You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Damage to a branch of the trigeminal nerve (eg, inferior alveolar, lingual, mental, and infraorbital nerves) is a potential untoward consequence of performing implant dentistry. Injury to a nerve can occur due to direct trauma, inflammation, or infection.1 Harm can happen during the following procedures: anesthesia, flap elevation or advancement, harvesting a bone graft, preparing an osteotomy, and implant placement. Because repair of a damaged nerve is problematic, wound avoidance is critically important. Therefore, knowledge is necessary regarding oral nerve anatomy, histology, familiarity with signs and symptoms of nerve damage, and testing for the presence of a neuropathy. Upon diagnosis of nerve injury, a decision must be made with respect to monitoring and documenting symptoms, pharmacologic therapy, implant removal, patient referral for nerve repair, or combinations of the previous concepts. This article addresses multiple issues associated with avoiding injuries and managing patients who experience damage to the trigeminal nerve due to dental implant-related procedures.
Trigeminal Nerve Anatomy and Histology
The trigeminal nerve is the fifth and largest cranial nerve.2 It has three main branches: ophthalmic (V1), maxillary (V2), and mandibular (V3). The mandibular segment is the largest branch and innervates the mandibular lip, chin, teeth, adjacent soft tissues, mandible, and part of the external ear. The motor fibers of the mandibular branch are not subject to injury during implant procedures, because they separate from V3 before they exit the foramen ovale.3
The basic unit of a nerve is a fiber.4 Myelinated nerve fibers predominate in V3. A single nerve axon and a Schwann cell are encased in a connective tissue covering called endoneurium. Groups of encased nerve fibers are referred to as a fascicle and they are surrounded by perineurium. A group of fascicles is surrounded by epineurium. Damage to any part of the nerve bundle can result in neurosensory impairment.
The trigeminal nerve has 7,000 to 12,000 axons and the number of fascicles varies in different regions of the mouth.5 The inferior alveolar nerve (IAN) is polyfascicular (>10 fascicles), whereas the lingual nerve (LN) has few fascicles.6 Because the IAN has more fascicles than the LN, it has greater capacity to repair after injury due to innervation from uninjured fascicles.3
Types of Nerve Damage
Injuries to the trigeminal nerve can include compression, stretching, and partial or complete transection. Damage can result in neurosensory alterations with respect to touch, pressure, temperature, and pain.7 Trigeminal nerve malfunctions can interfere with speaking, eating, kissing, shaving, applying makeup, toothbrushing, and drinking.7 In addition, these injuries can have psychological effects and affect social interactions.8 Altered sensations (eg, pain) may be detected during surgical manipulations or there may be a delayed onset of discomfort.
Terms used to describe injuries with respect to axonal damage are listed:9
Neurapraxia: There is no loss of continuity of the nerve, but it has been stretched or undergone blunt trauma. The altered sensations will subside and feeling returns in days to weeks.
Axonotmesis: The nerve is damaged, degeneration and regeneration occur, but the axon is not severed and feeling returns within 2 to 4 months. Eventual recovery of sensation is often less than normal, and it may be accompanied by dysesthesia.
Neurotmesis: The nerve is severed and there is a poor prognosis for resolution of all neurosensory alterations.
The International Association for the Study of Pain standardized nomenclature concerning nerve injuries. In particular, they altered the definition of paresthesia, which used to denote loss of feeling.10 Currently, the following definitions are employed:
Paresthesia: Altered sensation that is not unpleasant (eg, pins and needles).
Dysesthesia: Altered sensation that is unpleasant.
Anesthesia: Loss of feeling or sensation.
