous system (>50 Gy/5000 rad).3 They are related to the dose received and follow a particular sequence of events called the acute radiation syndrome. There is a prodromal period during which acute clinical symptoms occur followed by a latent period when there is no observable sign of radiation sickness. This is followed by manifest illness, a dose-related period during which the three syndromes occur. The survival time following whole-body radiation is dependent upon the amount of exposure. As the dose increases, the average time between exposure and death decreases. At doses above 10 Gy, death is certain and typically occurs within four days.3
Somatic and Genetic Factors
The types of tissues that are exposed to radiation have an influence on the biologic effects of radiation. Generally, the body is divided into two tissue categories, somatic and genetic. Somatic tissues include all tissues of the body except reproductive. The somatic effects of radiation are not passed along to future generations but affect the irradiated individual. Small exposures produce no observable damage but at increasingly higher doses, a threshold will be reached when specific effects are observable. For instance, an individual who receives a localized dose in the 3 to 6 Gy (300 to 600 rad) range will exhibit an erythema of the skin within the first or second day.3 After a brief latent period, the erythema reappears followed by skin desquamation and epilation. These types of somatic effects follow a nonlinear, threshold dose-response in which higher doses produce progressively more severe damage. Dental exposures are well below threshold doses and will not produce any observable effects. Genetic tissues include reproductive cells and embryonic tissues. The genetic or heritable effects of radiation occur only in reproductive cells and can be passed to future generations. These effects do not affect the irradiated individual but rather offspring. These effects include mutation and effects on the embryo that may not be expressed for years or generations. Genetic effects are believed to occur in a linear, non-threshold dose-response. This means that even small amounts have the potential to produce some mutations. The more the dose increases, the more mutations are produced. Therefore, no dose is considered safe. Gonadal absorbed doses from typical dental radiographic projections rarely, if ever, deliver any measurable dose to the embryo or fetus.8
Stochastic and Non-Stochastic Effects
The biologic effects of radiation can be described as stochastic (chance) and non-stochastic or deterministic. With regard to stochastic effects, the probability of the occurrence of an effect rather than the severity is proportional to the dose. These effects include cancer, mutation and effects on the embryo and do not have dose thresholds. Stochastic effects are all or none; a person either has or does not have the condition.4 The primary concern in dental radiology is cancer rather than reproductive cell mutation or embryonic alterations. With non-stochastic effects, the severity of the response is a function of the radiation dose. A threshold dose must be reached before an effect is observed. Non-stochastic effects occur in all people when the dose is large enough. These effects are proportional to dose, dose rate and the volume of tissues exposed. Dental exposures are well below threshold doses.
Units of radiation measurement
There are two different systems of nomenclature for describing radiation quantities, International or SI units (Système International D’Unités) and standard or traditional units. SI units include the coulomb/kilogram, gray, Sievert and bequerel and traditional units include the roentgen, rad, rem and curie. The SI units have replaced the use of traditional units. Table 2 outlines the most typical units used to measure quantities of radiation.
Exposure
Exposure is a measurement of radiation quantity and refers to the ability of x-rays to ionize air. This is the amount of radiation that is emitted from the x-ray tube and reaches the patient. The measurement is taken at the skin surface before radiation has penetrated the patient’s tissues to measure the intensity of the radiation. The traditional unit for exposure is the roentgen (R) and it measures the amount of x-ray or gamma radiation capable of ionizing 1.6 x 1012 ion pairs per gram of air. Although there is no specific SI unit, 1 roentgen would be equal to 2.58 x 104 C/kg of air and 1 C/kg = 3.88 x 103 R (3876 R).
Air Kerma
Kerma (kinetic energy released in matter) is the unit of radiation exposure or intensity measured in joules per kilogram in which 1 J/kg is equivalent to 1 Gya. This measurement has, for the most part, replaced the use of coulombs/kg and/or the roentgen (R). Air kerma represents the kinetic energy that is transferred from photons to electrons during the processes of ionization and excitation. Air kerma values represent those that are measured in air. The unit Gya is used to indicate radiation dose in air.
Absorbed Dose
The absorbed dose is the measurement of the quantity of any type of ionizing radiation received by a mass of any type of matter. This is the amount of radiation that is actually absorbed by the patient’s tissues. The biologic effects of radiation are related to the absorbed dose. The SI unit is the gray (Gyt) and the rad is the traditional unit for the absorbed dose. The unit Gyt is used to represent radiation dose in tissue. One gray (Gyt) is equal to 1 J/kg, while the rad is equal to the transfer of 100 ergs per gram of tissue. One gray is equivalent to 100 rads. To convert from SI units to traditional, multiply the gray (Gyt) by 100 to equal the rad. Absorbed dose units may be expressed in smaller units such as cGy or mGy. (Table 3)
Equivalent Dose
The equivalent dose (HT) is a measurement used to compare the biologic effects or damage an exposed individual might expect to occur from different types of ionizing radiation. The equivalent dose is the mean absorbed dose in a tissue or organ modified by the weighting factor (WT) for the type and energy of the radiation under consideration.8 As previously discussed, the LET varies among different types of radiation which has an impact on the biologic effects. For example, the weighting factor for x-rays is one, which means that x-rays are less effective in producing biologic effects than alpha particles, which have a weighting factor of 20. The sievert (Sv) is the SI unit for the equivalent dose and the rem (roentgen equivalent man) is the traditional unit. To convert from SI to traditional units, multiply the sievert by 100 to equal the rem. The equivalent dose may be expressed in smaller units such as mSv or μSv (Table 3). The equivalent dose is used to quantify the occupational exposure of radiation workers.
Effective Dose
The effective dose (E) is used to estimate risk or assess biologic consequences in humans. It is the sum of weighted equivalent doses for the radiosensitive tissues of the body expressed as E = Σ WT x HT. WT is the tissue weighting factor for tissue T and HT is the equivalent dose in tissue organ T.8 The tissue weighting factors vary, for example the weighting factor for the gonads is 0.08 while the weighting factor for thyroid is 0.04.17 (Table 4) The unit of measurement for the effective dose is the sievert (Sv).8
Radioactivity
Radioactivity is the emission of particles and energy to achieve stability. Some atoms exist in an excited state due to an unstable nucleus. This process is known as radioactive decay or disintegration. Alpha particles come from the nucleus of heavy elements and beta particles from the nucleus of radioactive atoms. Both are forms of ionizing particulate radiation. The SI unit of measurement for radioactivity is the becquerel (Bq) and the traditional unit is the curie (Ci). The becquerel is the activity of a quantity of radioactive material in which one nucleus disintegrates every second (1 Bq = 1 ds).
