You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Currently, clinicians have essentially three material choices available to them for direct restorations: amalgam, composite resin, and glass-ionomer cements (GICs). Amalgam is obviously no longer considered acceptable for anterior direct restorations in most cases, and its use in the posterior continues to decline annually.1 Knowing that therapeutic treatment of dental injuries or diseases can be accomplished without esthetic compromise, dentists and patients are demanding fewer metallic restorations and more tooth-colored options worldwide. For direct restorations, this leaves only glass ionomers and composite resins.
Modern dental composites are typically composed of mixtures of polymerizable dimethacrylate monomers, which are strengthened by the addition of glass fillers. They were made possible by the work of Bowen, who in 1962 synthesized a reaction product of glycidyl methacrylate and bisphenol A, which came to be known as bis-GMA.2 A key improvement to composite resins was the discovery of the ultraviolet (UV) and visible light photoinitiators, which allowed for the development of light- and dual-cured restorative composites. Highly esthetic and versatile, composite resins have clearly become accepted as an all-purpose anterior and posterior direct tooth-colored restorative material. They have been used extensively for all G.V. Black cavity classifications as well as many indirect applications, offering durability under function, insolubility once set, reasonable biocompatibility, and many variations, shades, and opacities. However, despite more than 50 years of clinical experience and many technical improvements, two substantial challenges remain. First, composite resins have a clinically significant volumetric shrinkage during polymerization, which places stresses on the tooth, the restoration itself, and the adhesive bond between restoration and tooth. The second challenge is creating and maintaining a long-term waterproof seal between composite restoration and tooth structure, which requires special surface preparation and usually a separate adhesive interface. Successful placement of composite resin restorations is certainly possible but calls for impeccable clinical technique and ideal conditions.
Glass-ionomer cement was first reported in 1972 by Wilson and Kent.3 The authors described it as an acid/base reaction between an ion-leachable silicate glass and an aqueous solution of polyacrylic acid and made the first suggestions that it might be useful for dentistry. Since their introduction, GICs have been widely accepted and used in dentistry: 1) as a base or liner under other direct filling materials or indirect restorative materials; 2) for crown buildup/foundation restorations; and 3) as luting cements for indirect restorations. They have also been advocated for use as direct placed restorations, however they have never quite reached the same level of acceptance for this modality of treatment. GICs do have some recognized disadvantages, such as their opacity, brittleness, rough surface finish, high wear rate, and prolonged setting time. However, these economical, biocompatible materials also have many unique advantages that make them highly useful for either a full-contour restoration or sandwich/hybrid restorations where they are synergistic with composite resins. For example, GICs are hydrophilic, have the ability to chemically bond quite strongly to both enamel and dentin, release fluoride, and, contrary to popular belief, have low solubility when properly protected with a glaze/varnish and are fully set.4 In recent years, new clinical techniques and manufacturer improvements have helped overcome limitations and advance GICs in the areas of durability, esthetics, and ease of clinical use. Also, as this article will discuss, GICs may actually perform as well as or better than both amalgam and composite resin when used for some direct clinical applications.
In spite of their own recognized disadvantages—such as polymerization shrinkage, the need for a separate adhesive interface, hydrophobicity, postoperative sensitivity, being technique-sensitive, and having a low fluoride release—composite resins are frequently used for clinical applications where another material or technique might actually be superior. The purpose of this article is to discuss the potential advantages of GIC for some direct applications where composite or another material may not be appropriate or the most ideal choice.
Bulk-Fill Applications
There are numerous reasons for the decline in the popularity of dental amalgam over the past several decades. It is far from an esthetic material, requires cavity preparations that are not minimally invasive, and its use remains controversial due to high mercury content. But amalgam has many virtues that continue to make it difficult to dismiss as a restorative material, especially in poorer countries where access to care, advanced training, and materials is limited. It lasts a long time, is very easy to use, and is relatively insensitive to moisture. But one of amalgam’s chief benefits is that it is dimensionally stable after proper placement and setting. Though it can actually expand or contract slightly during manipulation and setting, the current ADA Specification No. 1 requires that the dimensional change of amalgam be confined to the range of ± 15 µm/cm to 20 µm/cm (measured at 37°C, between 5 minutes and 24 hours after trituration).5 This very small dimensional change allows it to be placed clinically with a true “bulk-fill” technique. It is one property that makes amalgam exceptionally easy to use and has driven both dentists and manufacturers of alternative and esthetic restorative materials to search for what has been frequently described as a “white amalgam.”
