You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
This article addresses a type of papilla-sparing incision that can be used to place an implant or when soft- or hard-tissue augmentation is needed. These procedures can be performed separately or in combination with each other. The described papilla-sparing incision is not novel; however, different clinical scenarios (eg, submerged and nonsubmerged implant placement, fixed and removable provisionalization) using papillary-sparing incisions are presented and background information is provided to help understand the intricacies of this technique. First, some background information is provided, then details of the papilla-sparing technique are described.
Appearance of a Normal, Healthy Periodontium in the Maxillary Anterior
In a healthy dentition with no bone or clinical attachment loss, the underlying alveolar crest follows the scallop of the cementoenamel junction (CEJ) and is approximately 2 mm apical to the CEJ. The maxillary anterior interdental crests are approximately 3 mm coronal to the facial bone height (ranging from 2.1 mm to 4 mm).1 On average, the free gingival margin is approximately 3 mm coronal to the crest of the bone (biologic width plus sulcus depth).2,3 Interproximally, interdental papillae between the central incisors is 4.5 mm to 5.5 mm coronal to the osseous crest.2,3 The additional papillary height (1.5 mm to 2.5 mm) is caused by hypertrophy of the interdental tissue and includes the col area when a contact point is present.2,3 These papillae are supported by gingival supracrestal fibers from adjacent teeth. Therefore, preservation of these fibers and subjacent bone are important considerations in maintaining gingival form when surgery is needed in the esthetic zone.
Papilla-Sparing Incision Technique
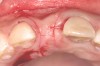
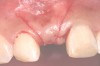
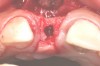
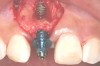
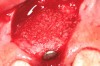
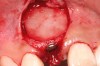
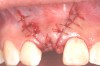
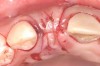
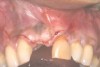
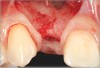
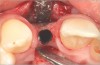
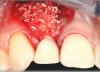
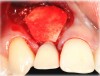
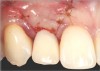
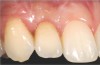
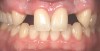
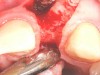
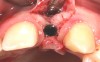
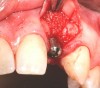
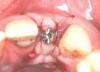
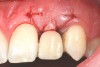
In the esthetic zone it is preferable to avoid elevating papillae because flap elevation may induce recession and create unesthetic black triangles.4 If a flap is necessary to perform a procedure in an edentate area when adjacent teeth are present, the following technique can be used to circumvent blunting papillae. Make a horizontal incision along the midcrestal or palatal aspect of the ridge and terminate the incision 1 mm from the adjacent teeth (Figure 1). The incision is created palatally if it is desired to transpose keratinized tissue to the buccal. From the horizontal incision, create bilateral buccal vertical releasing incisions that extend obliquely at an angle (Figure 2). The vertical incisions can also be extended palatally (for access), but this is not always necessary (Figure 3). Preserved papillae contain gingival supracrestal fibers that subsequently help maintain papillary height (Figure 4). The distance the incision is extended vertically on the buccal is dictated by the task to be accomplished (eg, implant insertion only requires short vertical incisions, while bone grafting needs longer incisions) (Figure 5 and Figure 6). At the end of the surgical procedure, the severed papillary segments are sutured to their retained counterparts (Figure 7 and Figure 8). Figure 1 through Figure 8 demonstrate a submerged implant protocol.
Papillae-Sparing Incisions Vs. Flap Elevation with Papillae—
In Figure 1 through Figure 8, tooth No. 8 is missing. If abundant bone and keratinized tissue is present, a punch procedure (flapless) can be used to access bone to place an implant. However, a flap should be elevated if there is a lack of keratinized tissue or if bone grafting or soft-tissue repair is required. There are two available options with respect to raising a flap: elevate it and include the papillae, or use papillae-sparing incisions. Each procedure provides advantages and disadvantages.
Papillae-sparing incisions can be made (as previously described) by creating vertical releasing incisions that provide a wider base for the flap than occurs coronally (Figure 2 and Figure 4 through Figure 6). The size of the flap and subsequent working area is initially dictated by the width of the edentate space. The flap can be enlarged by performing a papilla-sparing incision that is extended laterally by exaggerating the vertical incision angles, or the incisions can cross obliquely (laterally) at the mucogingival junction to a desired distance (Figure 9).