Risk summary
Dental radiographic x-ray examinations are not without risk. However, the risk is small in terms of other risks readily assumed in daily life that cannot be avoided. Every day humans are exposed to background radiation from the environment. Natural radiation or background radiation results from cosmic and terrestrial radiation including radon and internal source radiation. It accounts for approximately 50% of the average annual effective dose, 3.1 mSv.9 Humans are also subject to artificial sources of radiation which include medical and dental applications such as computed tomography, nuclear medicine, interventional and conventional radiography and fluoroscopy and other sources such as consumer products, occupational exposure and industrial products. Artificial sources account for nearly the same percentage and average annual effective dose as natural background radiation.9 It is important to note that radiation exposure from artificial sources has trended upward in recent years. This is due to the increased use of medical applications, particularly computed tomography (CT) and nuclear medicine examinations.10 Previously, medical imaging represented about one-sixth the amount of natural background radiation.7 Although dental applications contribute to only 2.5% of the conventional radiography/fluoroscopy category, this does not include cone beam computed tomography (CBCT) which is being used with increasing frequency in dentistry.7 In like manner, the annual effective dose per individual in the U. S. has increased from 3.6 to 6.2 mSv.7 To put this in perspective, several dental imaging examples using background radiation equivalence follow. A full mouth survey taken with F speed film or photostimulable phosphor plates using rectangular collimation is equivalent to 4.3 days of background radiation while a direct digital panoramic image is equivalent to 1.7 to 3.0 days (depending on the system) of background radiation.11
Another way to express risk is the probability of stochastic effects in which the occurrence of an effect increases as the dose of radiation exposure increases. The primary risk from dental radiography is radiation-induced cancer. The risk of cancer as a result of low dose radiation is very difficult to estimate. Chiefly, risks have been estimated by extrapolation of high dose data from Japanese atomic bomb survivors and other exposed populations. The generally accepted model to estimate risk is the linear non-threshold (LNT) dose hypothesis.11 The LNT hypothesis presents a linear relationship between dose and the risk of cancer induction in which there is no threshold dose below which there is no additional risk. In other words, there is no so called “safe dose.”
In 2007, the ICRP published new recommendations which replace those issued in Publication 60 in 1991.12 The 2007 Recommendations update the radiation and tissue weighting factors (Table 4) in the quantities equivalent and effective dose, radiation detriment and the method for calculating effective dose based on the latest scientific information available on the biology and physics of radiation exposure.12,13 The revised tissue weighting factors include tissues in the maxillofacial area including the extrathoracic region, lymph nodes, muscle and oral mucosa. In 2008, Ludlow et al, studied the impact of the new tissue weighting factors on the effective dose of common dental radiographic examinations and their detriment.11 The results of the study demonstrated increased effective doses for dental radiographic examinations when using the 2007 ICRP calculation method.11 The risks associated with dental radiography were 32% to 422% higher than when using 1990 ICRP guidelines.11 For example, a full mouth survey taken with D speed film and round collimation results in a 20 per million risk of death while the same survey with F speed film or PSP receptors and rectangular collimation reduced the risk ten fold.11 The outcomes of the study suggest an increased possibility of risk with dental exposures and reinforce the need to practice patient dose reduction measures as recommended by the American Dental Association and NCRP.8,14
In summary, the Committee to Assess Health Risks from Exposure to Low Levels of Ionizing Radiation (BEIR VII) concluded that the preponderance of evidence indicates that there is some risk at low doses but the risk is small.15 In addition, the NCRP concluded that radiation risks to dental patients and operators are very small and may be zero.8 However, until there is clear evidence for a threshold dose below which dental patients are not at risk, dental health professionals have an obligation to help patients avoid unnecessary radiation exposure. Methods to reduce radiation exposure to the patient will be discussed in the ongoing text.
Selection Criteria - Recommendations for Patient Selection
Guidelines for prescribing dental radiographs were first developed by the Food and Drug Administration (FDA) in 1987 to promote appropriate use of radiographic examinations in dentistry and to respond to concern about the U.S. population’s total exposure to radiation from all sources.16 After many years of use, the American Dental Association and the U.S. Department of Health and Human Services updated the document in 200417 and most recently in 2012.14 The underlying principles remain the same but the latest document includes recommendations with regard to the evaluation of dentoalveolar trauma, careful interpretation of radiographs, complete record review prior to conducting a radiographic examination as well as methods to limit radiation exposure to patients and clinicians alike.
The guidelines for prescribing radiographs or selection criteria include the following concepts: type of encounter (new or recall), patient age (child, adolescent, adult), stage of dental development (primary, transitional, permanent dentitions; partially edentulous, edentulous), vulnerability to known risk factors (caries, periodontal disease), growth and development monitoring and/or assessment of dental/skeletal relationships and other circumstances.14 The overarching theme is that the dentist should conduct a thorough clinical examination with consideration given to the patient’s medical and dental history; review of any prior radiographs, caries risk assessment and the patient’s dental and general health needs before prescribing a radiographic examination.
The use of selection criteria guidelines is a primary concept in reducing patient radiation exposure. Patient examinations should be individualized and only necessary, high yield radiographs should be taken rather than unjustified routine radiographs. Routine, fixed schedules for radiographic examinations for all patients are not recommended except as outlined in the bitewing (horizontal or vertical) guidelines presented below and reduces overall radiation exposure.
There are a number of indicators that the dentist can use to determine the need for dental radiographs. These indicators include the risk for caries, historical findings and positive clinical signs and symptoms. Indicators that would suggest that a patient is at high risk for caries may include evidence of poor oral hygiene, clinical or radiographic caries, recurrent caries, inadequate fluoride exposure, high sucrose diet, radiation therapy and xerostomia. Positive historical findings such as a history of tooth pain or trauma, previous periodontal, endodontic or implant treatment or a family history of dental anomalies may indicate a need for radiographs. In addition, positive clinical signs and symptoms observed during the clinical examination provide evidence that radiographs may be indicated. Examples of positive clinical signs and symptoms include clinical evidence of periodontal disease and/or carious lesions, large or deep restorations, tooth mobility, trauma, unusual eruption patterns and unexplained missing teeth.
The updated guidelines for the selection of patients for dental radiographic examinations are summarized in Table 5 and outlined in following text using the general framework of the type of encounter, and patient age and dental development stage. Under the types of encounters, clinical circumstances and oral diseases are presented. These include clinical caries or the increased risk for caries, no clinical caries or no increased risk for caries as well as periodontal disease or a history of periodontal treatment, monitoring of dentofacial growth and development and/or assessment of dental/skeletal relationships and other circumstances. Several examples of the latter category include existing implants, dental or craniofacial pathoses, restorative and endodontic needs, treated periodontal disease and caries remineralization. Applicable to all categories are the concepts of radiographic evaluation of dentoaveolar trauma; the thorough examination of all radiographs for any evidence of caries, alveolar bone loss, developmental anomalies and occult disease; prohibition of using radiographic screening prior to a clinical examination; and conduction of a thorough clinical examination, review of the patient history, prior radiographs, caries risk assessment and consideration of dental and general health needs of the patient prior to performing a radiographic examination.
Child Patient with a Primary Dentition – prior to eruption of first permanent tooth
New patient being evaluated for oral diseases: Individualized radiographic exam consisting of selected periapical/occlusal views and/or posterior bitewings if proximal surfaces cannot be visualized or probed. Patients without evidence of disease and with open proximal contacts may not require a radiographic exam at this time.
Recall patient with clinical caries or increased risk for caries: Posterior bitewings at 6 to 12 month intervals if proximal surfaces cannot be examined visually or with a probe.
Recall patient with no clinical caries and no increased risk for caries: Posterior bitewings at 12 to 24 month intervals if proximal surfaces cannot be examined visually or with a probe.
Recall patient with periodontal disease: Clinical judgment as to the need for and type of radiographic images necessary for evaluation of periodontal disease. Imaging may consist of, but is not limited to, selected bitewings and/or periapical images of areas where periodontal disease (other than nonspecific gingivitis) can be demonstrated clinically.
Patient (New or Recall) for the monitoring of growth and development and/or assessment of dental/skeletal relationships: Clinical judgment as to the need for and type of radiographic images for evaluation and/or monitoring of dentofacial growth and development or assessment of dental skeletal relationships.