Despite major technological improvements, composite resins have yet to prove ideal for bulk-fill applications. The overriding drawback is that all current “light-cured” direct composites have limited depth of cure, which requires the use of incremental layers to ensure complete polymerization in moderate to deeper cavities. Even one newer light-cured material that has been marketed expressly as a single-step bulk-fill composite cannot be used in increments greater than 5 mm,6 a depth limit that is easily reached in many Class I and II cavities. There are several modified bulk-fill products that require a 2-mm, secondary capping layer of hybrid composite on top of each 4-mm “bulk-fill base” increment in their user instructions. Incremental placement has also been advocated to help reduce the negative impact of polymerization shrinkage of composite resins, especially in cavities that possess a high C-Factor. While it is generally agreed that incremental layering does help overcome the effects of polymerization shrinkage versus bulk filling, the literature has not definitively resolved how the materials should be incrementally applied. Some authors have recommended the use of multiple horizontal layers,7 while others believe multiple oblique layers that do not bridge the buccal and lingual cusps8 work better. Still others have proposed unusual clinical techniques like working from the periphery of the cavity to the core9 or some combination of oblique and horizontal increments.10 Presently, there simply is no evidence to recommend one layering technique over another for light-cured composites,11 and none can claim to be superior to simply using self-curing composites.
Self-curing, or auto-curing, composites do not have a limited depth of cure and set slower with more favorable reaction kinetics. They have been shown to work better for bulk filling.12 However, they still possess a significant polymerization shrinkage factor that ultimately transfers shrinkage stress to the cavity walls and can challenge the adhesive bond similar to light-cured composites. Postoperative sensitivity, microleakage, and recurrent decay are often the results. Auto-curing composites also have a lower degree of conversion compared with light-cured composites, which can impact negatively on durability, wear, and other physical properties. Once mixed, they offer limited working time for placement and shaping. Finally, the self-curing chemistry for resin composites requires a much higher level of aromatic tertiary amines, which are known to result in some color instability over time. For these reasons, self-curing composites intended for direct restorative work are no longer prevalent and are only available commercially in limited shades. Though properties of composite resins have improved, and some materials have been produced that are intended for bulk filling, their use is still restricted by material properties and cavity geometry and have only limited and inconclusive clinical testing at this time. As such, composite resins are not considered ideal for bulk-fill applications.
GICs are believed by many dentists to have little or no dimensional change during setting; however, this is not entirely true. During the initial setting reaction, auto-cured GICs do show a limited degree of setting shrinkage of approximately 2% to 3%.13 However, due to many factors such as their slower reaction kinetics, lower elastic modulus, and the type of chemical bond formed with the tooth, this initial dimensional change does not appear to subject the tooth–restoration interface to undue stress. Moreover, subsequent water uptake will compensate for the initial shrinkage, and the restoration will stabilize within a few days of placement to the extent that the initial dimension change is neutralized.13 Resin-modified glass ionomers (RMGIs) do possess a small additional component of a light-cured, hydrophilic resin like hydroxyethyl methacrylate (HEMA), which can also shrink during polymerization. However, in most cases RMGIs contain only small amounts of resin (< 5% of the restoration), which do not form an adhesive bond with the tooth and will not appreciably impact the developing ionic bond with the tooth. Like standard auto-cured GICs, this additional shrinkage is believed to be compensated for by water sorption over a relatively short period of time. This makes both GICs and RMGIs a viable tooth-colored alternative to amalgam and superior to composite resin for bulk-filling applications with regards to dimensional stability.