In contrast, a full-thickness flap with papillae elevation can be extended across many teeth, thereby providing extended access for completing procedures. However, with this technique there is a risk of inducing recession of elevated papillae.5,6
The decision to use a papilla-sparing incision versus a flap procedure is subjective and depends on the clinician’s preference. In segments of the arch other than the esthetic zone, a papilla-sparing incision can be used if there is adequate space between the teeth; however, creation of vertical releasing incisions may add to treatment time with respect to closure. Furthermore, the potential of inducing recession in the segments of the arch is not as critical as in the esthetic zone. Deschner et al7 demonstrated that an insignificant amount of recession occurred (0.4 mm mesial, 0.2 mm buccal, and 0.3 mm distal) when flap surgery (eg, apicoectomy) was performed at sites where the gingiva was healthy. Contrastingly, Velvart et al8 reported a mean 0.98 mm papillary recession 1 year after flaps and papillae were elevated, and no recession when a type of papilla-sparing incision (incisions made horizontally across the base of the papilla) was used. The reported papillary recession when they were elevated may have been a result of suturing at the base of the papillae as opposed to close to their tips, which did not ensure primary closure. No studies were found in the literature that compared the amount of recession that occurs when papillae-sparing incisions as described above were contrasted to flaps that elevated papillae.
Width of Severed Retained Papilla—
When performing a papilla-sparing incision, it is recommended that the width of the papilla left adjacent to the tooth be approximately 1 mm.9-11 Selection of 1 mm is an arbitrary thickness. It is possible that a narrower piece of tissue could be left; however, no investigations have addressed this issue. The distance from the tooth where the incision is made must be selected bearing in mind that the retained piece of papilla must encompass the following structures: oral sulcular epithelium (0.2 mm to 0.3 mm), junctional epithelium (15 to 30 cell layers, which decreases apically), and the sulcus width (the periodontal ligament being 0.25 mm).12,13 In addition, since the epithelium is avascular, connection with its vascular plexus subjacent to the oral and junctional epithelium must be included. Therefore, although the minimum dimension of the retained papillary segment that facilitates papilla retention is unknown, clinically, 1 mm seems to provide adequate papillary stability.9-11
Beveled Vs. Unbeveled Incision—
The vertical incision can be made perpendicularly to the bone, or a beveled incision (approximately 45°) can be made. Conceptually, the benefit of a beveled incision is the retention of connective tissue on the bone. Therefore, if the flap is imperfectly repositioned, bone will not be left exposed. A study conducted on monkeys that qualitatively compared healing results suggested that beveled incisions healed faster than perpendicular incisions.14 However, the vertical releasing incisions were not sutured. Accordingly, these data should not be interpreted to indicate that beveled incisions routinely heal faster than perpendicular incisions. The width of the bevel will be limited by the thickness of the tissue. On average, gingiva is 1.56 mm thick (ranging from 0.53 mm to 2.62 mm)15 and the epithelium is 0.3 mm thick.16
Several other factors should be considered when using beveled incisions. Very thin tissue cannot be beveled. In addition, if the flap margin is too thin, it may necrose. Also, suturing thin tissue may be difficult. If the tissue is thin before suturing, take a small instrument and elevate part of the fixed tissue adjacent to the incision line so that a suture can be placed without tearing the flap margin.
Length of Vertical Releasing Incisions—
The length of the buccal vertical releasing incisions is dictated by the extent of access needed to complete a given task. For example, to place an implant, the vertical incisions are extended to provide access to the crestal bone. Conversely, if a bone graft will be placed or keratinized tissue from the palate will be moved to the buccal aspect, then the vertical releasing incisions should extend to the vestibule to provide the ability to apically (translocating keratinized tissue) or coronally position the flap to cover the guided bone regeneration procedure. If additional coronal flap positioning is needed to accommodate a bone graft and barrier, periosteal fenestration should be performed.17 When greater flap advancement is required, the submucosa can be incised.17 To increase the zone of keratinized tissue on the buccal, the initial horizontal incision should be created several millimeters lingual to the center of the ridge and the tissue should be relocated buccally. Once vertical releasing incisions are used, the elevated tissue is actually a pedicle flap. It should always include connective tissue, which contains blood vessels. Furthermore, to ensure adequate flap vascularity, its length-to-width ratio should not exceed 2.5:1.18
Incisions Over Osseous Defects—
It is preferable to make vertical releasing incisions over intact bone. Incisions over defects increase the difficulty of suturing flap margins together and enhance the risk of getting a soft-tissue dehiscence or fenestration. If an incision is inadvertently created over an osseous defect, place a graft material into the defect and cover it with a membrane to ensure that the flap is supported when the margins are coapted. Suturing over bone reduces tension on the closed incision line.
