You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
In the United States, dietary supplement use in adults aged 20 and older has increased significantly in the last 2 decades.1 The percentage of the US population taking at least one dietary supplement increased from 42% in 1988 to 1994 to 53% in 2003 to 2006, with women (compared to men) more likely to consume one or more dietary supplements and multivitamins.1-3 Globally, the supplement market is valued at $82 billion, with the US contributing approximately $23 billion. Growth is expected to remain strong through 2017 with a 5% and 6% increase per year both globally and in the United States, respectively.4
The latest data on the type of supplements purchased by supplements users, at the time of this publication, show that the most popular supplements are vitamin D (55.7%), followed by omega-3 (52.7%), coenzyme Q10 (CoQ10) (44.2%), multivitamins (43.4%), probiotics (42.5%), B vitamins (B1 through B12) (40.7%), magnesium (38.0%), curcumin (33.1%), vitamin C (31.5%), calcium (30.1%), and protein/nutrition powders/drinks (24.3%).5 For herbal and botanical supplements, green tea (12%) is the most used, followed by cranberry (8%), garlic (6%), ginseng (6%), echinacea (5%), Ginkgo biloba (5%), turmeric (5%), and milk thistle (4%).6
The growing popularity of dietary supplements has led to increased scrutiny, which has called into question the safety of these products. Bleeding has been one of the complications associated with the use of dietary supplements.7-9
A recent study10 estimated 23,005 emergency-department visits in 1 year were attributed to adverse events related to the use of dietary supplements, with 65.9% involving a single herbal or complementary nutritional product and 31.8% involving a single micronutrient product. A weight-loss product was implicated in 25.5% of emergency-department visits and an energy product in 10.0%. The principal adverse events were palpitations, chest pain, or tachycardia (71.8%) and choking or pill-induced dysphagia or globus (37.6%). The mean age of the patients receiving treatment was 32 years old and 28% of the visits involved persons 20 to 34 years of age.
Other complications included interaction with prescription medications and between different dietary supplements. In dentistry, special attention has to be taken in patients who are taking anticoagulants and cytochrome P450 3A4 substrates (such as clarithromycin, erythromycin, ketoconazole, itraconazole, midazolam, and triazolam) in addition to specific micronutrients such as vitamin D, vitamin E, vitamin K, calcium, fluoride, iron, magnesium, selenium, and zinc, and herbal-nutrient formulations such as cranberries, saw palmetto, garlic, G. biloba, ginseng, ginger, kava kava (removed from the market in Germany and Canada because of its hepatotoxicity), St. John’s wort, valerian, and echinacea. These augment the effects of anticoagulants and inhibit drugs that are absorbed by cytochrome P450 enzymes.11-13 Dietary supplements may interact with each other: possible synergistic effects between omega-3 fish oil and CoQ10 can lead to hypotension, calcium and vitamin D can cause hyperkalemia, and omega-3 fish oil, magnesium, flaxseed, and rosemary can produce an increased risk for bleeding. Others include a possible hypoglycemic effect caused by the use of sage with parsley, bay leaf, cinnamon, and flaxseed; flaxseed with parsley and almond; ginger with cinnamon; and almond with cumin and turmeric.14
For surgeons and their patients, intraoperative and postoperative bleeding are undesirable complications of periodontal and/or oral surgery. Any dietary supplement that contributes to bleeding should be identified and avoided whenever possible.
There is a paucity of literature regarding implant surgery, extraction, orthognathic surgery, or endodontic surgery and dietary supplements. However, a review of periodontal therapy reports some studies are exploring the use of the anti-inflammatory characteristics of certain dietary supplements to gain a better result after therapy and suggests taking supplements of vitamins and minerals, particularly vitamin C, may contribute to improvement of periodontal health.15
The aim of this article is to present a case report of a patient who had been taking dietary supplements for 1 month, did not reveal the usage to the dental team prior to second-stage implant abutment insertion surgery, and developed severe, continuous bleeding after the procedure. In addition, food and herbal supplements are reviewed that may increase the risk for operative and postoperative bleeding.
Case Report
In February 2016, a 36-year-old female presented to the Ashman Department of Periodontology and Implant Dentistry of New York University College of Dentistry in New York. Her chief complaint was the absence of tooth No. 19. She requested a fixed prosthesis. The patient did not smoke, had an unremarkable medical history, reported taking no medications, had no known drug allergies, and rarely consumed alcohol. The intraoral examination showed no caries, a healthy periodontal status, and good oral hygiene. The treatment plan was to place an implant in area No. 19. Periapical x-rays of the area were taken, and a surgical guide was made from an ideal wax-up.