Another benefit of GICs is their superior resistance to microleakage compared with composite resins. This is why they have been used and recommended in sandwich/hybrid restorations where the deeper layers are GIC and the superficial layer is composite. Although neither adhesive-bonded composites nor GICs form a perfect seal with the tooth interface, GICs are able to release fluoride into any gaps that form, which helps prevent recurrent decay. Furthermore, the matrix of a set GIC is actually a polyacrylate hydrogel, which allows for the free movement of ions like fluoride anion (Fl-) into, out of, and through the restoration, making the final restoration behave like a rechargeable fluoride sponge or reservoir. Some composite resin materials, most notably compomers, are by design initially high in fluoride release. However, once their polymer matrix has fully set, the free movement of fluoride ions within them is severely limited, and fluoride release is found to fall off significantly.
The properties that have challenged GICs as a bulk-fill material in the past are their lowered fracture strength and wear resistance and an opaque appearance. In spite of these negatives, even full-contour/monolithic applications of conventional GICs have performed much better than expected in durability when compared with amalgam and composites. For example, a recent study found no difference in the survival rates of single-surface and multiple-surface GICs and amalgam (primary and permanent teeth) after 6 years and 4 years, respectively.14 A 10-year clinical trial of high-viscosity GICs placed into permanent posterior teeth showed 86.5% of single-surface and 57.6% of multiples were still in service.15 Furthermore, a 2-year study comparing a packable composite versus a high-viscosity GIC showed very similar retention rates for both Class I and Class II restorations.16 Figure 1 shows a Class II, silver cermet restoration (Ketac™ Silver, 3M ESPE, www.3MESPE.com) from the author’s private practice that has been in clinical service for 26 years. Recently, a clinical technique in which a nanofilled light-cured resin glaze is applied to the surface of a high-viscosity GIC (EQUIA™ Fil and EQUIA™ Coat, GC America, www.gcamerica.com) has been found to significantly increase physical properties and durability of the final restoration.17,18 This coating is reported to penetrate 30 µ to 50 µ into the surface of the GIC and will last ≥ 6 months. The final GIC restoration can be recoated with the glaze periodically to extend the clinical benefit. Thus, if bulk-filling applications are desired, GICs may be superior to composite resins in many cases.
The Adhesive/Resin Bond Vs. Glass Ionomer
In the current understanding of adhesive resin bonding it is necessary to remove some of the mineralized components (calcium hydroxyapatite) from enamel and dentin, creating microporosities into which methacrylate resins must infiltrate and polymerize in situ. Enamel is highly uniform throughout its depth and contains very little organic material. In fact, it is the most highly mineralized tissue in the human body, being approximately 96 vol% inorganic mineral and the remainder water and collagen. Acid-etching enamel selectively removes only a small amount of the inorganic material and creates small interprismatic spaces and microporosities within the cores of individual enamel prisms, but they remain largely intact. Resin readily penetrates these spaces and partially surrounds the prisms through capillary action and forms a very stable, strong adhesive connection to enamel. The enamel/resin bond is entirely micromechanical in nature; no chemical bond between resin and enamel has ever been shown or proven to exist. Research and long-term clinical experience has shown this enamel/resin bond to be extremely durable.19 Unfortunately, most cavities and restorations are not entirely confined to enamel, while others may not possess a complete border of enamel.