Patient with other circumstances including but not limited to, proposed or existing implants, other dental and craniofacial pathoses, restorative/endodontic needs, treated periodontal disease and caries remineralization: Clinical judgment as to the need for and type of radiographic images for evaluation and/or monitoring of these conditions.
Child Patient with a Transitional Dentition – after eruption of first permanent tooth
New patient being evaluated for oral diseases: Individualized radiographic exam consisting of posterior bitewings with a panoramic exam or posterior bitewings and selected periapical images.
Recall patient with clinical caries or increased risk for caries: Posterior bitewings at 6 to 12 month intervals if proximal surfaces cannot be examined visually or with a probe.
Recall patient with no clinical caries or no increased risk for caries: Posterior bitewings at 12 to 24 month intervals if proximal surfaces cannot be examined visually or with a probe.
Recall patient with periodontal disease: Clinical judgment as to the need for and type of radiographic images for evaluation of periodontal disease. Imaging may consist of, but is not limited to, selected bitewings and/or periapical images of areas where periodontal disease (other than nonspecific gingivitis) can be demonstrated clinically.
Patient (New or Recall) for monitoring of dentofacial growth and development and/or assessment of dental/skeletal relationships: Clinical judgment as to the need for and type of radiographic images for evaluation and/or monitoring of dentofacial growth and development or assessment of skeletal relationships.
Patient with other circumstances including but not limited to, proposed or existing implants, other dental and craniofacial pathoses, restorative/ endodontic needs, treated periodontal disease and caries remineralization: Clinical judgment as to the need for and type of radiographic images for evaluation and/or monitoring these conditions.
Adolescent Patient with a Permanent Dentition – prior to eruption of third molars
New patient: Individualized radiographic exam consisting of posterior bitewings with a panoramic exam or posterior bitewings and selected periapical images. A full mouth intraoral radiographic exam is preferred when the patient has clinical evidence of generalized oral disease or a history of extensive dental treatment.
Recall patient with clinical caries or increased risk for caries: posterior bitewings at 6 to 12 month intervals if proximal surfaces cannot be examined visually or with a probe.
Recall patient with no clinical caries or no increased risk for caries: Posterior bitewing exam at 18 to 36 month intervals.
Recall patient with periodontal disease: Clinical judgment as to the need for and type of radiographic images for the evaluation of periodontal disease. Imaging may consist of, but is not limited to, selected bitewing and/or periapical images of areas where periodontal disease (other than nonspecific gingivitis) can be demonstrated clinically.
Patient (New or Recall) for monitoring of dentofacial growth and development, and/or assessment of dental/skeletal relationships: Clinical judgment as to the need for and type of radiographic images for evaluation and/or monitoring of dentofacial growth and development, or assessment of dental and skeletal relationships. Panoramic or periapical exam to assess developing third molars.
Patient with other circumstances including, but not limited to, proposed or existing implants, or other dental and craniofacial pathoses, restorative/ endodontic needs, treated periodontal disease and caries remineralization: Clinical judgment as to the need for and type of radiographic images for evaluation and/or monitoring of these conditions.
Adult, Dentate or Partially Edentulous
New patient being evaluated for oral diseases: Individualized radiographic exam consisting of posterior bitewings with panoramic exam or posterior bitewings and selected periapical images. A full mouth intraoral radiographic survey is preferred when the patient has clinical evidence of generalized oral disease or a history of extensive dental treatment.
Recall patient with clinical caries or increased risk for caries: Posterior bitewings at 6 to 18 month intervals.
Recall patient no clinical caries or no increased risk for caries: Posterior bitewings at 24 to 36 month intervals.
Recall patient with periodontal disease: Clinical judgment as to the need for and type of radiographic images for the evaluation of periodontal disease. Images may consist of, but is not limited to, selected bitewings and/or periapical images of areas where periodontal disease (other than nonspecific gingivitis) can be demonstrated clinically.
Patient (New or Recall) for monitoring of dentofacial growth and development, and/or assessment of dental/skeletal relationships: Usually not indicated for monitoring of growth and development. Clinical judgment as to the need for and type of radiographic images for evaluation of dental and skeletal relationships.
Patient with other circumstances, including but not limited to, proposed or existing implants, other dental and craniofacial pathoses, restorative/endodontic needs, treated periodontal disease and caries remineralization: Clinical judgment as to the need for and type of radiographic images for evaluation and/or monitoring of these conditions.
Adult, Edentulous
New patient being evaluated for oral diseases: Individualized radiographic exam based on clinical signs and symptoms.
Recall patient with clinical caries or at increased risk for caries: Not applicable.
Recall patient with no clinical caries or not at increased risk for caries: Not applicable.
Recall patient with periodontal disease: Not applicable.
Patient (New or Recall) for monitoring of dentofacial growth and development, and/or assessment of dental/skeletal relationships: Usually not indicated for monitoring of growth and development. Clinical judgment as to the need for and type of radiographic image for evaluation of dental and skeletal relationships.
Patient with other circumstances including, but not limited to, proposed or existing implants, other dental and craniofacial pathoses, restorative/endodontic needs, treated periodontal disease and caries remineralization: Clinical judgment as to the need for and type of radiographic images for evaluation and/or monitoring of these conditions.
In addition, the following recommendations and considerations should be kept in mind when evaluating the need for dental radiographic images for all categories of patients. All radiographic images taken or obtained should be carefully examined for any evidence of caries, bone loss from periodontal disease, developmental anomalies and occult disease. Radiographic screening for the purpose of detecting disease before a clinical examination should not be undertaken. Intraoral radiography is useful for the evaluation of dentoaveolar trauma but if the area of interest extends beyond the dentoalveolar complex, extraoral imaging may be indicated. When a radiographic examination is indicated, care should be taken to follow the ALADA Principle to limit the patient’s exposure.14
Radiation Safety and Protection - Limiting Radiation Exposure
ALADA
The guiding concept in radiation protection is the ALADA principle: As Low As Diagnostically Acceptable. Dental professionals have an ethical and professional obligation to the patient to minimize exposure and maximize the diagnostic result. The dentist is responsible for prescribing radiographs based on the application of selection criteria. The radiographer who carries out the radiographic procedures must implement the recommended safety precautions and utilize optimal imaging, exposure and processing techniques to ensure that quality results are produced without re-exposure to the patient.
A number of past as well as recent studies have documented deficiencies in radiographic procedures that increase exposure to patients. The most common factors that contribute to increased exposure were improper processing, kilovoltage miscalibration, receptor selection, improper exposure settings and round rather than rectangular collimation.18-20 The American Academy of Oral and Maxillofacial Radiology (AAOMR) has published several articles to assist dental practitioners in the assessment of and compliance with recommended practices and safety measures.21,22 The ADA Council on Scientific Affairs has published several articles, most recently in collaboration with the Food and Drug Administration to update patient selection guidelines and address methods to limit radiation exposure.14,23 In addition, the Image Gently® Alliance for the Radiation Safety in Pediatric Imaging, a coalition of health care organizations including dentistry, are collaborating to improve the safety and effectiveness of pediatric imaging. (http://www.imagegently.org/Roles-What-can-I-do/Parent/Dentist).24 These resources can provide the impetus for improving image quality and reducing patient exposure.
Minimizing patient exposure
There are a variety of safety and protection measures that can be implemented to reduce exposure to the dental patient and adhere to the ALADA principle.