Duration of Healing—
Long incisions will heal at the same rate as short ones.19 Healing occurs across the lateral aspects of the incision line. Because the lengths of incisions do not delay healing time, adequate access for procedures should be attained. In general, healing time of epithelium (after a 12-hour lag time) is 0.5 mm to 1 mm/day, and healing time of connective tissue is approximately 0.5 mm/day.20 It takes 4 days for angiogenesis to be restored across an incision line.21
Hiatt et al22 assessed the time needed before a flap is resistant to displacement. After flap surgery on dogs, they used a tensiometer and reported the force required to separate a flap from bone. At 2 to 3 days, 225 g of force was necessary; at 1 week, 340 g of force was required; and at 2 weeks, 1700 g of force could not displace the flap. In general, flaps are well attached in 10 days (dog model). However, it may take longer in humans. During the first week, collagen fibrils hold the flap weakly in place.
Blood Supply to Papillae and Edentate Areas—
When executing papillae-sparing incisions, the vascular plexus adjacent to the oral and junctional epithelium remains present. Similarly, the blood supply from the periodontal ligament and from the alveolar process is retained. However, it is unknown if segmenting the papillae temporarily alters papillary vascularity. Based on clinical results, healing of the papillae does not appear to be compromised. Contrastingly, the supraperiosteal vessels providing vascularity to the bone are disrupted when the flap is raised.
Bone Loss After Flap Elevation—
Gomez-Roman et al9 compared the amount of mean bone loss that occurs after a papilla-sparing incision versus a flap with elevated papilla (0.29 mm [± 0.38] versus 1.12 mm [± 1.14], respectively). The range of bone loss after papillae-sparing incisions versus post-flap elevation was 0 to 1 mm versus 0 to 3.5 mm, respectively. They concluded that papillae-sparing incisions resulted in less bone loss interproximally. However, standard deviations (SD) around the means were large, which indicates there was a great deal of variation among assessed patients. Pertinently, the SD concerning bone loss was much smaller related to papillae-sparing incisions.
The quantity of bone loss that occurs when a flap is raised is a relevant subject when discussing papillae-sparing incisions. In general, raising a flap without tooth removal results in a mean bone diminution of 0.5 mm.23,24 The extent of bone loss is typically larger over the buccal aspect of the roots than interproximally where the bone is thicker.23 The bone loss is a result of the elevation of the periosteum, which reduces blood supply to the bone.
In this regard, several investigators reported that avoiding flap elevation decreased buccal vertical bone resorption after tooth extraction and immediate implant insertion in humans25 and in animal experimental studies.26-28 Contrastingly, researchers in humans29-31 and in animal studies32-34 reported that even with a flapless approach, the degree of vertical bone loss was comparable with and without flap elevation. With respect to horizontal bone loss, several recent articles indicated there is greater preservation of the horizontal bone width if a flap is not raised.35-37
Despite contradictory data with respect to vertical bone loss associated with and without flap elevation, it is the authors’ opinion that if tissues are left untouched (no flap elevation) there is an improved chance of decreasing soft-tissue recession. In this regard, not elevating a segment of papillae adjacent to teeth in the esthetic zone has consistently been reported to be beneficial by numerous authors.9,38-40
Relationship Between Bone Loss and Recession—
Gomez-Roman et al9 demonstrated increased bone loss without papillae-sparing incisions; however, they did not record soft-tissue measurements. Therefore, it cannot be assumed that papillae-sparing incisions resulted in less recession. Furthermore, it should be noted that it is possible to have bone loss and increased probing depths without recession (eg, a periodontal pocket). Another factor that may affect this relationship is tissue biotype. In general, after surgical procedures, it is expected that a thick biotype will recede less than a thin biotype.41
Including or Excluding the Maxillary Anterior Frenum in the Incision Design—
The maxillary anterior frenum is a mucosal structure that usually extends from the midline of the maxillary gingiva between the central incisors into the vestibular region and the middle of the upper lip.42 The size, shape, and point of attachment may vary. It rarely contains muscle fibers from the orbicularis oris muscle. Depending on the location of the frenum, the papillary-sparing incision can include or exclude this structure. However, excluding it should not come at the expense of altering the flap design, which could result in the coronal aspect of the flap being wider than the base of the flap. Avoidance of incising the frenum is recommended since it is easier to suture the vertical incision line when it is not included.