Dentin is considerably different from enamel, especially when it comes to adhesive resin bonding. In its native state, dentin contains substantially less mineral content and much higher amounts of organic material and water (approximately 70 vol% mineral and 30 vol% collagen and water).20 But at least one source lists the composition to be as low as 50 vol% inorganic mineral, with a 30 vol% of Type I collagen and 20 vol% fluid.21 When dentin is etched, nearly all of the inorganic mineral content from a shallow portion of the surface is removed, exposing the dentin’s matrix collagen structure. These exposed collagen fibers need to be encapsulated by the polymeric resins to be fully protected from the hydrolytic action of oral and dentinal fluids. However, following acid-etching, demineralized dentin is 30% collagen and 70% water, and the adhesive must now diffuse through water to form the hybrid layer.22 This has necessitated the development and use of hydrophilic adhesive comonomers (eg, hydroxyethyl methacrylate [HEMA] and 4-methacryloxyethyl-trimellitic acid [4-META]). However, the key component within the adhesive that contributes most to its strength and durability is the so-called “cross-linker” monomer because of its ability to form cross-linked polymers. Cross-linker monomers have at least two polymerizable groups and are typically dimethacrylates (ie, bis-GMA, UDMA, TEGDMA). They are also hydrophobic and, as such, have been found to infiltrate only a fraction of the wet, demineralized dentin. Thus, a portion of the hybrid layer remains predominantly collagen with contribution from the hydrophilic monomethacrylate comonomer. Because of their low cross-link density, monomethacrylates are inherently unstable in aqueous environments, and this phase will thus eventually degrade when exposed to oral fluids. If the cavity has a continuous border of sound enamel, the resin/enamel bond may effectively block fluid penetration and serve as a protective barrier to the resin/dentin bond. However, without this barrier, the objective of encapsulating the exposed collagen fibers of the demineralized dentin is not always achieved and the zone of demineralized dentin/exposed collagen is not completely infiltrated by adhesive resin.23
In addition to the hydrolytic instability of the monomethacrylate polymers, there is a secondary mechanism that leads to early degradation of the resin/dentin interface. Matrix metalloproteinases (MMPs) are a class of zinc- and calcium-dependent endopeptidases24 that are trapped within the mineralized dentin matrix during tooth development.25 The release and subsequent activation of these endogenous enzymes during acid-etching and dentin bonding procedures26,27 are thought to be responsible for the in-vitro manifestation of thinning and disappearance of collagen fibrils from incompletely infiltrated hybrid layers in aged, bonded dentin.28,29 Thus, the dentin/resin interface does not appear to be nearly as stable as the enamel/resin interface.30 Recent studies have demonstrated that there may be as much as a 32% reduction in the microtensile strengths of the resin/dentin bond in only 6 months.31
One additional challenge of bonding to dentin is that it is not uniform throughout its depth nor even in different parts of the tooth and root. It is well understood that the dentinal tubule is anatomically an inverted cone, with its widest dimension at the pulp and its smallest dimension at the dentinoenamel junction (DEJ). Over the course of its run, its lumen narrows from approximately 3 µm nearest to the pulp to approximately 0.6 µm to 0.8 µm in the superficial dentin. Using this information, Pashley calculated the tubular surface area to be ~ 1% at the DEJ and ~ 22% for dentin nearest the pulp.32 Since this tubular area is occupied by dentinal fluid, which is 95% water, these areas are also approximately equal to the tubular water content for these areas. As indicated above, this difference in intrinsic moisture has been deemed at least partly responsible for the differences observed in bond strengths between superficial and deeper dentin. Many studies clearly show that superficial dentin normally results in higher composite/dentin bond strengths than deeper dentin.33-35
These studies point out the critical importance of the enamel margin to the marginal seal as well as long-term stability and durability of the adhesive resin bond. It seems highly unlikely that durable adhesive bonding can occur when all the margins are located in dentin,36 when the cavity lacks a continuous margin of enamel, or when the clinician needs to bond to dentin that is located in the root of the tooth or very near the pulp.
GICs are in many ways superior to composite resins when it comes to their adhesive interaction with dentin and enamel. In fact, they remain the only material that is truly self-adhesive to tooth tissues without any type of surface pretreatment. Although the bond strength reaches only about 25% of that which can be obtained with resin-based bonding systems, the bond is reliable and far more degeneration-resistant than the resin systems, where the hybridized layer can degrade with time. The adhesive bond between GIC and tooth is understood to be created through a two-fold interaction. The first is a true chemical (ionic) bond between the carboxyl group from the polyalkenoic acid of the GIC and the calcium from the hydroxyapatite mineral found in both enamel and dentin. This chemical bond occurs independent of the depth or locations of enamel and dentin. When a polyalkenoic acid surface “pretreatment” is used, there is a second interaction that can be thought of as a mild self-etching effect leading to shallow hybridization and micromechanical interlocking. Unlike traditional acid-etching with phosphoric acid, this milder pretreatment is effective in enhancing adhesion by cleaning the tooth surface of smear layer without opening dentinal tubules. While this is similar to what is observed with self-etch, resin-based adhesives, the smear layer is rinsed away with GICs while it remains solubilized in the adhesive layer with self-etch adhesive resins. Moreover, in the case of GICs, the polycarboxyl-based polymers are moderately high in molecular weight (8000 to 15,000) versus the relatively low molecular weight of the acidic hydrophilic monomers that are typically used in resins (100 to 1000).