Other terms used to describe changed responses include: allodynia—pain to a stimulus that does not normally hurt; causalgia—persistent burning pain; hypoesthesia—decreased sensitivity to stimulation; and hyperesthesia—increased sensitivity to stimulation.10
When nerves are stretched or compressed, the perineurium protects the fascicles. However, elongation >30% can result in structural failure of the nerve.11 With respect to partial or complete transection of a nerve, total transections usually cause an anesthetic response with poor function. In contrast, partial nerve injuries can have a varied response with respect to dysesthesia.11 Others mentioned that persistent postsurgical pain is a poor predictor for spontaneous rejuvenation of a nerve injury.12,13
Subsequent to peripheral nerve injury, Wallerian degeneration starts and continues over weeks to months.14 Distally, past the place of injury, the axons undergo necrosis. This degeneration is progressive and becomes irreversible at zero to 18 months.14 Factors affecting healing include the patient’s general health, age, and type of injury.14 A defining moment for the damaged nerve is reached when a large mass of endoneurial tubules has changed into scar tissue.15
Assessing Patients’ Injuries Concerning the Trigeminal Nerve
The most commonly injured nerve during implant dentistry is the IAN.3,16 Damage to this structure may manifest itself as anesthesia or paresthesia or dysesthesia of the skin adjacent to the mental foramen, the lower lip, buccal mucous membranes, and gingiva as far posteriorly as the second molar.17 In contrast, patients with an LN injury may report drooling, tongue biting, burning, loss of taste, changes in speech, swallowing, alterations of taste perception, and numbness of the lingual mucosa or tongue.15
During surgery or post-implantation, all signs or symptoms (eg, pain, altered sensation, numbness) of a nerve injury should be documented. Areas of neurosensory deficit should be mapped (eg, altered sensation areas ought to be measured in millimeters). This will facilitate monitoring recovery and help determine if micro-reconstructive surgery may be needed.17 Both subjective and objective sensory tests can be employed to document and evaluate injuries. There are two basic categories of tests: mechanoceptive (response to mechanical pressure or distortion) and nociceptive (perception of pain) (Table 1). Mechanoceptive tests include static light touch, two-point discrimination, and brush-stroke direction.18,19 Pin-tactile discrimination and thermal testing are nociceptive tests. The side opposite to the injury should be stimulated to help confirm there has been an alteration of sensation at the supposedly damaged location. If loss of taste was reported, it can be assessed with salt or sugar on a cotton swab.
Incidence of Nerve Injuries
Subsequent to implant procedures, the occurrence of permanent nerve damage that results in altered lip sensations ranges from 0% to 36% in different studies.20-22 However, citations of old literature can be misleading and not relevant to contemporary implant dentistry. Historically, many damaged nerves may have been due to vestibular incisions that were employed to facilitate implant placement. Currently, midcrestal incisions are usually used and computerized tomography helps avoid injuries. In this regard, recent articles have noted injury rates lower than previously reported. Dannan et al reported a transient trigeminal nerve damage rate after dental implants of 2.95% (5/169 patients) and 1.7% of the patients had permanent neuropathy.23 Another recent university study in an outpatient setting indicated an injury rate of 2.69% (42/1,559) after oral surgical procedures and the permanent injury rate was less (not reported for all types of surgery).24 In the authors’ opinion, even these reduced incidences of nerve damage are too high. It should also be noted that transient altered lip sensations can be due to edema for 1 to 2 weeks and are not considered nerve damage.
Lingual Nerve Damage During Surgical Procedures
The LN in the mandibular molar areas resides within the lingual soft tissue. It may be coronal to the bone within the tissue and lies close to the lingual cortical plate.25 Therefore, caution must be exercised when performing surgical procedures in this region. After third-molar removal, damage to the LN occurs 0.5% to 2.1% of the time.26 However, it is unusual for the LN to be damaged during periodontal or implant surgery.27 As a general rule, implants placed in the mandibular molar region should be performed as follows: intrasulcular incisions, no vertical releasing incisions on the lingual aspect, and a full-thickness mucoperiosteal flap on the lingual aspect; avoid overstretching the flap and maintain safety distance to the nerve when creating the osteotomy. It has been documented that around 90% of LN injuries are transitory and resolve 8 to 10 weeks postoperatively.28
Preoperative Planning: Preventing Nerve Injuries
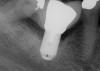
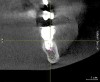