Management of a Large Diastema Between Anterior Teeth—
If there is a large diastema between the central incisors (eg, teeth Nos. 8 and 9) and one incisor is missing (eg, tooth No. 8), the vertical releasing incision should not include a piece of the mesial papilla on tooth No. 9 when employing a papilla-sparing incision to insert an implant at the site of tooth No. 8. The vertical releasing incision should be made mesial to the papilla of tooth No. 9.
Scar Formation—
A scar is a histological disturbance of the normal structure of tissue. It is characterized by a lack of specific organization of cellular and matrix elements and an increased amount of collagen when compared to uninjured tissue. Hypertrophic scars and keloids are examples of fibroproliferative disorders with an excessive accumulation of collagen within a wound. An atrophic scar is characterized by an indentation or a groove; it is not a real scar. If primary closure of the vertical incisions of papillae-sparing incisions is not achieved, then an indentation may be noticed.
Clinical observations indicate that surgical wounds—especially in attached gingiva and palatally—do not usually scar; however, mucosa may heal with a scar.8,43 It is possible that lack of scarring in gingiva versus mucosa is the result of a lack of elastic fibers in the gingiva compared to mucosa.44 It is also possible that the explanation could be more complicated. Guo et al45 reported that scarless tissue repair could be due to the reduced production of endothelin by gingival fibroblasts. Conversely, Eslami et al46 suggested that prolonged expression of αvβ6 integrin along with transforming growth factor-β3 in the gingival wound epithelium may be important in protecting gingiva from scar formation. Many possible reasons have been proposed, but the precise reason why gingiva heals without scarring remains unknown.
Patient Comfort—
When performing oral surgery, a number of general concepts should be considered to optimize patient comfort. These include: always managing tissues gently; keeping the periosteal elevator on the bone at all times; and suctioning on bone in a sweeping motion to avoid irritating the soft tissues. Precise incisions that are well coapted heal by primary intention and mend more quickly than incisions that repair by secondary intention. Before and after suturing, wet gauze should be pressed directly on the pedicle flap. This will help remove any excess blood under the flap and facilitate more rapid initial flap adherence.
Reduced treatment time results in less time that bone is exposed and also decreases soft-tissue desiccation. All surgeries cause increased interstitial fluid (edema), inflammation, and the release of bradykinins and histamines47; therefore, reduced surgical time and precise incisions result in less swelling and less postoperative discomfort for the patient.17
Suturing—
A 5-0 chromic gut suture with a half round needle is very effective for suturing vertical releasing incisions, especially when the tissue is thin. The needle should engage 2 mm to 3 mm of tissue, and the sutures should be placed every 3 mm to 5 mm. However, do not suture the pedicle to the retained papillary segment, because the tissue is too thin. Sutures should be snugged down but not pulled too tightly, otherwise they can cause pressure necrosis. Movable tissue should be sutured to fixed tissue, and sutures should not be directly suctioned.
Shape of the Temporary Healing Abutment and Crown—
After papilla-sparing incisions and graft procedures are completed, the tissue is distended. Therefore, if an immediate crown is placed, use straight (flat) healing abutments and a flat temporary crown, which will not expand the volume under the replaced flap. Furthermore, straight or concave prostheses facilitate moving gingiva coronally.48 After the tissue heals, a convex healing abutment and temporary crown can be placed to stretch and restore gingival contour. In this regard, a large interproximal convexity causes papillae to migrate coronally.48 In general, it takes 3 months for tissue to migrate,49 and 3 to 6 months should pass before restoring a tooth in a critically esthetic area.50
Other Types of Papillae-Sparing Incisions
Several different techniques have been suggested with respect to avoiding papillary shrinkage. In 1989, Takei et al51 recommended elevating only a palatal flap to circumvent inducing a lot of recession in the esthetic zone in periodontal patients. The papillae were severed on the lingual aspect but not elevated. This technique was a compromise with respect to pocket elimination but provided a better esthetic result than elevating a buccal flap. In contrast, Cortellini et al52 advocated elevating a palatal flap and elevating papillae from the lingual. This facilitated performing regenerative procedures and attaining primary closure. Another technique was proposed by Velvart et al.8 They made an incision at the base of the buccal papillae and raised a flap without elevating the papillae. This type of technique was recommended for apicoectomies. Each of these procedures has advantages; however, none provided access to the ridge and buccal bony plate without elevating the papilla as the procedure described in this article does.