Therefore, whenever a cavity lacks a complete margin of enamel, extends near to the pulp, or is found in root dentin it becomes prudent to consider whether a layered or monolithic GIC direct restoration might actually be superior than direct composite.
Case Report 1
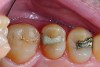
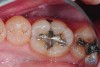
The case depicted in Figure 2 through Figure 5 was completed clinically in late 2009 and subsequently reported in a technique paper published by the author in 2010.37 The case describes the aforementioned EQUIA technique, which essentially employs a high-viscosity, posterior-grade GIC with a bulk-fill technique that is then surface-coated with a penetrating, nanofilled light-cured resin glaze. The reader is referred to the original paper for details about the case, materials, and technique. What is pertinent to this article, however, is the retention and continued durability of the restoration at approximately 42 months. Figure 2 and Figure 3 show the pretreatment view of defective Class I and Class V amalgam restorations, while Figure 4 and Figure 5 illustrate the 42-month recall of the EQUIA Fil restorations. Note that after nearly 4 years of clinical service both restorations were not only still in service but were in excellent condition and would be rated Alfa according to a modified US Public Health Service (USPHS) clinical rating criteria. There is clearly no evidence of excessive wear, staining, marginal microleakage, or recurrent decay. Though this single case is anecdotal, it supports the results found in more comprehensive clinical trials such as those referenced above.
Case Report 2
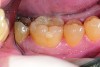
Non-carious cervical lesions (NCCLs), which are sometimes referred to as abfractions, are particularly challenging to restore and, therefore, are a good test of the restorative material or technique. Typically non-retentive, NCCLs have at least 50% of their margins in cervical dentin, and ineffective bonding will usually result in loss of the restoration. The etiology for these lesions remains controversial. Initially, it was believed that they were caused by vigorous or incorrect toothbrushing. However, based on the work of Grippo, most authorities now consider them to be caused by tensile and compressive forces created during tooth flexure, which disrupts the chemical bonds of the crystalline structure of enamel and dentin. This predisposes the area of flexure to cavitation.38
This type of lesion is often restored clinically with low-modulus composite resin. As discussed earlier, the portion of the restoration that is adhesively bonded to enamel typically produces an excellent seal and long-term bond, but the most cervical portions are often located in root dentin or cementum. This portion of the restoration is often found to have significant marginal staining or recurrent decay after only short periods of clinical service. On the other hand, GICs are not as sensitive to the tooth location because they bond to the mineral content of the tooth and are hydrophilic. They also have a low modulus, which seems ideal to resist cervical flexural forces.
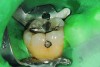
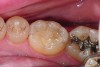
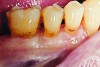
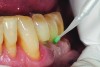
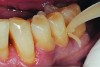
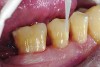
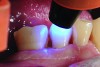
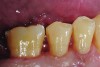
The case demonstrated in Figure 6 through Figure 11 shows the ease with which non-carious Class V lesions can be handled with bulk-filled GIC restorations. Several cervical lesions (abfractions) of significant depth in permanent posterior teeth showed signs of turning carious (Figure 6). The author elected to restore them with the EQUIA technique. For non-carious lesions it is only necessary to freshen the cavity surface and margins with a diamond or carbide bur and to remove any visibly damaged hard tissue. It is then most ideal to remove any smear layer by treating the cavities with a weak acidic solution such as 20% polyacrylic acid (Figure 7). It is important that the clinician not substitute this acidic solution with a conventional phosphoric acid solution or gel used for etching tooth structure prior to resin bonding, as using the conventional solution will remove important mineral content that the GIC requires to bond with chemically. Once the cavities were isolated, a high-viscosity auto-curing GIC was mixed and syringed into the cleaned cavities with a bulk-fill technique (Figure 8). A plastic instrument was then used to condense and shape the material to the cavity walls and margins. The GIC will set in approximately 2:30 seconds to a sufficient level where it is safe to begin adjusting and finishing with rotary instruments. After the restorations were finished, they were coated with a nanofilled, light-cured resin glaze (Figure 9 and Figure 10). The glaze protects the surface of the GIC while it sets completely and enhances the physical strength of the final restoration. The completed GIC restorations (Figure 11) showed excellent esthetics comparable to composite resin.