Image Receptor Selection
Intraoral receptors
Presently, there is no standardized system in place to classify the sensitivity of digital receptors to radiation like there is for film. As such, dose reduction comparisons and performance claims can be difficult to accurately assess. Generally speaking, digital receptors offer equal or greater radiation dose reduction than E/F speed film.4 The actual amount of exposure reduction achieved with digital receptors is dependent on a number of factors including speed, collimation, technique and retakes. In a systematic review of digital intraoral radiography, Wenzel and Moystad concluded that achievement of patient radiation dose reduction with digital receptors may not hold true in clinical practice.25 The factors that undermined patient dose reduction were the number of images taken, more errors committed and more retakes needed when compared to film. This was particularly true for rigid digital receptors because of their thick, rigid construction, small periphery of dead space reducing the area of image capture, positioning errors and patient discomfort that produced errors causing retakes.25
Radiographic film remains a widely used image receptor. The speed of radiographic film represents the sensitivity of the emulsion to x-rays. The faster the film, the less radiation exposure that is required. Letters of the alphabet denote film speed with D, E, E/F and F speed film commercially available. F speed is the fastest film currently available and its use can significantly decrease the exposure (60% compared to D and 20-25% compared to E) to the patient without diminishing image quality.26 The FDA recommends that film below E speed not be used for dental radiography because it contributes to greater than necessary patient exposure.26
Regardless of the receptor used, standard infection control measures must be used. Digital receptors, both plates and rigid sensors, cannot be sterilized so the clinician must use careful disinfection and barrier coverage techniques to avoid cross-contamination of the receptor.27-30 (Figure 3) A number of studies have indicated that both types of intraoral digital receptors can become contaminated with saliva during use through perforation or improper handling.28-30 Therefore, these devices should be covered with an FDA-cleared disposable barrier during use. The Centers for Disease Control and Prevention (CDC) recommends the use of a double barrier method to minimize the chance of oral fluid contamination.
Extraoral receptors
Extraoral imaging can be accomplished with both digital and film-based systems.
Digital receptors can be used for panoramic and cephalometric imaging, as well. PSP plates are available in panoramic and cephalometric sizes and can be placed inside the respective film cassettes without the intensifying screens. A laser scanner capable of scanning 6” x 12” and/or 8” x 10” plates is required. With direct digital systems, the receptor consists of a vertical CCD/CMOS detector several pixels wide which on-loads and off-loads data as it moves in concert with the x-ray source during the exposure cycle with the image appearing in real time on the monitor.
With regard to film-based extraoral radiography, rare earth intensifying screen phosphors are recommended to reduce radiation exposure. Rare earth elements like lanthanum and gadolinium have replaced calcium tungstate crystals in intensifying screens. Upon x-ray exposure, rare earth phosphors emit a green light that is more intense than the blue light emitted by calcium tungstate phosphors. When rare earth screens are combined with green light sensitive film, exposure can be reduced approximately 55% for panoramic and cephalometric radiographs.4 (Figure 4a, Figure 4b)
No significant dose reduction is achieved by using digital receptors instead of rare-earth intensifying screens combined with matched high-speed film for extraoral radiography.4,31 When imaging children, care should be taken to “child-size” the exposure factors and utilize digital systems that have pediatric programs which restrict the size of the focal trough and thus reduce exposure.32
Receptor Instruments
A variety of intraoral receptor instruments are available to assist the clinician in the acquisition of periapical and bitewing radiographic images. Most are designed for the paralleling technique with alignment rings to guide the x-ray beam with the goals of improving accuracy and reducing errors. (Figure 5) The use of disposable or heat sterilization tolerant instruments are recommended for optimal infection control. The clinician should be well-versed in in both the paralleling and bisecting angle techniques to accommodate situations in which receptor placement, patient anatomy or patient cooperation are less than ideal.
Collimation
The purpose of collimation is to restrict the size of the x-ray beam and as a result reduce patient exposure. Rectangular collimation restricts the x-ray beam the most and significantly reduces the tissue volume exposed when compared to round collimation.8,14 (Figure 6) Rectangular collimation decreases the effective dose to the patient approximately fivefold.32 Therefore, it is recommended that intraoral x-ray equipment provide rectangular collimation with the x-ray beam not to exceed the dimensions of the receptor by more than 2% of the source-to-receptor distance.8 In addition, it is recommended that source-to-skin lengths for PIDs should be open-ended and long (16” or 40 cm) rather than short (8” or 20 cm).8 Other devices that provide rectangular collimation include rectangular collimator that attached to round PIDs, metal rings that clip into the instrument beam guide or facial shield collimators incorporated into the receptor-holding instrument. Several commercial products are available to collimate the primary beam or that can be attached to current radiographic equipment to achieve rectangular collimation. A primary concern with the use of rectangular collimators is an increase in technical errors, specifically cone cuts, because the clinician must be much more precise in receptor placement, angulation and beam alignment. Although round collimators produce fewer placement and cone cut errors than rectangular collimators, several studies have demonstrated that the use of rectangular collimation usually produced minor errors that did not significantly affect the diagnostic yield of intraoral radiographs.34-36 However, clinician training with rectangular collimation and modification of some of the currently available rectangular collimation devices could facilitate error reduction and improve utility.36
Filtration
The x-ray beam generated inside the x-ray tube is not homogeneous but rather contains both high and low-energy photons. The purpose of filtration is to remove low-energy photons that will be absorbed by the patient and, thus, decrease exposure. Aluminum is the most typical metal used for beam filtration. Dental x-ray machines that operate at < 70 kVp are required to have 1.5 mm of aluminum filtration (Figure 7) while machines that operate at ≥ 70 kVp are required to have 2.5 mm of aluminum filtration.
Exposure Factors
Dental x-ray machines that operate below 60 kVp result in higher radiation doses to the patient. Low-energy x-rays are absorbed by the patient’s tissues and do not contribute to image production. The acceptable kilovoltage range is between 60 and 80 with optimal settings between 60 and 70 kV.8 Most intraoral x-ray machines available today have fixed kilovoltage and milliamperage with the exposure time as the only adjustable variable. The length of the exposure time is dependent on several factors including the kilovolltage and milliamperage settings, collimation of the x-ray beam, patient size, area of interest and the type of receptor. Recommended exposure time settings provided in x-ray machine operator manual are designed for the average adult patient. Therefore, adjustments must be made for patients who fall outside this parameter as well as lower settings when imaging children. By necessity, exposure times increase as the survey progresses from the anterior to the posterior regions of the mouth as structures increase in both size and thickness. Technique charts should be used for all x-ray systems which require adjustment of any exposure factor. Technique charts, posted near the control panel, provide guidance to the clinician regarding the appropriate settings for the patient (adult/child), patient size (small/average/large), area of interest (anterior/posterior) and type of receptor (sensor/plate/film). The goal is to consistently produce a diagnostically acceptable image without re-exposure of the patient.