To ensure correct implant placement with the least amount of complications, preoperative planning is necessary. Avoiding nerve injuries starts with proper patient selection, which is done in association with good diagnostics. If it is believed that a computerized tomography (CT) scan or surgical guide would be beneficial, then it should be utilized. Individuals performing surgeries must confirm the path of the mandibular nerve that may have been outlined on a scan by a radiologist. When placing dental implants, a 2-mm safety zone should be left apical to implants over the IAN to accommodate minor drilling errors and drill lengths should be adjusted to take into account radiographic distortion.19,29 In addition, the 2-mm safety zone may help avoid pressure placed on the nerve due to bone compression when the implant is placed close to the mandibular or mental canals (Figure 1 and Figure 2). If necessary, short implants can be used to remain in the safety zone.30 Clinicians should also be aware that drill markings to denote bur length do not take into consideration the extra length of the tapered drill tip, which can add anywhere from 0.4 mm to 1.5 mm to the actual drill size.19,31 In addition, over the IAN or mental nerve, it is advantageous to use drill stops to avoid over-drilling.19,31 It should be underscored that the thickness or density of the bone surrounding the IAN does not provide substantial resistance to drill penetration and excess force should be avoided when drilling over the IAN.32
Finally, it should be noted that 50% of lawsuits related to nerve injury after implant therapy are associated with a lack of informed consent obtained prior to surgery; thus this document must be signed by the patient.33 It also is a good idea to do a neurosensory assessment before initiating procedures to rule out pre-existing sensation impairments.16
Local Anesthesia: Potential to Cause Nerve Damage
Injury to the IAN or LN can occur during block injections due to needle trauma, hematoma formation, or injected chemicals.17 However, it is unknown how the needle or injection ingredients cause nerve damage. In one retrospective study, it was estimated that the incidence of nerve injury was 1/26,762 to 1/160,571,6 whereas Haas and Lennon34 projected it happened 1/785,000. Others, after assessing the literature, indicated the incidence of short-term impaired LN and IAN damage after injections was 0.15% to 0.54%,26 whereas permanent damage due to injection of local analgesics is very unusual, at 0.0001% to 0.01%.35
When performing a mandibular block injection, patients may feel an electric shock around 3% to 7% of the time.36,37 Needle trauma usually resolves spontaneously.37 However, when a clinician sees a patient react (wince in pain) to an injection, the needle should be withdrawn a little and repositioned. Furthermore, with respect to nerve damage due to local anesthesia, there is no known treatment or method of prevention,6 besides avoiding block injections.
It has been noted that 70% to 89% of the injuries that occur as a result of block injections are to the LN.38,39 A possible explanation for this is that the LN has few fascicles, whereas the IAN is polyfascicular and has a greater potential for healing. From a geometric perspective, which may influence the potential for damage or repair, the tip of a 25-gauge needle is 0.45 mm, and the LN and IAN are 1.86 mm and 3 mm wide, respectively.40,41
After block injections, the greatest incidence of neuropathy occurred among individuals who were injected with 4% articaine or prilocaine.39,42 Studies assessing 4% prilocaine and 4% articaine noted an increased number of neuropathies occurred when compared to lidocaine, 7.3 and 3.6 times more, respectively.36,43 Garisto et al reported 4 of 9 investigations that demonstrated 4% prilocaine or articaine was associated with a higher incidence of paresthesia than anesthetics with lower concentrations.39 They and others believe that regional blocks with these drugs should be avoided to reduce the risk of creating a neuropathy.39,42 However, Malamed indicated there was no supporting data besides anecdotal reports that articaine caused an increase in neurosensory alteration when compared to lidocaine.44 Similarly, in 2013, after an extensive literature review, Toma et al concluded that studies suggesting articaine caused increased neurotoxicity were retrospective, biased in data recruitment, and provided a low level of evidence.45 They concluded that procedural trauma emerged as a valid explanation for reported neurological issues. There is controversy in the literature regarding this issue; therefore, clinicians need to make decisions with respect to using higher concentrations of anesthetics based on the data in the literature, their interpretation of this information, and recommendations by drug manufacturers.
Osteotomy Preparation for Dental Implants
Osteotomies should be prepared using sharp drills with copious irrigation. Conceptually, it is possible that too much generated heat could result in postoperative nerve damage.46 The size of the necrotic areas induced by heat is directly proportional to the heat generated during the surgery.47 Eriksson and Albrektsson suggested 47º for 1 minute could produce bone resorption.48 However, the few seconds used to drill an osteotomy probably will not cause nerve damage.