Conclusions
For placing direct esthetic restorations, clinicians have two primary material choices: composite resins and glass ionomers. Composite resins are available in many different forms and with various dispensing options. Today’s composites and the adhesives used with them are highly advanced yet still require significant attention to detail to achieve clinical success. Clinicians tend to utilize composites routinely for most anterior and posterior applications. However, in some situations the other direct esthetic restorative option—glass-ionomer cements—might be the better option. Like composite resins, today’s glass ionomers are also available in various forms and with assorted dispensing options. Though glass ionomers may not have the same esthetics, wear resistance, or durability of composites, much progress has been made to significantly improve these parameters. GICs currently compete quite well with composite resin and may actually be easier to use and prove superior in some clinical situations. Clinicians should consider GICs for applications in which: a good bond to deep dentin or root dentin is needed; there is a high caries index; true bulk filling is desired; or sufficient enamel is lacking to create an effective seal for the permanent restoration.
DISCLOSURE
The author has no affiliation with any of the products mentioned in this article.
ABOUT THE AUTHOR
Mark L. Pitel, DMD
Associate Clinical Professor of Operative Dentistry, Director of Predoctoral and CDE Esthetic Studies, Columbia University College of Dental Medicine, New York, New York
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
REFERENCES
1. Mitchell RJ, Koike M, Okabe T. Posterior amalgam restorations—usage, regulation, and longevity. Dent Clin North Am. 2007;51(3):573-589.
2. Albers HF. Tooth-Colored Restoratives: Principles and Techniques. 9th Ed. Hamilton, Ontario, Canada: BC Decker Inc.; 2002:81.
3. Wilson AD, Kent BE. A new translucent cement for dentistry. The glass ionomer cement. Br Dent J. 1972;132(4):133-135.
4. Rodrigues Garcia RC, De Góes MF, Del Bel Cury AA. Influence of protecting agents on the solubility of glass ionomers. Am J Dent. 1995;8(6):294-296.
5. ANSI/ADA Standard No. 1—Alloy for Dental Amalgam: 2003. Chicago, IL: American Dental Association; 2003.
6. SonicFill™ [directions for use]. Orange, CA: Kerr Sybron Dental Specialties. http://www.kerrdental.com/cms-filesystem-action?file=/kerrdental-products-dfu/2011_SonicFill_[NA].pdf. Accessed November 22, 2013.
7. Jackson RD, Morgan M. The new posterior resins and a simplified placement technique. J Am Dent Assoc. 2000;131(3):375-383.
8. Chi HH. A posterior composite case utilizing the incremental and stratified layering technique. Oper Dent. 2006;31(4):512-516.
9. Bichacho N. The centripetal build-up for composite resin posterior restorations. Pract Periodontics Aesthet Dent. 1994;6(3):17-23.
10. Ward DH. Treating patients with CARE (comfortable aesthetic restorations): reducing postoperative sensitivity in direct posterior composite restorations. Dent Today. 2004;23(8):60-65.
11. Park J, Chang J, Ferracane J, Lee IB. How should composite be layered to reduce shrinkage stress: incremental or bulk filling? Dent Mater. 2008;24(11):1501-1505.
12. Kuijs RH, Fennis WM, Kreulen CM, et al.Does layering minimize shrinkage stresses in composite restorations? J Dent Res. 2003;82(12):967-971.
13. Mount GJ. An Atlas of Glass-Ionomer Cements: A Clinician’s Guide. 3rd ed. London, England: Informa Healthcare; 2001:38.
14. Mickenautsch S, Yengopal V. Failure rate of atraumatic restorative treatment using high-viscosity glass-ionomer cement compared to that of conventional amalgam restorative treatment in primary and permanent teeth: a systematic review update - III. Journal of Minimum Intervention in Dentistry. 2012;5(6):273-331.
15. Zanata RL, Fagundes TC, Freitas MC, et al. Ten-year survival of ART restorations in permanent posterior teeth. Clin Oral Investig. 2011;15(2):265-271.