Quality Assurance
Other quality assurance measures that need to be performed are x-ray machine inspection and patient shield evaluation. X-ray machines must be surveyed by a qualified expert upon installation, re-evaluated at regular intervals (typically 3 to 4 year intervals) as established by individual state regulations or after any change has been made to the unit that might affect safe operation or exposure to patients and/or clincians.8,14 Patient shields including thyroid collars and lap aprons should be evaluated on a monthly basis for any visual or physical signs of damage such as creases or clumping of the material which compromise the integrity of the device. Damaged shields should be replaced.8,14
Patient Shields
Thyroid collar
The thyroid gland, particularly in children, is among the most sensitive organs to radiation-induced tumors.8 Even with proper radiographic techniques, the x-ray beam may pass near or through the thyroid.8 In addition, there is some evidence that radiation exposure of the thyroid during pregnancy is associated with low birth weight38 and that dental exposures may contribute to thyroid cancer.39 A thyroid collar should be used on all patients for intraoral radiography.8,14 Use of the thyroid collar results in significant exposure reduction to the thyroid gland.14,40-42 (Figure 8) The NCRP recommends use of the thyroid collar for children and adults when it does not interfere with the image study such as in panoramic radiography.8 In cephalometric imaging, there is evidence that a cephalometric thyroid protector can be employed in lateral cephalometric imaging to reduce exposure to the thyroid without compromising the image.43
Lap apron
The use of the lead apron in dentistry has been controversial. The AAOMR states that the gonadal dose from dental radiography is so minimal that use of the lead apron (Figure 9) should be considered optional unless required by law.22 The updated selection criteria guidelines recommend that a protective thyroid collar should be used whenever possible but lap lead apron shielding may not be necessary if all recommendations for limiting radiation exposure are put into practice.14 The NCRP recommends that a lead apron need not be provided if all other recommendations in the report are followed rigorously.14 NCRP Report 145 includes a large number of recommendations, which would require compliance in order to dispense with lap apron shielding.8 The recommendations include the use of: rectangular collimation with specifications, thyroid shielding, fast film or digital intraoral receptors, rare earth intensifying screens with matched film for film-based extraoral radiography or digital extraoral radiography, soft tissue filteration and beam collimation in cephalometric imaging, open-ended PIDs with lengths of 20 cm or longer and other directives related to dental radiology personnel and safety practices.8 Additionally, the dentist is expected to utilize selection criteria in the prescription of patient radiographic examinations, to obtain qualified expert guidance in facility design and radiation protection, to establish office quality assurance and radiation protection program including radiation safety training of staff and continuing education in radiology.8 The majority of these recommendations have been discussed in the preceding text. If any measure is not implemented, then a lead apron should be provided. In summary, the ALADA principle infers that all precautions be taken to keep exposure as low as reasonably achievable. To err on the side of patient protection and dose reduction is prudent and reflects best practice.
Lead shields should have at least .25 mm of lead or lead equivalent. Some states have requirements regarding the thickness of lead or lead equivalent for patient shields. Since lead is a very soft metal, care should be taken when handling or storing lead shields. Avoid bending, creasing or folding shields and use apron hangers to allow the shield to hang flat and properly distribute its weight. The NCRP recommends that patient shields be visually inspected for defects on a monthly basis and more frequently if damaged. Further, fluoroscopic examination for hidden defects is recommended on an annual basis.8 As an alternative to lead aprons, light weight lead-equivalent shields are available that utilize materials that effectively absorb scatter radiation but are not as heavy and may be more comfortable for patients and more durable.
Training and Education
Dental auxiliary personnel can perform intraoral and extraoral imaging procedures where permitted by law. The qualifications and regulations for dental auxiliary personnel for dental imaging vary by individual state. Regardless, certified personnel should receive appropriate education and training.
Practitioners should remain current regarding radiation safety measures, new equipment, technology and techniques that could further reduce radiation exposure and improve image quality.8,14 In addition, dentists should be prepared to conduct informed discussions with their patients regarding both the benefits and the risks of radiographic examinations.
Operator protection principles
Sources of Radiation
There are three sources of radiation in the dental office, primary, secondary and leakage. Primary and leakage radiation emanate from the x-ray machine. Primary radiation is generated at the anode target, collimated by the PID and directed toward patient to produce radiographs. Secondary radiation is scatter radiation created by primary beam interaction with matter such as the patient’s face and oral structures. And leakage, a form of secondary radiation, is emitted from the tube head encasement when x-rays are generated inside the x-ray tube. The dental assistant should avoid all three sources to minimize occupational exposure.
Dose Limits
Individuals who use radiation to carry out their professional responsibilities are classified as occupationally exposed persons. Dose limits (DLs) have been established for occupationally exposed persons, occupationally exposed pregnant women, trainees under age 18 and nonoccupationally exposed persons (general public). The dose limit (DL) is the dose of whole body radiation that is not expected to produce any significant somatic or genetic effects in a lifetime. Table 6 outlines the DL for all four categories in both SI and traditional units and well as a formula for calculating the cumulated whole body dose limit for occupationally exposed persons. DL compliance is achieved through adherence to radiation safety practices. The ultimate goal of any clinician should be zero occupational exposure.
Minimizing operator exposure
Standard Rules
Minimizing occupational exposure is not difficult to achieve. The dental assistant should consistently practice standard safety measures and utilize a radiation barrier or the distance and position rule to avoid exposure. In addition, safety measures employed to reduce exposure to dental patients also reduce operator occupational exposure. In simple terms, the dental assistant should avoid the primary beam. The dental assistant SHOULD NOT stand in or near the primary beam or its path, hold the x-ray head or PID in place, hold receptor in the patient’s mouth or hold the patient in position. If assistance is required to stabilize a patient, a parent or guardian should be shielded and restrain the dental patient. If the x-ray head or PID drifts, the unit should be serviced professionally rather than risk occupational exposure.
A study by Kumazawa et al. indicated that the average annual occupational dose for dental personnel was 0.2 mSv.44 Few dental personnel received more than 1 mSv and the vast majority received exposures well below detectable levels.44 The vast majority of dental radiation workers do not require individual monitoring. The NCRP recommends that monitoring of dental personnel be considered for individuals who are expected to receive an annual effective dose in excess of 0.1 rem or 1 mSv and that personal dosimeters be provided for known pregnant occupationally exposed individuals8 The DL for a pregnant radiation worker is limited to 0.5 rem or 5 mSv for the duration of the pregnancy and monthly monitoring can help keep occupational exposure below this limit. Any work restrictions for pregnant radiation workers should be based on recommendations by their physician and compliance with institutional policies or state law.8,24 The preceding NCRP recommendations regarding occupational exposure during pregnancy are advisory only.
Monitoring for Pregnant Dental Healthcare Providers
The U. S. Nuclear Regulatory Commission (NCR) has specific regulations (10 CFR 20.1208 and as adopted in 17 CCR 30253) which govern conditions requiring individual monitoring for the declared pregnant woman likely to receive during the entire pregnancy from radiation sources external to the body, a deep dose equivalent in excess of 0.1 rem (1 mSv).45,46 The dose equivalent to an embryo is the sum of the deep-dose equivalent to the woman who has declared her pregnancy and the dose equivalent to the embryo resulting from radionuclides in the embryo and the radionuclides in the woman who has declared pregnancy.46 These regulations require that such individuals be instructed in the health issues associated with occupational exposure to radiation including estimated doses and associated risks as described in Regulatory Guides 8.29 and 8.13.46,47 The purpose of providing this instruction is to help pregnant women make informed decisions regarding radiation exposure during pregnancy. The information should be given orally, in printed form or in any other effective communication and the worker should be given an opportunity to discuss the information and ask questions of the supervisor.47 In order for the pregnant worker to take advantage of the lower radiation dose limit and dose monitoring in 10 CFR 20.1208, the woman must declare her pregnancy in writing to the licensee.47 The declaration remains in effect until she is no longer pregnant or she withdraws the declaration in writing. The worker can elect to complete a guide form letter declaring pregnancy and agrees to comply with a radiation dose not to exceed 0.5 rem (5 mSv) during the entire pregnancy with the understanding that meeting the lower dose may require a change in job or job responsibilities during pregnancy.47 If the worker chooses not to declare pregnancy, she will be subject to the radiation dose limits that apply to any other occupational worker.47
Barriers
When exposures are made, it is preferable that the operator stands behind a barrier with a window to observe the patient. There are two types of barriers, primary and secondary. Primary barriers are designed to be in the path of the primary beam, commonly operatory walls. Secondary barriers are designed to absorb scatter and leakage radiation, commonly the ceiling and floor. Barriers need not be lead-lined, in fact, dental office operatory walls constructed of drywall are found to be adequate. Barrier specifications for dental operatories are based on factors such as total workload of the x-ray machine; the maximum kVp used, the distance from the x-ray source to people occupying the facility, the radiation status of people in the facility and the occupancy for all areas adjacent to the operatory housing the x-ray unit. When barriers are not available, the distance and position rule should be employed. The operator should stand 2 meters (6 feet 8 inches)8 from the source of x-rays and positioned between 90˚ to 135˚ angle to the primary beam. This rule is based on the Inverse Square Law, which states that the intensity of the x-ray beam diminishes as the distance between the source increases. The further the operator is from the source of radiation, the less intense the x-ray beam and the less potential for occupational exposure.