When there is advanced resorption of the mandibular alveolar ridge the position of the mental foramen may be at the alveolar crest; therefore, a midcrestal incision in the edentate area is contraindicated. The incision should be made on the lingual aspect of the ridge to avoid injuring the emerging mental nerve.19,39
If an implant is to be placed anterior to the mental foramen and its length is greater than the distance from the alveolar ridge to the infundibulum of the mental foramen, it is important to verify with a CT scan that there is no anterior loop to the mental nerve.39
Flap Advancement Procedures
Flap advancement will not usually predispose a patient to nerve damage, but caution must be exercised especially in the mental foraminal area.49 In this regard, the clinician should know precisely where the mental nerve emerges from the mental canal to avoid damaging the nerve when a flap is advanced in this region.
Extracting Teeth
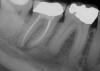
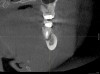
Before extracting a mandibular molar or premolar in preparation for an implant, assess the location of their roots in relation to the IAN and mental nerve, because if a nerve is juxtaposed to the roots of teeth, tooth removal has the potential to induce nerve damage. Furthermore, caution should be exercised when debriding large periapical radiolucencies, because these lesions may communicate with a nerve canal (Figure 3 and Figure 4).
Pharmacologic Therapies for Neuropathies Associated with Dental Implant Placement
There is no consensus with respect to use of pharmaceuticals after a nerve injury. However, some authors suggest the use of corticosteroids and nonsteroidal anti-inflammatory drugs (NSAIDs).3,12,16,19,50-53 Pharmaceuticals may be appropriate when signs or symptoms of neurosensory alterations occur and the clinician is certain that the nerve was not transected.
Table 2 lists several situations where drug application may be beneficial. In conjunction with pharmaceutical intervention, Renton and Yilmaz recommend that patients with chronic neuropathy engage in psychological counseling with respect to pain management.12 The goals of these therapies are to decrease discomfort and to help patients manage their pain. Contrastingly, Bagheri and Meyer stated that it is unlikely that corticosteroids would be of benefit after injury of the IAN, because it is contained within the inferior alveolar canal and drug penetration would be minimal.54 Pertinently, no clinical trials were found that investigated the use of corticosteroids or NSAIDs after nerve injury caused by dental implants.
When to Refer a Patient to a Microsurgeon
At present, there are no uniform guidelines concerning when a patient with an injured trigeminal nerve should be referred to a microsurgeon. If altered sensations occur post-implantation, some authors suggest immediate referral.33 Others recommend seeking consultation after different monitoring intervals: 2 months,19,55 3 months,3,14 before 4 months,18 and 3 to 6 months.56 Ziccardi and Zuniga stated that microsurgery should be done before 1 year, because after that the surgery’s efficacy is diminished.57
Subsequent to nerve injury, the clinician needs to determine if immediate referral is necessary or a pharmacologic approach is warranted or if implant removal or reverse-torquing it a little would best serve a patient. There are diverse opinions in the literature pertaining to when and under what conditions referral to a microsurgeon is needed.58,59 It is generally agreed that if a clinician believes that a nerve has been transected as a result of an implant procedure, immediate referral is warranted.12,51,54,60,61 On the other hand, if a patient manifests neurologic symptoms post-implantation, but the clinician is sure that the drill never entered the mandibular canal, it is possible that postoperative altered sensation is caused by traction of a nerve or an inflammatory process. Then a pharmacologic approach may be warranted.3 A clinician can be almost sure that the drill did not enter the mandibular or mental canal if after each drill, the floor of the osteotomy was checked with an implant probe or if a radiograph clearly depicts the osteotomy terminated several millimeters from the nerve canal. The previous remark is qualified, because there is the remote possibility that some unusual branch of the IAN was present and damaged. From another perspective, a CBCT scan can be ordered to attain an enhanced view of the implant’s relationship to vital structures if a 2-dimensional radiograph was initially used to assess the situation (Figure 1 and Figure 2).
