You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Though cleaning, shaping, and disinfection of the root canal space is essential for endodontic success,1 complete obturation is equally important. Superficially, the root filling is the radiographic legacy left behind by the treating clinician. Realizing that the root canal space can never be sterilized, it is important that obturation materials entomb any remaining bacteria within root canal systems in order to isolate them from a substrate. Obturation also serves to slow the percolation of saliva and oral bacteria down the root canal system.2
The dental marketplace is replete with obturation materials, devices, and recommendations for techniques. As healthcare professionals, dentists take an oath to, above all else, do no harm. We have the responsibility to ensure that the materials being used are safe, effective, and offer a good prognosis according to high-quality, peer-reviewed literature, when available. Though dentistry is ripe with new technologies that improve quality, efficiency, and the patient experience, adopting new materials requires strong enough evidence that they offer significant improvements over existing gold standards to justify a change. Many choices exist for obturation materials supported by similarly high levels of evidence, and it is truly an individual decision to choose a particular protocol that balances safety, efficacy, and success as well as convenience. Many obturation materials have been utilized for decades, as some materials that have come to market have not demonstrated the necessary improvements for wider adoption within the field. That said, as endodontic protocols in general have exhibited significant change as supported by evidence, endodontic obturation will likely evolve with time.
Obturation Materials
When considering a material for obturation, clinicians should consider factors that would make the material well suited for use within the root canal system. Grossman proposed several qualities of importance in selecting an obturation material (Figure 1).3 In addition to the factors listed, the means of placement ultimately matters equally.
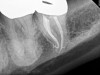
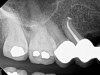
No obturation material nor method for placement available today is truly ideal. Though the dental manufacturing industry periodically introduces new materials with promise, many have been abandoned given major deficiencies in their efficacy or safety. For example, silver points were once well accepted as root filling materials, but were abandoned decades ago due to moisture-related corrosion creating leakage and staining.4 Additionally, silver points proved especially difficult to remove during orthograde retreatment procedures due to the corrosion (Figure 2). Other historic techniques, like the now infamous paraformaldehyde-based Sargenti Method, proved dangerous due to toxicity to the adjacent periapical and surrounding tissues5 (Figure 3). More recently, the technique of complete bioceramic cement obturation has been developed. Though useful with open apices for apexification or in repairing other root deficiencies such as strip perforations, its inability to be removed readily from the root canal system means it should be reserved for those cases lacking suitable alternatives.6
Unlike the previously discussed techniques, gutta percha-based systems meet many of the requirements of an ideal obturation material. Gutta percha itself is a rubber-based material, though in commercially available cones and thermoplasticized preparations it is complexed with zinc oxide, waxes, resins, and metals for opacification.7 Gutta percha is dimensionally stable and biocompatible, sterilizable, radiopaque, easy to manipulate, and well-sealing when placed properly. Though gutta percha is not inherently antibacterial, combining it with endodontic sealers achieves this goal.8 Additionally, if root canal retreatment becomes necessary, gutta percha is easy to remove from the canal space with solvents,9 rotary instrumentation,10 and heat. This does not necessarily include carrier-based gutta percha systems, which often prove difficult to remove. Gutta percha-based obturation is not entirely ideal, however, as it lacks the ability to seal in moist environments and will leak with exposure to saliva.11
Gutta percha cannot be utilized alone and must be placed in conjunction with a sealer. The choice of sealer is perhaps the area of greatest variability among providers practicing endodontics today. Many options exist, and most are considered safe and effective with some variability noted with safety. For example, zinc oxide-eugenol-based sealers were exceptionally popular until about 10 years ago. These sealers are antibacterial8 and inexpensive, and mix and handle easily. Concern exists regarding these sealers, however, as eugenol exhibits significant neurotoxicity.12 Though any material extruded beyond the apex proximal to the mental or inferior alveolar nerves could be damaging,13 the particular neurotoxicity of eugenol raises additional concerns. As a result of this information, many practitioners began to limit the use of this sealer, particularly in the treatment of mandibular molars, due to its risk of paresthesia when in contact with nerve tissue. Case reports relating fungal infections in the maxillary sinus to zinc-containing sealers (due to their metabolic requirements for heavy metals) have also been published,14 raising concerns about their use in the posterior maxilla. Resin-based sealers, known to have antibacterial properties as well, offer particular utility in these instances.8 It is notable that certain variations of epoxy resin-based sealers are known to release formaldehyde, a toxic by-product, though modifications in formulations have eliminated this as a concern.15 As no studies currently exist proving greater effectiveness of zinc oxide-eugenol sealers over epoxy resin sealers, and using several materials on a daily basis is simply inefficient, many practitioners exclusively utilize an epoxy resin endodontic sealer today.