Hand-held Portable Devices
Hand-held, chargeable battery powered x-ray generating systems are available and are being used instead of conventional wall-mounted intraoral x-ray units and/or in environments outside the dental office such as nursing homes and missions of mercy or public health settings. (Figure 10) Handheld x-ray units legally marketed in the U.S. must comply with FDA radiation safety and medical device requirements.48 All certified handheld dental x-ray units must display a fixed certification label/tag, a warning label in English, and an identification (ID) label/tag on the unit’s housing.48 It is important that the clinician has the technical skills necessary to properly use the device and that they receive instruction on its operation and proper storage conditions. (Table 8)
When used according to manufacturer instructions, these hand-held devices present no greater radiation occupational exposure risk than conventional x-ray machine installations.14,49 The potential radiation sources include secondary radiation from the patient and leakage radiation from the housing and the ring shield.50
To reduce risk and to ensure proper utilization, the clinician should comply with the recommendations outlined in Table 7. Patient head and chair positions may require adjustment in order to maintain the device in the correct position to ensure adequate operator protection. If the handheld x-ray device is not used according to these recommendations, additional radiation safety precautions need to be taken. Depending on state regulations, these precautions may include lap lead apron and lead thyroid collar shielding for the clinician and extremity and whole body monitoring dosimetry devices.
Occupational Radiation Monitoring
There are a number of companies that offer personal monitoring with a film badge dosimeter, TLD monitor (thermoluminescent dosimeter) or an optically stimulated luminescence dosimeter. (Figure 10) The collar level is the recommended location but the waist or chest level is an acceptable location to wear the dosimeter.3 A dosimeter monitors the DL for an occupationally exposed individual and indicates the degree of compliance with recommended safety practices. It does not provide any protection for the clinician. Dosimeters are typically issued on a quarterly or monthly basis. Whole body dosimeters are issued on a quarterly basis when the potential for significant exposure on a monthly basis is low. Extremity dosimeters such as a ring TLD can be used for clinician’s operating radiation producingequipment. Film badge dosimeters are inaccurate when used for a period longer than a month.3 Dosimeters are returned on a regular basis for analysis and reports are generated and returned to the dentist for review and permanent record keeping. (Figure 11) The benefits of monitoring are that the information obtained may lead to the identification of improper practices and unsuspected sources of high exposure.8
Summary
Dental radiographic examinations are not without risk. X-radiation has the potential to damage tissue through either the indirect effect or direct effect of radiation. There are many factors that influence the biologic effects of ionizing radiation such as tissue radiosensitivity, linear energy transfer, dose factors, the volume of tissues exposed to radiation, somatic and genetic effects and stochastic and non-stochastic effects. The biologic effects of radiation are cumulative and every effort must be taken to keep radiation exposures as low as reasonably achievable. A variety of radiation safety and protection measures can be employed to reduce exposure to dental patients and minimize occupational exposure.
Glossary
absorbed dose – measure of the energy absorbed per unit mass of matter by any type of ionizing radiation; expressed in rad or Gray units
absorption - transfer of some or all of x-ray photon energy to material or matter; dependent on the energy of the x-ray beam and composition of the absorber
ALADA — safety principle that states that radiation exposure should be kept to minimum or as low as diagnostically acceptable.
atrophy - diminished size or shrinkage in the size of a cell, tissue or organ caused by cell death; a wasting process or progressive degeneration
background radiation – radiation encountered in daily living arising from natural and artificial sources
charge coupled device (CCD) – solid-state, silicon chip detector that converts light or x-ray photons to electrons
collimation – device used to restrict the size and shape of the x-ray beam
complementary metal oxide sensor (CMOS) – solid-state detector similar to the CCD with built-in control functions, smaller pixel size and lower power requirements
coulomb per kilogram – measures the number of electrical charges or ion pairs in a kilogram of air
cross-linking – side spur creation by radiation exposure and attachment to adjacent macromolecule or portion of the same molecule
cumulative effect – additive biologic effect from repeated exposure to radiation
desquamation – ulceration and shedding or loss of the skin
deterministic effect – biologic response whose severity varies with radiation dose; usually a threshold dose exists
detriment – Total harm to health experienced by an exposed group and its descendants as a result of the group’s exposure to a radiation source
direct effect – damage that occurs when ionizing radiation interacts directly with a radiosensitive molecule
dissociation – to disjoin or separate
dose – amount of energy absorbed by an irradiated object
dose limit (DL) — maximum permissible occupational radiation dose
dose rate – dose in rad/Gy absorbed per unit time
dosimeter – device that detects and measures exposure to ionizing radiation
effective dose – the sum of the weighted equivalent doses for the radiosensitive tissues and organs of the body; used to estimate risk, expressed in sieverts
electromagnetic radiation — transmission of energy by oscillating electrical and magnetic fields traveling through space at the speed of light; includes ionizing energies such as x-rays and gamma rays and nonionizing forms including visible light, microwaves and radio waves
epilation — loss of hair
equivalent dose – mean absorbed dose in a tissue or organ modified by the radiation weighting factor; compares the biologic effect of different types of radiation on tissue; expressed in rem or sievert units
ergs – a unit of work or energy equivalent to 0.624 x 1012 electron volts
excitation – addition of energy to a system through x-ray exposure; result of low energy photon interaction with outer shell electron
erythema – redness of the skin that resembles sunburn due to congestion of the capillaries from high doses of radiation
exposure – measure of radiation quantity; ability of radiation to ionize air by x-rays or gamma rays; expressed in roentgen (R) or Coulomb per kilograms (C/kg)
film badge – photographic film used for personnel monitoring to measure radiation exposure to radiation workers
filtration – removal of the longer wavelength x rays from the primary beam with aluminum or other metal; improves beam quality and reduces patient exposure
free radical – uncharged molecule containing a single unpaired electron in the valence shell
genetic effect – effects produced in reproductive cells that affect future unexposed generations
gray (Gy) — A derived unit of ionizing radiation dose in the International System of Units (SI). It is defined as the absorption of one joule of radiation energy per kilogram of matter. It is used as a measure of absorbed does, specific energy (imparted), and kerma (an acronym for kinetic energy released per unit.; 1 Gy = 1 J/kg = 100 rad
heritable – capable of being inherited as a genetic trait
image receptor – devices such as film, intensifying screens and digital sensors used to capture a latent image by exposure to x-rays and made visible by chemical, laser or computer processing
indirect effect – radiation effect that results from the interaction of radiation with water and the production of free radicals
ionization – removal of orbital electrons from the atom creating positive and negative ions
irradiation — process by which an object, including a human subject, is exposed to radiation