Following administration of local anesthesia (lidocaine hydrochloride 2% with epinephrine 1:100,000), a crestal incision was performed from teeth Nos. 18 through No. 20 and a full-thickness flap was reflected in the No. 19 area. A 5-mm x 10-mm implant was placed with good initial stability (>35 N). Two single interrupted resorbable sutures were used to obtain primary closure. No untoward bleeding was detected at any point during or immediately after the implant surgery. One week later the patient presented for postoperative evaluation and healing was uneventful.
Three months later, the patient was scheduled for second-stage surgery to place a healing abutment. When asked, the patient stated that she had no changes in her medical history. Following local anesthesia infiltration, a crestal incision was made and a full-thickness flap was elevated in the No. 19 area to expose the implant cover screw. A healing abutment measuring 5 mm x 5 mm was placed, and two single interrupted resorbable sutures were used to close the flap.
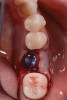
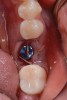
The following day, the patient presented for an emergency, reporting she began to bleed after she left the office and had continuous bleeding that day and night. Vital signs were normal (blood pressure was 115/65 mmHg, and pulse was 53 beats per minute). The first attempt to control the bleeding included having the patient bite on wet collagen wound dressing and alternate using wet- and dry-gauze compression for 30 minutes. After the removal of the collagen wound dressings, the bleeding restarted spontaneously. The sutures were then removed, and a collagen wound dressing was placed under the flap. Two single interrupted sutures were used, and a third suspended suture was placed around the healing abutment to obtain compression in the area. The patient was observed for 1 hour. During this time, wet-gauze compression was used on the area until the bleeding stopped. The patient was then dismissed and told to reapply gauze with pressure if bleeding recurred (Figure 1 through Figure 5).
An updated history revealed that after the time of implant placement, the patient started taking dietary supplements, including a multivitamin, an iron capsule (85 mg), a zinc capsule (15 mg), branched-chained amino-acid powder (1500 mg), a vitamin E soft gel (400 UI), glucosamine (750 mg) and chondroitin tablets (600 mg), and a vitamin C tablet (500 mg) (Table 1). However, prior to the second-stage surgery, when she was asked about any changes in her medical history, medication, or diet, she said, “Nothing new.”
Discussion
The growing popularity of herbal and homeopathic supplements has called into question the safety of such products. As defined by the US Congress in the Dietary Supplement Health and Education Act, which became law in 1994, a dietary supplement (other than tobacco) is intended to supplement the diet, contains one or more dietary ingredients (including vitamins, minerals, herbs or other botanicals, amino acids, and other substances) or their constituents, is intended to be taken by mouth as a pill, capsule, tablet, or liquid, and is labeled on the front panel as being a dietary supplement.3 The absence of direct regulation of the US Food and Drug Administration (FDA) on herbs and supplements as drugs translates to a lack of standardization that over-the-counter medications have. Critics contend that without FDA oversight, consumers have little assurance of predictable potencies, proper identification of plants, contamination control, and accurate labeling from manufacturers.
Some voluntary programs for quality control exist. The USP Verified Mark, started in 2002 by the scientific nonprofit U.S. Pharmacopeial Convention (USP), is given to dietary supplements that have been tested for identity, quality, purity, potency, performance, and consistency of the ingredients listed on the label and are manufactured according to USP internationally recognized standards and FDA current good manufacturing practice. This program is based on manufacturers’ voluntary participation.16
Another organization is ConsumerLab.com, an independent laboratory offering a quality-certification program for manufacturers. Since its inception in 1999, ConsumerLab.com has reviewed more than 3400 products.17 Certification means that a product adheres to standard requirements for consistency between labeling and ingredients and a lack of contaminants. Certification does not guarantee product effectiveness or safety. NSF International, founded in 1944, is an international organization dedicated to developing uniform consensus-based national standards for certifying products and systems in addition to providing consumer education.18
Supplement users have purchased supplements from mass merchandisers (41%), drug stores/pharmacies (34%), supermarkets (23%), and warehouse clubs (12%) or specialty-retailer web-based retailers (12%).6
Typically, patients assume dietary supplements can be taken without proper medical control, as in the case of this patient who did not reveal before surgery that she was using supplements, but only did so when questioned after the occurrence of postsurgical bleeding. Data from the 2014 Council for Responsible Nutrition Consumer Survey on Dietary Supplements18 show more than three-quarters of adults (77%) believe taking supplements is “a smart choice for a healthy lifestyle.” The principal reason for usage is for overall health/wellness benefits (54%), followed by filling in nutritional deficits (29%), bone health (25%), immune health (25%), healthy aging (24%), energy (22%), heart health (22%), joint health (17%), and reduction in the risk for serious illness (17%).