With respect to discomfort after implant placement, if it can be confirmed that the implant is not near the nerve canal, there are different recommendations. Bagheri and Meyer suggest waiting 3 to 4 months to see if altered sensations improve before referring a patient to a microsurgeon.14 They also advise that if an implant is close to the nerve, it could be reversed a little to decrease bone compression. Contrastingly, after surgical placement, if a patient has neurosensory discomfort, despite no apparent transection of the nerve, it was recommended to remove the implant within 36 hours and prescribe a steroid.33,51 Pertinently, Khawaja and Renton discussed four cases where patients became symptomatic after implant placement without apparently encroaching upon the IAN.51 The implants were removed, and then two of the four patients’ neural issues quickly resolved.12 With respect to the four cases, implants were removed after 18 and 36 hours vs 3 and 4 days.
With regard to the controversy as to whether surgical intervention is needed prior to 3 months if there is no observed transection of the nerve—there is no definitive answer.58 Renton, Dawood, and colleagues suggest that clinicians should not wait too long, because after 3 months they believe neural changes occur that diminish positive responses to microsurgical repair.59 Contrastingly, Ziccardi and Steinberg in their review article suggest that when there are altered sensations, but unobserved damage to the nerve, the patient should be monitored for 1 month, and as long as symptoms are improving, monitoring should be continued.58 However, if there is no improvement, or pain worsens, consider microsurgery. It was concluded that patients treated 6 to 8 months after injury do as well as patients treated earlier.58 They pondered that earlier intervention may be better, but at present the data does not support this conclusion. It was also mentioned that minor altered sensations are best left untreated, because surgery is not totally predictable to achieve a desired result.
Others mentioned the “12 week rule,” which refers to the timeline in which surgeons often wait before making decisions about microsurgery for the patient who manifests intolerable continual loss of sensory function.54 This monitoring period would be curtailed for a patient who has pain. In summary, the patient’s radiographic and clinical findings, symptoms, and concern about neural scarring dictate when a patient should be referred for microsurgical consultation. Pertinently, there are medico-legal considerations with respect to a timely referral. Therefore, when in doubt pertaining to the etiology of altered neurosensory issues, early transfer to a nerve specialist is prudent.
Surgical Repair of a Damaged Trigeminal Nerve
There are specific reasons for undergoing nerve repair and different factors impact the success of these procedures. Ziccardi and Zuniga listed several indications for microsurgery: altered sensations that persist for more than 3 months and interfere with daily functions, observed nerve transection, no improvement of hypoesthesia, or development of pain caused by nerve entrapment.57 When nerve repair procedures are performed, numerous factors can affect the results: time between injury and repair, the type and extent of injury, the vascularity of the injury site, skill of the surgeon, harvesting and preparation of the graft, the tension (if any) across the repair, and the age and general health of the patient.60
Microsurgical repair of injured branches of the trigeminal nerve (IAN and LN) can be accomplished.62 However, the successful repair rate and amount of sensory restoration are variable (Table 3).60,62-66 Furthermore, it should be noted that most of the cited studies had limited populations and the final assessment methods were different; therefore it is not possible to directly compare the success rates of these studies.
It appears that 50% to 60% of the time there is a perceived neurological improvement by patients after microsurgery. However, Ziccardi and Zuniga caution that patients with moderate to severe nerve damage need to be informed that they will not usually experience complete sensory recovery.57 Pertinently, others lamented that the success of microsurgical procedures has been overstated and that signs of anesthesia, dysesthesia, and spontaneous pain are negative predictors for repair even with surgical interventions.51 Overall, it can be concluded that microsurgery can help some individuals; however, it cannot predictably resolve all issues.63 Therefore, prevention of injuries is the best way to ensure patients a speedy recovery after dental implant procedures.
Conclusion
There are situations when a decision must be made as to whether to retain an implant that is osseointegrated, but its insertion has caused a tolerable paresthesia (no pain). There are two sides to this dilemma. Implant removal may not improve the patient’s altered sensation; therefore, the implant can be restored. On the other hand, a patient must be informed that there is a remote possibility that a traumatic neuroma could form if an implant rests on a damaged nerve. A neuroma results from exaggerated neural healing and hyperplasia and may need to be surgically removed. To take into account both points of view, a treatment plan needs to be made after discussion with a patient and this conversation should be documented in the chart.
Disclosure
The authors had no disclosures to report.