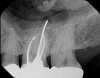
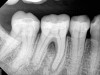
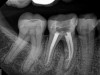
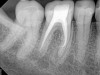
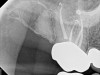
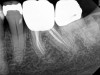
Due to reported sensitivities by patients to certain materials, the use of alternative materials does occasionally become necessary. The endodontic literature demonstrates the safety and efficacy of calcium hydroxide-based sealers, and consequently these represent a suitable alternative (Figure 4 through Figure 6).
New products are constantly introduced to the marketplace, and dentists are heavy targets of corporate marketing. New bioceramic sealers are intriguing given the strong research supporting the use of these materials in other aspects of dental care.6 The evidence so far has shown their safety and efficacy as endodontic sealers.16 However, their superiority to other materials has not been demonstrated. These materials require nuanced differences in technique and are more expensive.17 Wider utilization of these products is likely to occur, though, if the literature evolves in their favor.
Obturation Methods
Though Grossman idealized that an obturation material should be easily placed, some degree of training and skill is necessary in the placement of any dental material. The obturation of a perfectly shaped root canal system is no exception. Obturation methodologies are generally divided into cold lateral and warm vertical compaction. Though lateral compaction techniques still have utility, the warm vertical methodologies are thought to completely fill canal morphology more reliably.18 That said, early iterations of this methodology were inefficient and technique sensitive. Complete backfill techniques using thermoplasticized gutta percha are even more technique-sensitive and are not useful when a prominent apical stop is not present.
A version of warm vertical compaction, termed the continuous wave of condensation, is one of the most widely utilized obturation techniques19 (Figure 7 and Figure 8). This method involves placement of a single, tapered gutta percha cone fit to the same size as the master apical file with sealer, and downpacked with a controlled electric heat source. Based on the rotary instrumentation system used, either 04 or 06 tapered, ISO standardized apical size gutta percha cones corresponding to the final size and shape of the prepared canal can be placed with an appropriate sealer. A plugger heated to 200°F is introduced into the apical third of the root as determined by the working length to condense the apical portion of gutta percha. The heating element is turned off but left in place for an additional 5 seconds to allow slight cooling before a quick burst of heat. The heating element is removed with the coronal portion of excess gutta percha. Following this, the remainder of the canal space is filled using flowable gutta percha delivered via a motorized backfill device to the level of the chamber floor. This method can be hybridized with lateral compaction, in which spreaders are used to add accessory cones, when the shape of the canal does not allow for a tight fit of the ISO sizes of gutta percha points.
The Sealer Puff
The sealer puff achieved by any method is the stuff of lore among endodontic residency programs. Certain schools of thought (and schools) find it to be the marker of successful treatment, in that it proves a complete seal to the apex.18 Others argue that it represents a foreign body that could create long-standing inflammation resulting in impaired healing.20 Certainly, one could argue against a puff of sealer in cases when the root apices are in contact with the mandibular nerve, but in most cases the presence or absence of a sealer puff has not been shown to create an inflammatory reaction (Figure 9 and Figure 10). This will likely remain an area of controversy for as long as there are dentists doing endodontics.