Potential Complications
There are several issues that can arise when executing papillary-sparing incisions, including the inability to close incision lines. If graft material prevents primary closure of buccal vertical incision lines because of increased tissue volume, the vertical incisions should be extended apically and a periosteal fenestration incision is created at the base of the flap to advance it. This facilitates coronally positioning a wider part of the flap. Accordingly, it is important that the base of the flap be wider than the coronal aspect.
Another issue involves soft-tissue dehiscences along a vertical incision line. This can be avoided if the coronally positioned flap is tension-free before suturing. If a dehiscence occurs, a subepithelial connective tissue graft will need to be performed.
Lastly, there is potential for postoperative soft-tissue dehiscence or failure of the bone graft to mature. Transitional acrylic partials should not touch the incision lines or induce pressure on a grafted area, as this will result in a soft-tissue dehiscence or the bone graft being unsuccessful.
Case Reports: Utility of Papillae-Sparing Incisions
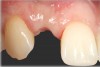
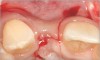
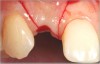
There are numerous scenarios when a papilla-sparing incision can be advantageously used, including implant placement, the reconstruction of soft tissue and bone, or both. Figure 10 through Figure 18 and Figure 19 through Figure 25 illustrate how papillae-sparing incisions can be used to restore form and function using a nonsubmerged implant protocol. (Surgeries were performed by DT.)
Conclusion
Papillae-sparing incisions facilitate accessing the bone to place dental implants and restore compromised osseous and gingival architecture. Their major benefit is to enable surgical procedures to be performed without inducing recession of papillae adjacent to treated sites. It should be noted that sometimes more than one surgical procedure may be required to correct osseous or gingival deficiencies. In addition, at times, it may be necessary to restore teeth adjacent to a compromised implant site to attain optimal esthetics. Pre-planning using diagnostic radiographs, wax-ups, and attention to detail before and during implant-related procedures can help avoid problems and assist in achieving desired clinical outcomes.
About The Authors
Gary Greenstein, DDS
Clinical Professor, College of Dental Medicine, Columbia University, New York, New York;
Private Practice, Surgical Implantology and Periodontics, Freehold, New Jersey
Dennis Tarnow, DDS
Director of Dental Implant Education, Clinical Professor, Department of Periodontology, College of Dental Medicine, Columbia University, New York, New York; Private Practice, Surgical Implantology and Prosthodontics, New York, New York
Queries to the author regarding this course may be submitted to authorqueries@aegiscomm.com.
References
1. Becker W, Ochsenbein C, Tibbets L, Becker BE. Alveolar bone anatomic profiles as measured from dry skulls. J Clin Periodontol. 1997;24(10):727-731.
2. Spear FM. The esthetic management of multiple missing anterior teeth. Inside Dentistry. 2007;3(1):72-76.
3. Kois JC. Altering gingival levels: the restorative connection part I: biologic variables. J Esthet Restor Dent. 1994;6(1):3-7.
4. Sharma AA, Park JH. Esthetic considerations in interdental papilla: remediation and regeneration. J Esthet Restor Dent. 2010;22(1):18-28.
5. Buser D, Wittneben J, Bornstein MM, et al. Stability of contour augmentation and esthetic outcomes of implant-supported single crowns in the esthetic zone: 3-year results of a prospective study with early implant placement postextraction. J Periodontol. 2011;82(3):342-349.
6. Buser D, Bornstein MM, Weber HP, et al. Early implant placement with simultaneous guided bone regeneration following single-tooth extraction in the esthetic zone: a cross-sectional, retrospective study in 45 subjects with a 2- to 4-year follow-up. J Periodontol. 2008;79(9):1773-1781.
7. Deschner J, Wolff S, Hedderich J, et al. Dimensional changes of periodontal soft tissues after intrasulcular incision. Clin Oral Investig. 2009;13(4):401-408.