About the Authors
Gary Greenstein, DDS, MS
Clinical Professor
College of Dental Medicine
Columbia University
Private Practice
Surgical Implantology and Periodontics
Freehold, New Jersey
Joseph R. Carpentieri, DDS
Clinical Assistant Professor
College of Dental Medicine
Columbia University
Private Practice
Surgical Implantology and Prosthodontics
White Plains, New York
John Cavallaro, DDS
Clinical Professor
College of Dental Medicine
Columbia University
Private Practice
Surgical Implantology and Prosthodontics
Brooklyn, New York
References
1. Nazarian Y, Eliav E, Nahlieli O. Nerve injury following implant placement: prevention, diagnosis and treatment modalities. Refuat Hapeh Vehashinayim. 2003; 20(3):44-50.
2. Joo W, Yoshioka F, Funaki T, et al. Microsurgical anatomy of the trigeminal nerve. Clin Anat. 2014;27(1):61-88.
3. Misch CE, Resnik R. Mandibular nerve neurosensory impairment after dental implant surgery: management and protocol. Implant Dent. 2010;19(5):378-386.
4. Sharawy M. Applied anatomy for dental implants. In: Misch CE, ed. Contemporary Implant Dentistry. St. Louis, MO: Mosby Elsevier; 2008:353-358.
5. Assael LA. The nerve under the microscope. J Oral Maxillofac Surg. 2002;60(5):483-484.
6. Pogrel MA, Thamby S. Permanent nerve involvement resulting from inferior alveolar nerve blocks. J Am Dent Assoc. 2000;131(7):901-907.
7. Ziccardi VB, Assael LA. Mechanisms of trigeminal nerve injuries. Atlas Oral Maxillofac Surg Clin North Am. 2001;9(2):1-11.
8. Abarca M, van Steenberghe D, Malevez C, et al. Neurosensory disturbances after immediate loading of implants in the anterior mandible: an initial questionnaire approach followed by a psychophysical assessment. Clin Oral Investig. 2006;10(4):269-277.
9. Seddon HJ. Three types of nerve injury. Brain. 1943;66(4):237-240.
10. Merskey H, Bogduk N, eds. Classification of Chronic Pain. 2nd ed. International Association for the Study of Pain Task Force on Taxonomy, IASP Press, IASP Council in Kyoto. 2007;29-30.
11. Hubbard JH. The quality of nerve regeneration. Factors independent of the most skillful repair. Surg Clin North Am. 1972;52(5):1099-1105.
12. Renton T, Yilmaz Z. Managing iatrogenic trigeminal nerve injury: a case series and review of the literature. Int J Oral Maxillofac Surg. 2012;41 (5):629-637.
13. Rodríguez-Lozano FJ, Sanchez-Pérez A, Moya-Villaescusa MJ, et al. Neuropathic orofacial pain after dental implant placement: review of the literature and case report. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2010;109(4):e8-12.
14. Bagheri SC, Meyer RA. When to refer a patient with a nerve injury to a specialist. J Am Dent Assoc. 2014;145(8):859-861.
15. Bagheri SC, Meyer RA, Khan HA, et al. Retrospective review of microsurgical repair of 222 lingual nerve injuries. J Oral Maxillofac Surg. 2010;68(4):715-723.
16. Juodzbalys G, Wang HL, Sabalys G, et al. Inferior alveolar nerve injury associated with implant surgery. Clin Oral Implants Res. 2013;24:183-190.
17. Juodzbalys G, Wang HL, Sabalys G. Injury of the inferior alveolar nerve during implant placement: a literature review. J Oral Maxillofac Res. 2011;2(1):1-20.
18. Hegedus F, Diecidue RJ. Trigeminal nerve injuries after mandibular implant placement—practical knowledge for clinicians. Int J Oral Maxillofac Implants. 2006;21(1):111-116.
19. Kraut RA, Chahal O. Management of patients with trigeminal nerve injuries after mandibular implant placement. J Am Dent Assoc. 2002;133(10):1351-1354.
20. Bartling R, Freeman K, Kraut RA. The incidence of altered sensation of the mental nerve after mandibular implant placement. J Oral Maxillofac Surg. 1999;57(12):1408-1410.
21. Wismeijer D, van Waas MA, Vermeeren JI, Kalk W. Patients’ perception of sensory disturbances of the mental nerve before and after implant surgery: a prospective study of 110 patients. Br J Oral Maxillofac Surg. 1997;35(4):254-259.