Conclusion
The continuous wave method of condensation, combined with gutta percha and epoxy resin sealers, covers most of Grossman's requirements for the ideal obturation of a root canal space. The deficiencies that remain, namely that gutta percha can still undergo coronal leakage, follow suit for other conventionally available materials. Given the sometimes varied nature of canal systems, it is useful to have several techniques in one's toolbox. Lateral condensation is a useful adjunctive skill to complement warm vertical methodologies when anatomy demands. That being said, it is "warm vertical or bust" for endodontists most of the time.
References
1. Torneck CD. Reaction of rat connective tissue to polyethylene tube implant-Part I. Oral Surg Oral Med Oral Pathol. 1966;21(3):379-387.
2. Delivanis PD, Mattison GD, Mendel RW. The survivability of F43 strain of Streptococcussanguis in root canals filled with gutta-percha and procosol cement. J Endod. 1983;9(10):407-410.
3. Grossman L. Endodontic Practice. Philadelphia, Lea & Febiger, 1981.
4. AAE position statement: Use of silver points. American Association of Endodontists website. https://www.aae.org/specialty/wp-content/uploads/sites/2/2017/06/silverpointsstatement.pdf. Updated 2017. Accessed March 14, 2020.
5. AAE position statement: Concerning paraformaldehyde-containing endodontic filling materials and sealers. American Association of Endodontists. https://www.aae.org/specialty/wp-content/uploads/sites/2/2017/06/paraformaldehydefillingmaterials.pdf. Updated 2017. Accessed March 14, 2020.
6. Parirokh M, Torabinejad M. Mineral trioxide aggregate: A comprehensive literature review-Part III: Clinical applications, drawbacks, and mechanism of action. J Endod.2010;36(3):400-413.
7. Goodman A, Schilder H, Aldrich W. The thermomechanical properties of gutta-percha-II: The history and molecular chemistry of gutta-percha. Oral Surg Oral Med Oral Pathol.1974;37(6):954-961.
8. Saleh IM, Ruyter IE, Haapasalo M, Ørstavik D. Survival of enterococcus faecalis in infected dentinal tubules after root canal filling with different root canal sealers in vitro. Int Endod J.2004;37:193-198.
9. Kaplowitz GJ. Evaluation of gutta-percha solvents. J Endod. 1990;16(11):539-540.
10. Ferreira JJ, Rhodes JS, Ford TR. The efficacy of gutta-percha removal using ProFiles. Int Endod J. 2001;34(4):267-274.
11. Swanson K, Madison S. An evaluation of coronal microleakage in endodontically treated teeth. Part I: Time periods. J Endod. 1987;13(2):56-59.
12. Kozam G. The effect of eugenol on nerve transmission. Oral Surg Oral Med Oral Pathol. 1977;44(5):799-805.
13. Ørstavik D, Brodin P, Aas E. Paraesthesia following endodontic treatment: survey of the literature and report of a case. Int Endod J. 1983;16(4):167-72.
14. Giardino L, Pontieri F, Savoldi E, Tallarigo F. Aspergillus mycetoma of the maxillary sinus secondary to overfilling of a root canal. J Endod. 2006;32(7):692-694.
15. Leonardo MR, Bezerra da Silva LA, Filho MT, Santana da Silva R. Release of formaldehyde by 4 endodontic sealers. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1999;88(2):221-225.
16. Zhang W, Li Z, Peng B. Ex vivo cytotoxicity of a new calcium silicate-based canal filling material. International Endodontic Journal. 2010; 43(9):769-774.
17.Chybowski EA, Glickman GN, Patel Y, et al. Clinical outcome of non-surgical root canal treatment using a single-cone technique with endosequence bioceramic sealer: A retrospective analysis. J Endod.2018;44(6):941-945.
18. Schilder H. Filling root canals in three dimensions. Dent Clin North Am. 1967 Nov:723-744.
19. Buchanan LS. The continuous wave of obturation technique: "Centered" condensation of warm gutta percha in 12 seconds. Dent Today. 1996;15(1):60-67.
20. Seltzer S, Bender IB, Turkenkopf S. Factors affecting successful repair after root canal therapy. JADA. 1963 Nov;67:651-662.