joule — unit of energy; work or force applied times the distance over which it is applied
kerma — energy absorbed per unit mass from the initial kinetic energy released in matter by all electrons freed by x-rays
kilovoltage – potential difference between the anode and cathode in an x-ray tube; controls the quality or penetrating power of the x-ray beam
latent period – time period between irradiation and the manifestation of an effect
Law of Bergonié and Tribondeau – the radiosensitivity of cells is directly proportional to their reproductive activity and inversely proportional to their degree of differentiation
leakage radiation – form of secondary radiation emitted from the tubehead housing
linear energy transfer – measure of the rate that energy is transferred from ionizing radiation to the tissue
linear nonthreshold response – biologic response that is directly proportional to dose; no threshold dose necessary for damage to occur
macromolecule - a very large molecule with a polymeric chain structure (combination of simpler molecules) such as proteins and polysaccharides
mitotic rate – rate or frequency of somatic cell division or mitosis in which a parent cell divides to form two daughter cells identical to the parent cell
mucositis – radiation-induced redness and inflammation of the mucous membranes
non-stochastic effects – biologic effects of radiation that demonstrate a threshold; increased severity of damage with increased dose
nonlinear threshold dose – varied biologic responses produced by varied doses of radiation with a certain level below which no response occurs
occupational exposure – radiation exposure that is received by radiation workers
osteoradionecrosis – radiation-induced damage and death of bone
photon – electromagnetic radiation in the form of x-rays and gamma rays that interact with matter like a particle or small bundle of energy rather than a wave
photostimulable phosphor plate (PSP) – receptor composed of polyester base coated with a crystalline halide emulsion which converts x-ray energy into stored energy
point lesion – change that causes impairment or loss of function at the point of a single chemical bond as a consequence of irradiation of a macromolecule
position-indicating device (PID) – open-ended cylinder or rectangular device that is inherent or attached to the x-ray tubehead; guides and collimates the primary beam of radiation toward the patient
primary barrier – protective barrier adequate to absorb the primary or useful beam
radiation caries – rampant form of dental decay as a result of radiotherapy and exposure of the salivary glands; altered salivary flow, pH and viscosity hamper buffering and cleansing actions
rad — radiation absorbed dose; 1 rad = 100 ergs/g = 0.01 Gy
radiolysis – dissociation or break up of the water molecule as a result of irradiation
radiosensitivity – relative susceptibility of cells, tissues and organs to the effects of ionizing radiation
relative biologic effectiveness – compares the biologic effectiveness of any type of radiation to a test radiation to produce the same effect
rem – roentgen equivalent man; a unit of radiation dose equivalent.
roentgen – x-ray quantity based on the ability of x-rays or gamma rays to ionize air; expressed as R or coulomb per kilogram; named for Wilhelm Conrad Roentgen
scatter radiation – x-rays that have been diverted and scattered back toward the x-ray beam
secondary radiation – scatter and leakage radiation produced as a result of primary beam interaction with matter
secondary barrier – protective barrier adequate to absorb secondary radiation
secondary electron – ejected electron from the outer shell of an atom
side spurs – small, spur-like structures that extend from the main chain macromolecule
sievert (Sv) – the SI unit for the dose equivalent and effective dose; 1Sv = 1J/kg = 100 rems
somatic effects – effects of radiation limited to the exposed individual and not passed one to future generations
stochastic effects – the probability of a biologic response to radiation as a function of dose; no threshold dose
thermoluminescent dosimetry – emission of light by a thermally stimulated crystal after radiation; used for occupational and environmental monitoring
threshold dose – dose at which a biologic response first occurs
viscosity – physical property of a substance that is dependent on the friction of the component molecules; a sticky or gummy flow
whole-body exposure – radiation exposure to the entire body rather than a specific or localized area
xerostomia – dryness of the mouth resulting from reduced or absent salivary flow
x-ray — a form of electromagnetic radiation which can penetrate and ionize matter
About the Author
Gail F. Williamson is a Professor of Dental Diagnostic Sciences in the Department of Oral Pathology, Medicine and Radiology at Indiana University School of Dentistry in Indianapolis, Indiana. She received an A. S. in Dental Hygiene, a B. S. in Allied Health and a M. S. in Education from Indiana University. She serves as Director of Allied Dental Radiology and Course Director for Dental Assisting and Dental Hygiene Radiology Courses. A veteran teacher, Prof. Williamson has received numerous awards for teaching excellence. She is a published author and presents numerous continuing education courses on Oral and Maxillofacial Radiology on the national, regional, state and local levels.
References
1. Langland OE and Langlais RP. Early pioneers of oral and maxillofacial radiology. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1995;80:496-511.
3. Bushong SC: Radiologic science for technologists: Physics, biology, and protection 10 St. Louis 2013, Elsevier-Mosby.
4. White SC, Pharoah MJ: Oral radiology: Principles and Interpretation, ed 7, St. Louis, 2013, Mosby-Elsevier.
5. Turbiana M. Radiation risks in perspective: radiation-induced cancer among cancer risks Radiation Environ Biophys 2000;39:3-16.
6. Brenner DJ, Sachs RK. Estimating radiation-induced risks at very low doses: rationale for using a linear no-threshold approach. Radiat Environ Biophys 2006;44:3-253-6.
7. White SC, Mallya SM. Update on the biological effects of ionizing radiation, relative dose factors and radiation hygiene. Australian Dental Journal 2012;57(Suppl): 2-8.
8. National Council on Radiation Protection and Measurements. Radiation protection in dentistry. NCRP Report No. 145. Bethesda, MD, 2003.
9. National Council on Radiation Protection and Measurements. Ionizing radiation exposure of the population of the United States. NCRP Report No. 160. Bethesda, MD, 2009.
10. Brenner DJ, Hall EJ. Computed tomography – an increasing source of radiation exposure. N Eng J Med 2007;357:2277-2284.
11. Ludlow JB, Davies-Ludlow LE, White SC. Patient Risk Related to Common Dental Radiographic Examinations: Impact of 2007 International Commission on Radiological Protection Recommendations Regarding Dose Calculation. JADA 2008;139;1237-1243.
12. Valentin J. The 2007 Recommendations of the International Commission on Radiological Protection. Oxford, England: Elsevier; 2007.
13. Nuclear Energy Agency Organization for Economic Co-operation and Development. Evolution of ICRP recommendations 1977, 1990 and 2007: Changes in underlying science and protection policy and their impact on European and UK domestic regulation. NEA No. 6920, OECD, 2011.
14. American Dental Association, Council on Scientific Affairs and U.S. Department of Health and Human Services, Public Health Service, Food and Drug Administration. Dental radiographic examinations: Recommendations for patient selection and limiting radiation exposure. Rev. 2012. Available at: http://www.ada.org/~/media/ADA/Member%20Center/FIles/Dental_Radiographic_Examinations_2012.ashx Accessed November 30, 2015.
15. The National Research Council. Health effects of exposure to low levels of ionizing radiation: Report in Brief. Based on the findings of the Committee on the Biological Effects of Ionizing Radiation (BEIR VII Phase 2). . Available at: http://dels.nas.edu/resources/static-assets/materials-based-on-reports/reports-in-brief/beir_vii_final.pdf Accessed April 9, 2016.
16. U.S. Department of Health and Human Services. The selection of patients for x-ray examinations. Washington, DC, Health and Human Services Publ (Food and Drug Administration) 88-8273, 1987.