22. Ellies L, Hawker P. The prevalence of altered sensation associated with implant surgery. Int J Oral Maxillofac Implants. 1993;8(6):674-679.
23. Dannan A, Alkattan F, Jackowski J. Altered sensations of the inferior alveolar nerve after dental implant surgery: a retrospective study. Dentistry. 2013;S13:1-5.
24. Deppe H, Mücke T, Wagenpfeil S, et al. Trigeminal nerve injuries after mandibular oral surgery in a university outpatient setting—a retrospective analysis of 1,559 cases. Clin Oral Investig. 2015;19(1):149-157.
25. Behnia H, Kheradvar A, Shahrokhi M. An anatomic study of the lingual nerve in the third molar region. J Oral Maxillofac Surg. 2000;58(6):649-651.
26. Hillerup S, Jensen R. Nerve injury caused by mandibular block analgesia. Int J Oral Maxillofac Surg. 2006;35(5):437-443.
27. Chan HL, Leong DJ, Fu JH, et al. The significance of the lingual nerve during periodontal/implant surgery. J Periodontol. 2010;81(3):372-377.
28. Blackburn CW. A method of assessment in cases of lingual nerve injury. Br J Oral Maxillofac Surg. 1990;28(4):238-245.
29. Greenstein G, Tarnow D. The mental foramen and nerve: clinical and anatomical factors related to dental implant placement: a literature review. J Periodontol. 2006;77(12):1933-1943.
30. Lee SA, Lee CT, Fu MM, et al. Systematic review and meta-analysis of randomized controlled trials for the management of limited vertical height in the posterior region: short implants (5 to 8 mm) vs longer implants (> 8 mm) in vertically augmented sites. Int J Oral Maxillofac Implants. 2014;29(5):1085-1097.
31. Greenstein G, Greenstein B, Desai RN. Using drill stops on twist drills to promote safety and efficiency when creating osteotomies for dental implants. J Am Dent Assoc. 2014;145(4):371-375.
32. Başa O, Dilek OC. Assessment of the risk of perforation of the mandibular canal by implant drill using density and thickness parameters. Gerodontology. 2011;28(3):213-220.
33. Renton T. Prevention of iatrogenic inferior alveolar nerve injuries in relation to dental procedures. Dent Update. 2010;37(6):354-356.
34. Haas DA, Lennon D. A 21-year retrospective study of reports of paresthesia following local anesthetic administration. J Can Dent Assoc.1995;61(4):319-330.
35. Hillerup S. Iatrogenic injury to oral branches of the trigeminal nerve: records of 449 cases. Clin Oral Investig. 2007;11(2):133-142.
36. Harn SD, Durham TM. Incidence of lingual nerve trauma and postinjection complications in conventional mandibular block anesthesia. J Am Dent Assoc. 1990;121(4):519-523.
37. Pogrel MA, Bryan J, Regezi J. Nerve damage associated with inferior alveolar nerve blocks. J Am Dent Assoc. 1995;126(8):1150-1155.
38. Miller PA, Haas DA. Incidence of local anesthetic-induced neuropathies in Ontario from 1994-1998. J Dent Res. 2000;79(special issue):627.
39. Garisto GA, Gaffen AS, Lawrence HP, Tenenbaum HC, Haas DA. Occurrence of paresthesia after dental local anesthetic administration in the United States. J Am Dent Assoc. 2010;141(7):836-844.
40. Kiesselbach JE, Chamberlain JG. Clinical and anatomic observations on the relationship of the lingual nerve to the mandibular third molar region. J Oral Maxillofac Surg. 1984;42(9):565-567.
41. Ikeda K, Ho KC, Nowicki BH, et al. Multiplanar MR and anatomic study of the mandibular canal. AJNR Am J Neuroradiol. 1996;17(3):579-584.
42. Gaffen AS, Haas DA. Retrospective review of voluntary reports of nonsurgical paresthesia in dentistry. J Can Dent Assoc. 2009;75(8):579.
43. Krafft TC, Hickel R. Clinical investigation into the incidence of direct damage to the lingual nerve caused by local anaesthesia. J Craniomaxillofac Surg. 1994;22(5):294-296.