Searches on PubMed and MedBase databases were performed using the following keywords for every dietary supplement: “coagulation,” “bleeding,” and “circulation.” An analysis of dietary supplements, dosage, and effects on bleeding are shown in Table 2. Among those, vitamin E, glucosamine, selenium, lycopene, magnesium, and CoQ10 have been reported to adversely alter hemostasis in humans.
Considering that 58% of the patients are taking supplements with potentially toxic cardiovascular medications, such as warfarin,38 the clinician should be aware of other common dietary supplements that can cause bleeding and can interfere with blood-thinner therapy, in particular garlic, G. biloba, ginseng, ginger, and fish oil. Garlic has been used medicinally for millennia especially for infections and has complex cardiovascular effects, including antiplatelet, antithrombotic, and fibrinolytic activity.38-40 Clinical studies have reported significant reductions in platelet aggregation by inhibition of adrenaline-induced platelet aggregation and mixed effects on fibrinolytic activity.41 Evidence suggests some of the antiplatelet activity may be irreversible, and thus it has been proposed patients cease ingestion of garlic at least 7 days prior to surgery.42
G. biloba is primarily used to improve memory and concentration43 and is one of the most widely used herbal medicines in Europe and the United States. Ginkgolides, one of the active components of G. biloba, are inhibitors of platelet activating factor (PAF). A few case reports have documented bleeding with G. biloba supplementation.44
Ginseng is a traditional herbal medicine that has been used therapeutically for more than 2000 years. Ginsenosides are the bioactive components in ginseng and may act on the immune system by inhibiting PAF.45 Findings from one study reported 2 weeks of ginseng administration (1 gram daily) in combination with warfarin reduced the peak international normalized ratio (INR) values compared with those of the placebo group.46
Ginger is the rhizome of the plant Zingiber officinale and has been used for its antinausea property. Its active component is gingerol and reportedly inhibits platelet aggregation in vitro by acting on prostaglandin and thromboxane synthesis.47 Ginger inhibits arachidonic acid-induced platelet aggregation.48 A study, comparing ginger supplementation (0.8 grams 3 times daily for 1 week) in association with warfarin (25 mg daily for 1 week), found no alterations in INR values, pharmacokinetics, and pharmacodynamics of warfarin.43
Regarding fish oil, commercially available supplements contain, on average, 1000 mg of fish oil.49 The bioactive ingredients in fish oil, eicosapentaenoic acid and docosahexaenoic acid (omega-3 fatty acids), have been showed to inhibit the synthesis of TXA2 and to reduce platelet adhesion and reactivity.50,51 One study reported increased bleeding time with the combination of fish-oil supplementation and aspirin.52
As in many cases similar to the one presented, many dietary-supplement users base their decisions on inadequate information from advertisements or take counsel from nonmedical figures (such as gym personal trainers or friends), despite the paucity or absence of clinical guidance and little or no information about interactions or side effects. For example, the iron tablet that the patient was taking contained a formulation that was more similar to a multivitamin when correctly analyzed and could have led to overdoses of a single supplement.
The dentist should exercise as much precaution as possible to avoid complications. Therefore, dentists should take a thorough medical and medication history at every appointment. A complete history should include the exact dosages and frequencies of ingestion of vitamins, minerals, and herbs that the patient reports.
Dentists should also be familiar with the various supplements’ possible adverse events and interactions with medications. If possible, the dental team should ask the patient to bring the bottles and packages of the supplements to the dental appointments. If the dentist is not familiar with the dietary supplements taken, the dental team can retrieve information from the National Institutes of Health’s Office of Dietary Supplements, which retains a health-professional database of fact sheets.53 For specific interactions and side effects, reputable sources are also available online or via smartphone apps.54-57 If a complication should occur, the dentist should first treat the problem and then send the patient for a medical workup. Although the amount of scientific evidence remains sparse, the American Society of Anesthesiologists recommends patients discontinue herbal and supplemental medications 2 to 3 weeks prior to surgery.11
A stricter regulation and surveillance of the dietary supplements market is therefore advised, and randomized large clinical studies of their effects and side effects are needed in order to avoid unwanted consequences for the patients.58,59 Fortunately, in this case, the patient did not experience other adverse events because she was not taking any prescribed medication that could have interacted with her dietary supplements.