8. Velvart P, Ebner-Zimmermann U, Ebner JP. Comparison of long-term papilla healing following sulcular full thickness flap and papilla base flap in endodontic surgery. Int Endod J. 2004;37(10):687-693.
9. Gomez-Roman G. Influence of flap design on peri-implant interproximal crestal bone loss around single-tooth implants. Int J Oral Maxillofac Implants. 2001;16(1):61-67.
10. Cavallaro JS Jr, Greenstein G, Tarnow DP. Clinical pearls for surgical implant dentistry: Part 3. Dent Today. 2010;29(10):134-139.
11. Al-Sabbagh M. Implants in the esthetic zone. Dent Clin North Am. 2006;50(3):391-407.
12. Nanci A, Bosshardt DD. Structure of periodontal tissues in health and disease. Periodontol 2000. 2006;40:11-28.
13. Hassell TM. Tissues and cells of the periodontium. Periodontol 2000. 1993;3:9-38.
14. Kon S, Caffesse RG, Castelli WA, Nasjleti CE. Vertical releasing incisions for flap design: clinical and histological study in monkeys. Int J Periodontics Restorative Dent. 1984;4(1):48-57.
15. Goaslind GD, Robertson PB, Mahan CJ, et al. Thickness of facial gingiva. J Periodontol. 1977;48(12):768-771.
16. Müller HP. Anatomy and physiology. In: Müller HP, ed. Periodontology: The Essentials. Stuttgart, Germany: Thieme; 2004:2-11.
17. Greenstein G, Greenstein B, Cavallaro J, et al. Flap advancement: practical techniques to attain tension-free primary closure. J Periodontol. 2009;80(1):4-15.
18. Cranin AN. Implant surgery: the management of soft tissues. J Oral Implantol. 2002;28(5):230-237.
19. Incisions and Healing in Animals. Doctors Foster and Smith Web site. www.peteducation.com/article.cfm?c=2+2116&aid=3025. Accessed March 13, 2014.
20. Engler WO, Ramfjord SP, Hiniker JJ. Healing following simple gingivectomy. A tritiated thymidine radioautographic study. I. Epithelialization. J Periodontol. 1966;37(4):298-308.
21. Midwood KS, Williams LV, Schwarzbauer JE. Tissue repair and the dynamics of the extracellular matrix. Int J Biochem Cell Biol. 2004;36(6):1031-1037.
22. Hiatt WH, Stallard RE, Butler ED, Badgett B. Repair following mucoperiosteal flap surgery with full gingival retention. J Periodontol. 1968;39(1):11-16.
23. Donnenfeld OW, Hoag PM, Weissman DP. A clinical study on the effects of osteoplasty. J Periodontol. 1970;41(3):131-141.
24. Wilderman MN, Pennel BM, King K, Barron JM. Histogenesis of repair following osseous surgery. J Periodontol. 1970;41(10):551-565.
25. Job S, Bhat V, Naidu EM. In vivo evaluation of crestal bone heights following implant placement with “flapless” and “with-flap” techniques in sites of immediately loaded implants. Indian J Dent Res. 2008;19(4):320-325.
26. Fickl S, Zuhr O, Wachtel H, et al. Tissue alterations after tooth extraction with and without surgical trauma: a volumetric study in the beagle dog. J Clin Periodontol. 2008;35(4):356-363.
27. Barros RRM, Novaes Jr AB, Papalexiou V. Buccal bone remodeling after immediate implantation with a flap or flapless approach: a pilot study in dogs. The Int J of Dent Implants and Biomaterials Titanium. 2009;1:45-51.
28. Blanco J, Nuñez V, Aracil L, et al. Ridge alterations following immediate implant placement in the dog: flap versus flapless surgery. J Clin Periodontol. 2008;35(7);640-648.
29. Maló P, Nobre M. Flap vs. flapless surgical techniques at immediate implant function in predominantly soft bone for rehabilitation of partial edentulism: a prospective cohort study with follow-up of 1 year. Eur J Oral Implantol. 2008;1(4):293-304.
30. Nickenig HJ, Wichmann M, Schlegel KA, et al. Radiographic evaluation of marginal bone levels during healing period, adjacent to parallel-screw cylinder implants inserted in the posterior zone of the jaws, placed with flapless surgery. Clin Oral Implants Res. 2010;21(12):1386-1393.