17. U.S. Department of Health and Human Services, Public Health Service, Food and Drug Administration and American Dental Association, Council on Dental Benefit Programs, Council of Dental Practice, Council on Scientific Affairs. The selection of patients for radiographic examinations. Rev. ed. 2004.
18. Platin E, Janhom A, Tyndall. A qualitative analysis of dental radiography quality assurance practices among North Carolina dentists. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1998;86:115-20.
19. Button TM, Moore WC, Goren AD. Causes of excessive bitewing exposure: Results of a survey regarding radiographic equipment in New York. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1999;87:513-17.
20. Yakoumakis EN, Tierris CE, Stefanou EP et al. Image quality assessment and radiation doses in intraoral radiography. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2001;91:362-68.
21. Goren AD, Lundeen RC, Deahl ST at al. Updated quality assurance self-assessment exercise in intraoral and panoramic radiography. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2000;89:369-74.
22. White SC, Heslop EW, Hollender LG, Mosier KM, Ruprecht A, Shrout MK. Parameters of radiologic care: An official report of the American Academy of Oral and Maxillofacial Radiology. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2001;91:498-511.
23. American Dental Association Council on Scientific Affairs. The use of dental radiographs: update and recommendations. JADA 2006;137:1304-12.
24. Image Gently Campaign Centers on Safety. JADA 2014;145:922.Image Gently Alliance for the Radiation Safety in Pediatric Imaging. Available at: http://www.imagegently.org/Roles-What-can-I-do/Parent/Dentist Accessed May 20, 2016.
25. Wenzel A, Moystad A. Work flow with digital intraoral radiography: A systematic review. Acta Odonttologica, 2010;68:106-114.
26. U. S. Food and Drug Administration Center for Devices and Radiological Health. Dental radiography: Doses and film speed. Available at: http://www.fda.gov/Radiation-EmittingProducts/RadiationSafety/NationwideEvaluationofX-RayTrendsNEXT/ucm116524.htm Accessed April 9, 2016.
27. Infection Control in Practice. Dentistry’s Resource for Infection Control & Safety. OSAP chart & checklist: Health and safety considerations for dental radiology. Annapolis, MD. OSAP 2005;4:1-8.
28. Choi JW: Perforation rate of intraoral barriers for direct digital radiography. Dentomaxillofac Radiol 2015;44(3):20140245.
29. Kalathingal SM, Moore S, Kwon S, Schuster GS, Shrout MK, Plummer K. An evaluation of microbiologic contamination on phosphor plates in a dental school. Oral Surg Oral Med Oral Pathol oral Radiol Endod 2009;107:279-82.
30. Castellanos S: Ensure the safe use of digital radiography. Dimensions of Dental Hygiene 2015:13;26-33.
31. Lee GS, Kim JS, Seo YS, Kim JD. Effective dose from direct and indirect digital panoramic units. Imaging Science in Dentistry 2013;43:77-84.
32. Davis AT, Safi H, Maddison SM. The reduction of dose in paedistric panoramic radiography: the impact of collimator height and programme selection. Dentomaxillofac Radiol 2015;44(2): 20140223.
33. Gibbs SJ. Effective dose equivalent and effective dose: Comparison for common projections in oral and maxillofacial radiology. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2000;90:538-45.
34. Parrott LA, Ng SY. A comparison between bitewing radiographs taken with rectangular and circular collimators in UK military dental practices: A retrospective study. Dentomaxillofac Radiol 2011;40(2):102-9.
35. Parks, ET. Errors generated with the use of rectangular collimation.Oral Surg Oral Med Oral Pathol Oral Radiol Endod 1991;71(4):509-13
36. Zhang W, Abramovitch K, Thames W, Leon IK, Colosi, DC, Goren AD. Comparison of the efficacy and technical accuracy of different rectangular collimators for intraoral radiography. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2009;108:e22-e28.
37. Miles DA, Van Dis ML, Williamson GF, Jensen CW. Radiographic Imaging for the Dental Team. Ed. 4, St. Louis, 2009, Saunders- Elsevier.
38. Hujoel P, Bollen A-M, Noonan CJ, del Aguila MA. Antepartum dental radiography and infant low birth weight. JAMA 2004;291:1187-93.
39. Memon A, Godward S, Williams D, Siddique I, Al-Saleh K. Dental x-rays and the risk of thyroid cancer: A case-control study. Acta Oncologica 2010:49:447-453.
40. Sikorski PA, Taylor KW. The effectiveness of the thyroid shield in dental radiology. Oral Surgery 58:225–236; 1984.
41. Marshall NW, Faulkner K, Clarke P. An investigation into the effect of protective devices on the dose to radiosensitive organs to the head and neck. Br J Radiol 1992;65:799-802.
42. Preece J. NCRP radiation protection in dentistry – a challenge for the dental profession. AADMRT Newsletter, Winter 2011.
43. Hoogeveen RC, Rottke D, van der Stelt PF, Berkout WER. Dose reduction in orthodontic lateral cephalographyy: dosimetric evaluation of novel cephalometric thyroid protector (CTP) and anatomical cranial collimation. Dentomaxillofac Radiol 2015;44:20140260.
44. Kumazawa S, Nelson DR, Richardson AC. Occupational exposure to ionizing radiation in the United States: A comprehensive review for the year 1980 and a summary for the years 1960-1985. Washington. U.S. Environmental Protection Agency; 1984. EPA 520/1-84/005.
45. United States Nuclear Regulatory Commission. NRC Regulations 10 CFR 20.1208 Dose equivalent to an embryo/fetus. December 2015. Available at: http://www.nrc.gov/reading-rm/doc-collections/cfr/part020/part020-1208.html Accessed on May 30, 2016.
46. United States Nuclear Regulatory Commission. Regulatory Guide 8.29 Instruction concerning risks from occupational radiation exposure. February 1996. Available at: http://pbadupws.nrc.gov/docs/ML0037/ML003739401.pdf Accessed on September 23, 2015.
47. United States Nuclear Regulatory Commission. Regulatory Guide 8.13 Instruction Concerning Prenatal Radiation Exposure. Available at: http://www.nrc.gov/reading-rm/doc-collections/reg-guides/occupational-health/rg/8-13/ Accessed on September 23, 2015.
48. FDA U.S. Food and Drug Administration. Illegal sale of unsafe hand-held dental x-ray units: FDA Safety Communication. February 10, 2012. http://www.fda.gov/Radiation-EmittingProducts/RadiationSafety/AlertsandNotices/ucm291214.htm Accessed, April 11, 2016.FDA U.S. Food and Drug Administration.
49. Gray JE, Bailey ED, Ludlow JB. Dental staff doses with handheld dental intraoral x-ray units. Health Physics 2012;102:137-42.
50. Radiation safety considerations for x-ray equipment designed for hand-held use. December 24, 2008. http://www.fda.gov/MedicalDevices/DeviceRegulationandGuidance/GuidanceDocuments/ucm094345.htm Accessed, April 11, 2016.
Suggested Readings
American Dental Association, Council on Scientific Affairs, U.S. Department of Health and Human Services, Public Health Service, Food and Drug Administration. Dental radiographic examinations: Recommendations for patient selection and limiting radiation exposure. Rev. 2012. Available at: http://www.ada.org/~/media/ADA/Member%20Center/FIles/Dental_Radiographic_Examinations_2012.ashx
Image Gently® The Alliance for Radiation Safety – What Can I Do as a Dental Professional? http://www.imagegently.org/Roles-What-can-I-do/Parent/Dentist