Conclusion
The case presented of a patient with a bleeding complication following second-stage implant abutment insertion surgery illustrates possible etiology and importance of dietary supplements and glucosamine on potential postsurgical bleeding problems. Although this patient took a combination of multivitamins, iron, branched-chained amino-acid powder, glutamine, glucosamine, zinc, vitamin C, and vitamin E, clinicians need to be aware of any dietary supplements and dosages the patient takes prior to surgical procedures to prevent excessive bleeding. Recommendations to stop these supplements prior to surgery similar to those given to patients on any anticoagulant (eg, aspirin, warfarin, rivaroxaban, dabigatran etexilate mesylate) should be discussed with patients undergoing periodontal and implant surgery.
About the Authors
Alessandro Grisa, DDS
Resident
Ashman Department of Periodontology and Implant Dentistry
New York University College of Dentistry
New York, New York
Salvatore Florio, DDS
Resident
Ashman Department of Periodontology and Implant Dentistry
New York University College of Dentistry
New York, New York
Elisabetta Bellia, DDS, PhD
Resident
Ashman Department of Periodontology and Implant Dentistry
New York University College of Dentistry
New York, New York
Sang-Choon Cho, DDS
Clinical Assistant Professor
Director of Advanced Program for International Dentists in Implant Dentistry, and Co-Director of Clinical Research
Ashman Department of Periodontology and Implant Dentistry
New York University College of Dentistry
New York, New York
Stuart J. Froum, DDS
Clinical Professor and Director of Clinical Research
Ashman Department of Periodontology and Implant Dentistry
New York University College of Dentistry
New York, New York
Private Practice
New York, New York
Queries to the authors regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Gahche J, Bailey R, Burt V, et al. Dietary supplement use among U.S. adults has increased since NHANES III (1988–1994). NCHS Data Brief. 2011;(61):1-8.
2. Bailey RL, Gahche JJ, Lentino CV, et al. Dietary supplement use in the United States, 2003-2006. J Nutr. 2011:141(2):261-266.
3. Kennedy ET, Luo H, Houser RF. Dietary supplement use pattern of U.S. adult population in the 2007–2008 National Health and Nutrition Examination Survey (NHANES). Ecol Food Nutr. 2013;52(1):76-84.
4. Teichner W, Lesko M. Cashing in on the booming market for dietary supplements. Consumer and Shopper Insights. McKinsey & Company. https://www.mckinseyonmarketingandsales.com/sites/default/files/pdf/CSI_VMHS_FNL_0.pdf Published December 2013. Accessed September 27, 2016.
5. Vitamin D supplements jump to top spot in popularity, surpassing fish oil, as probiotics rise to #5 spot behind CoQ10 and multivitamins in consumerlab.com survey [news release]. ConsumerLab.com. February 25, 2016. http://www.consumerlab.com/news/ConsumerLab_Survey_Findings/2_24_2016/. Accessed September 27, 2016.
6. Council for Responsible Nutrition (CRN) Web site. The Dietary Supplement Consumer. 2015 CRN Consumer Survey on Dietary Supplements. http://www.crnusa.org/CRNconsumersurvey/2015/index.html#confidence. Accessed September 27, 2016.
7. Randhawa S, Abowd M, Sharma A, Weiss JS. Anterior segment complications of a nutritional supplement. J Cataract Refract Surg. 2007;33(5):918-920.
8. Vale S. Subarachnoid hemorrhage associated with Ginkgo biloba. Lancet. 1998;352(9121):36.
9. Rowin J, Lewis SL. Spontaneous bilateral subdural hematomas associated with chronic Ginkgo biloba ingestion. Neurology. 1996;46(6):1775-1776.
10. Geller AI, Shehab N, Weidle NJ, et al. Emergency department visits for adverse events related to dietary supplements. N Engl J Med. 2015;373(16):1531-1540.
11. Shankland WE Jr. Four common herbs seen in dental practice: properties and potential adverse effects. Cranio. 2009;27(2):118-124.
12. Trivedi R, Salvo MC. Utilization and safety of common over-the-counter dietary/nutritional supplements, herbal agents, and homeopathic compounds for disease prevention. Med Clin North Am. 2016;100(5):1089-1099.