44. Malamed SF. Local anesthetics: dentistry’s most important drugs, clinical update 2006. J Calif Dent Assoc. 2006;34(12):971-976.
45. Toma M, Berghahn M, Loth S, et al. Articaine and paresthesia in dental anesthesia: neurotoxicity or procedural trauma? Dentaltown. 2013:64-71.
46. Eriksson AR, Albrektsson T. Temperature threshold levels for heat-induced bone tissue injury: a vital-microscopic study in the rabbit. J Prosthet Dent. 1983;50(1):101-107.
47. Tehemar SH. Factors affecting heat generation during implant site preparation: a review of biologic observations and future considerations. Int J Oral Maxillofac Implants. 1999;14(1):127-136.
48. Eriksson AR, Albrektsson T. Temperature threshold levels for heat-induced bone tissue injury: a vital-microscopic study in the rabbit. J Prosthet Dent. 1983;50(1):101-107.
49. Greenstein G, Greenstein B, Cavallaro J, et al. Flap advancement: practical techniques to attain tension-free primary closure. J Periodontol. 2009;80(1):4-15.
50. Vecht CJ, Haaxma-Reiche H, van Putten WL, et al. Initial bolus of conventional versus high-dose dexamethasone in metastatic spinal cord compression. Neurology. 1989;39(9):1255-1257.
51. Khawaja N, Renton T. Case studies on implant removal influencing the resolution of inferior alveolar nerve injury. Br Dent J. 2009;206(7):365-370.
52. Park JH, Lee SH, Kim ST. Pharmacologic management of trigeminal nerve injury pain after dental implant surgery. Int J Prosthodont. 2010;23(4):342-346.
53. Martin WJ, Forouzanfar T. The efficacy of anticonvulsants on orofacial pain: a systematic review. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2011;111(5):627-633.
54. Bagheri SC, Meyer RA. Management of mandibular nerve injuries from dental implants. Atlas Oral Maxillofac Surg Clin North Am. 2011;19(1):47-61.
55. Alhassani AA, AlGhamdi AS. Inferior alveolar nerve injury in implant dentistry: diagnosis, causes, prevention, and management. J Oral Implantol. 2010;36(5):401-407.
56. Jones RHB. Repair of the trigeminal nerve: a review. Australian Dent J. 2010;55(2):112-129.
57. Ziccardi VB, Zuniga JR. Nerve injuries after third molar removal. Oral Maxillofac Surg Clin North Am. 2007;19(1):105-115.
58. Ziccardi VB, Steinberg MJ. Timing of trigeminal nerve microsurgery: a review of the literature. J Oral Maxillofac Surg. 2007;65(7):1341-1345.
59. Renton T, Dawood A, Shah A, et al. Post-implant neuropathy of the trigeminal nerve. A case series. Br Dent J. 2012;212(11):1-6.
60. Bagheri SC, Meyer RA, Cho SH, et al. Microsurgical repair of the inferior alveolar nerve: success rate and factors that adversely affect outcome. J Oral Maxillofac Surg. 2012;70(8):1978-1990.
61. Meyer RA, Bagheri SC. Microsurgical reconstruction of the trigeminal nerve. Oral Maxillofac Surg Clin North Am. 2013;25(2):287-302.
62. Susarla SM, Lam NP, Donoff RB, et al. A comparison of patient satisfaction and objective assessment of neurosensory function after trigeminal nerve repair. J Oral Maxillofac Surg. 2005;63(8):1138-1144.
63. Pogrel MA. The results of microneurosurgery of the inferior alveolar and lingual nerve. J Oral Maxillofac. 2002;60(5):485-489.
64. Lam NP, Donoff RB, Kaban LB, Dodson TB. Patient satisfaction after trigeminal nerve repair. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2003;95(5):538-543.
65. Strauss ER, Ziccardi VB, Janal MN. Outcome assessment of inferior alveolar nerve microsurgery: a retrospective review. J Oral Maxillofac Surg. 2006;64(12):1767-1770.
66. Gregg JM. Studies of traumatic neuralgia in the maxillofacial region: symptom complexes and response to microsurgery. J Oral Maxillofac Surg. 1990;48(2):135-140.