16. Ersin NK, Candan U, Aykut A, et al. A clinical evaluation of resin-based composite and glass ionomer cement restorations placed in primary teeth using the ART approach: results at 24 months. J Am Dent Assoc. 2006;137(11):1529-1536.
17. Shinohara M, Antunes A, Padovano J, Bedran-Russo AK. Effect of resin coating on the ultimate strength of glass ionomers [abstract]. J Dent Res. 2010;89(spec iss A). Abstract 796.
18. Turkun LS, Kanik O. Clinical evaluation of new glass ionomer-coating combinated systems for 18-months [abstract]. J Dent Res. 2010;89(spec iss B). Abstract 402.
19. Frankenberger R, Krämer N, Petschelt A. Long-term effect of dentin primers on enamel bond strength and marginal adaptation. Oper Dent. 2000;25(1):11-19.
20. ten Cate AR. Oral Histology: Development, Structure and Function. 5th ed. St. Louis, MO: Mosby; 1998:150.
21. Marshall GW Jr, Marshall SJ, Kinney JH, Balooch M. The dentin substrate: structure and properties related to bonding. J Dent. 1997;25(6):441-458.
22. Nakabayashi N, Pashley DH. Hybridization of Dental Hard Tissues. Chicago, IL: Quintessence Publishing; 1998.
23. Misra A, Spencer P, Marangos O, et al. Parametric study of the effect of phase anisotropy on the micromechanical behaviour of dentin-adhesive interfaces. J R Soc Interface. 2005;2(3):145-157.
24. Visse R, Nagase H. Matrix metalloproteinases and tissue inhibitors of metalloproteinases: structure, function, and biochemistry. Circ Res. 2003;92(8):827-839.
25. Tjäderhane L, Larjava H, Sorsa T, et al. The activation and function of host matrix metalloproteinase in dentin matrix during breakdown in carious lesions. J Dent Res. 1998;77(8):1622-1629.
26. Pashley DH, Tay FR, Yiu C, et al. Collagen degradation by host-derived enzymes during aging. J Dent Res. 2004;83(3):216-221.
27. Nishitani Y, Yoshiyama M, Wadgaonkar B, et al. Activation of gelatinolytic/collagenolytic activity in dentin by self-etching adhesives. Eur J Oral Sci. 2006;114(2):160-166.
28. Armstrong SR, Vargas MA, Chung I, et al. Resin-dentin interfacial ultrastructure and microtensile dentin bond strength after five-year water storage. Oper Dent. 2004;29(6):705-712.
29. García-Godoy F, Tay FR, Pashley DH, et al. Degradation of resin-bonded human dentin after 3 years of storage. Am J Dent. 2007;20(2):109-113.
30. Okuda M, Pereira PN, Nakajima M, et al. Long-term durability of resin dentin interface: nanoleakage vs. microtensile bond strength. Oper Dent. 2002;27(3):289-296.
31. Lenzi TL, Soares FZ, Rocha Rde O. Degradation of resin-dentin bonds of etch-and-rinse adhesive system to primary and permanent teeth. Braz Oral Res. 2012;26(6):511-515.
32. Pashley DH. Smear layer: physiological considerations. Oper Dent Suppl. 1984;3:13-29.
33. Stanford JW, Sabri Z, Jose S. A comparison of the effectiveness of dentin bonding agents. Int Dent J. 1985;35(2):139-144.
34. Mitchem JC, Gronas DG. Effects of time after extraction and depth of dentin on resin dentin adhesives. J Am Dent Assoc. 1986;113(2):285-287.
35. Suzuki T, Finger WJ. Dentin adhesives: site of dentin vs. bonding of composite resins. Dent Mater. 1988;4(6):379-383.
36. Gamborgi GP, Loguercio AD, Reis A. Influence of enamel border and regional variability on durability of resin-dentin bonds. J Dent. 2007;35(5):371-376.
37. Pitel ML. A rapid and aesthetic alternative to a direct posterior composite. Dent Today. 2010;29(11):148-151.
38. Grippo JO. Abfractions: a new classification of hard tissue lesions of teeth. J Esthet Dent. 1991;3(1):14-19.