13. Donaldson M, Touger-Decker R. Vitamin and mineral supplements: friend or foe when combined with medications? J Am Dent Assoc. 2014;145(11):1153-1158.
14. Levy I, Attias S, Ben Arye E, et al. Interactions between dietary supplements in hospitalized patients. Intern Emerg Med. 2016;11(7):917-927.
15. Najeeb S, Zafar MS, Khurshid Z, et al. The role of nutrition in periodontal health: an update. Nutrients. 2016;30:8(9).
16. U.S. Pharmacopeial Convention website. USP verified dietary supplements. For manufacturers. http://www.usp.org/verification-services/manufacturers. Accessed September 26, 2016.
17. ConsumerLab.com. About ConsumerLab.com. http://www.consumerlab.com/aboutcl.asp. Accessed September 26, 2016.
18. NSF International website. About NSF. http://www.nsf.org/about-nsf/. Accessed September 26, 2016.
19. Institute of Medicine. Dietary Reference Intakes: Vitamin C, Vitamin E, Selenium, and Carotenoids. Washington, DC: National Academies Press; 2000:186-263.
20. Stampfer MJ, Jakubowski JA, Faigel D, et al. Vitamin E supplementation effect on human platelet function, arachidonic acid metabolism, and plasma prostacyclin levels. Am J Clin Nutr. 1988;47(4):700-706.
21. Marsh SA, Coombes JS. Vitamin E and alpha-lipoic acid supplementation increase bleeding tendency via an intrinsic coagulation pathway. Clin Appl Thromb Hemost. 2006;12(2):169-173.
22. Mabile L, Bruckdorfer KR, Rice-Evans C. Moderate supplementation with natural alpha-tocopherol decreases platelet aggregation and low-density lipoprotein oxidation. Atherosclerosis. 1999;147(1):177-185.
23. Steiner M. Influence of vitamin E on platelet function in humans. J Am Coll Nutr. 1991;10(5):466-473.
24. Zbikowska HM, Olas B. Antioxidants with carcinostatic activity (resveratrol, vitamin E and selenium) in modulation of blood platelet adhesion. J Physiol Pharmacol. 2000;51(3):513-520.
25. Suttnar J, Masova L, Scheiner T, et al. Role of free radicals in blood platelet activation. Cas Lek Cesk. 2002;141(suppl):47-49.
26. Freedman JE, Keaney JF Jr. Vitamin E inhibition of platelet aggregation is independent of antioxidant activity. J Nutr. 2001;131(2):374S-377S.
27. Steiner M. Vitamin E, a modifier of platelet function: rationale and use in cardiovascular and cerebrovascular disease. Nutr Rev. 1999;57(10):306-309.
28. Kakishita E, Suehiro A, Oura Y, Nagai K. Inhibitory effect of vitamin E (alpha-tocopherol) on spontaneous platelet aggregation in whole blood. Thromb Res. 1999;60(6):489-499.
29. Toivanen JL. Effects of selenium, vitamin E and vitamin C on human prostacyclin and thromboxane synthesis in vitro. Prostaglandins Leukot Med. 1987;26(3):265-280.
30. Ricetti MM, Guidi GC, Tecchio C, et al. Effects of sodium selenite on in vitro interactions between platelets and endothelial cells Int J Clin Lab Res. 1999;29(2):80-84.
31. Vitoux D, Chappuis P, Arnaud J, et al. Selenium, glutathione peroxidase, peroxides and platelet functions Ann Biol Clin (Paris). 1996;54(5):181-187.
32. Davila JC, Edds GT, Osuna O, Simpson CF. Modification of the effects of aflatoxin B1 and warfarin in young pigs given selenium. Am J Vet Res. 1983;44(10):1877-1883.
33. O’Kennedy N, Crosbie L, van Lieshout M, et al. Effects of antiplatelet components of tomato extract on platelet function in vitro and ex vivo: a time-course cannulation study in healthy humans. Am J Clin Nutr. 2006;84(3):570-579.
34. O’Kennedy N, Crosbie L, Whelan S, et al. Effects of tomato extract on platelet function: a double-blinded crossover study in healthy humans. Am J Clin Nutr. 2006;84(3):561-569.
35. Hsiao G, Wang Y, Tzu NH, et al. Inhibitory effects of lycopene on in vitro platelet activation and in vivo prevention of thrombus formation. J Lab Clin Med. 2005;146(4):216-226.
36. Gawaz M, Ott I, Reininger AJ, Neumann FJ. Effects of magnesium on platelet aggregation and adhesion. Magnesium modulates surface expression of glycoproteins on platelets in vitro and ex vivo. Thromb Haemost. 1994;72(6):912-918.
37. Serebruany VL, Ordonez JV, Herzog WR, et al. Dietary coenzyme Q10 supplementation alters platelet size and inhibits human vitronectin (CD51/CD61) receptor expression. J Cardiovasc Pharmacol. 1997;29(1):16-22.
38. McEwen BJ. The influence of herbal medicine on platelet function and coagulation: a narrative review. Semin Thromb Hemost. 2015;41(3):300-314.
39. Capasso F, Gaginella TS, Grandolini G, Izzo AA. Phytotherapy. A Quick Reference to Herbal Medicine. Berlin, Germany: Springer-Verlag; 2003.
40. Borrelli F, Capasso R, Izzo AA. Garlic (Allium sativum L.): adverse effects and drug interactions in humans. Mol Nutr Food Res. 2007;51(11):1386-1397.
41. Ackermann RT, Mulrow CD, Ramirez G, et al. Garlic shows promise for improving some cardiovascular risk factors. Arch Intern Med. 2001;161(6):813-824.
42. Ang-Lee MK, Moss J, Yuan CS. Herbal medicines and perioperative care. JAMA. 2001;286(2): 208-216.
43. Jiang X, Williams KM, Liauw WS, et al. Effect of ginkgo and ginger on the pharmacokinetics and pharmacodynamics of warfarin in healthy subjects. Br J Clin Pharmacol. 2005;59(4):425-432.
44. Bent S, Goldberg H, Padula A, Avins AL. Spontaneous bleeding associated with ginkgo biloba: a case report and systematic review of the literature. J Gen Intern Med. 2005;20(7):657-661.
45. Kiefer D, Pantuso T. Panax ginseng. Am Fam Physician. 2003;68(8):1539-1542.
46. Yuan CS, Wei G, Dey L, et al. Brief communication: American ginseng reduces warfarin’s effect in healthy patients: a randomized, controlled trial. Ann Intern Med. 2004;141(1):23-27.
47. Verma SK, Singh J, Khamesra R, Bordia A. Effect of ginger on platelet aggregation in man. Indian J Med Res. 1993;98:240–242.
48. Srivastava KC. Aqueous extracts of onion, garlic and ginger inhibit platelet aggregation and alter arachidonic acid metabolism. Biomed Biochim Acta. 1984;43(8-9):S335-S346.
49. Stanger MJ, Thompson LA, Young AJ, Lieberman HR. Anticoagulant activity of select dietary supplements. Nutr Rev. 2012;70(2):107-117.
50. Connor SL, Connor WE. Are fish oils beneficial in the prevention and treatment of coronary artery disease? Am J Clin Nutr. 1997;66(4 suppl):1020S-1031S.
51. Goodnight SH Jr, Harris WS, Connor WE. The effects of dietary omega 3 fatty acids on platelet composition and function in man: a prospective, controlled study. Blood. 1981;58(5):880-885.
52. Svaneborg N, Kristensen SD, Hansen LM, et al. The acute and short-time effect of supplementation with the combination of n-3 fatty acids and acetylsalicylic acid on platelet function and plasma lipids. Thromb Res. 2002;105(4):311-316.
53. National Institutes of Health. The Office of Dietary Supplements website. https://ods.od.nih.gov. Accessed September 27, 2016.
54. WebMD web site. http://www.webmd.com/interaction-checker/default.htm. Accessed September 27, 2016.
55. Lexicomp website. Clinical Drug Information. Products for Dentists. Wolters Kluwer Health. http://webstore.lexi.com/Store/Products-for-Dentists. Accessed September 27, 2016.
56. Natural Medicines website. https://naturalmedicines.therapeuticresearch.com. Accessed September 27, 2016.
57. Epocrates website. https://www.epocrates.com. Accessed September 27, 2016.
58. Wallace RB, Gryzlak BM, Zimmerman MB, Nisly NL. Application of FDA adverse event report data to the surveillance of dietary botanical supplements. Ann Pharmacother. 2008;42(5):653-660.
59. Petroczi A, Taylor G, Naughton DP. Mission impossible? Regulatory and enforcement issues to ensure safety of dietary supplements. Food Chem Toxicol. 2011;49(2):393-402.