31. De Bruyn H, Atashkadeh M, Cosyn J, van de Velde T. Clinical outcome and bone preservation of single TiUnite™ implants installed with flapless or flap surgery. Clin Implant Dent Relat Res. 2011;13(3):175-183.
32. Caneva M, Botticelli D, Salata LA, et al. Flap vs. “flapless” surgical approach at immediate implants: a histomorphometric study in dogs. Clinical Oral Implants Res. 2010;21(12):1314-1319.
33. Araújo MG, Lindhe J. Ridge alterations following tooth extraction with and without flap elevation: an experimental study in the dog. Clin Oral Implants Res. 2009;20(6):545-549.
34. Becker W, Wikesjö UM, Sennerby L, et al. Histologic evaluation of implants following flapless and flapped surgery: a study in canines. J Periodontol. 2006;77(10):1717-1722.
35. Vera C, De Kok IJ, Chen W, et al. Evaluation of post-implant buccal bone resorption using cone beam computed tomography: a clinical pilot study. Int J Oral Maxillofac Implants. 2012;27(5):1249-1257.
36. Degidi M, Daprile G, Nardi D, Piattelli A. Buccal bone plate in immediately placed and restored implant with Bio-Oss(®) collagen graft: a 1-year follow-up study [published online ahead of print August 13, 2012]. Clin Oral Implants Res. doi: 10.1111/j.1600-0501.2012.02561.x.
37. Brownfield LA, Weltman RL. Ridge preservation with or without an osteoinductive allograft: a clinical, radiographic, micro-computed tomography, and histologic study evaluating dimensional changes and new bone formation of the alveolar ridge. J Periodontol. 2012;83(5):581-589.
38. Simon Z, Rosenblatt A. Challenges in achieving gingival harmony. J Calif Dent Assoc. 2010;38(8):583-590.
39. Present S, Levine R. Single maxillary anterior tooth restoration. Inside Dentistry. 2010;6(1):58-66.
40. Gottesman E. Ridge augmentation in the esthetic zone to achieve prosthetically guided implant positioning. Dent News Philippines. 2012;13-15.
41. Lee A, Fu JH, Wang HL. Soft tissue biotype affects implant success. Implant Dent. 2011;20(3):e38-e47.
42. Sadeghi EM, Van Swol RL, Eslami A. Histologic analysis of the hyperplastic maxillary anterior frenum. J Oral Maxillofac Surg. 1984;42(12):765-770.
43. von Arx T, Salvi GE, Janner S, Jensen SS. Gingival recession following apical surgery in the esthetic zone: a clinical study with 70 cases. Eur J Esthet Dent. 2009;4(1):28-45.
44. Roten SV, Bhat S, Bhawan J. Elastic fibers in scar tissue. J Cutan Pathol. 1996;23(1):37-42.
45. Guo F, Carter DE, Leask A. Mechanical tension increases CCN2/CTGF expression and proliferation in gingival fibroblasts via a TGFβ-dependent mechanism. PLoS One. 2011;6(5):e19756.
46. Eslami A, Gallant-Behm CL, Hart DA, et al. Expression of integrin alphavbeta6 and TGF-beta in scarless vs scar-forming wound healing. J Histochem Cytochem. 2009;57(6):543-557.
47. Petho G, Reeh PW. Sensory and signaling mechanisms of bradykinin, eicosanoids, platelet-activating factor, and nitric oxide in peripheral nociceptors. Physiol Rev. 2012;92(4):1699-1775.
48. Bichacho N, Landsberg CJ. Single implant restorations: prosthetically induced soft tissue topography. Pract Periodontics Aesthet Dent. 1997;9(7):745-752.
49. Kinsel RP, Capoferri D. A simplified method to develop optimal gingival contours for the single implant-supported, metal-ceramic crown in the aesthetic zone. Pract Proced Aesthet Dent. 2008;20(4):231-236.
50. Small PN, Tarnow DP. Gingival recession around implants: a 1-year longitudinal prospective study. Int J Oral Maxillofac Implants. 2000;15(4):527-532.
51. Takei H, Yamada H, Hau T. Maxillary anterior esthetics. Preservation of the interdental papilla. Dent Clin North Am. 1989;33(2):263-273.
52. Cortellini P, Prato GP, Tonetti MS. The modified papilla preservation technique. A new surgical approach for interproximal regenerative procedures. J Periodontol. 1995;66(4):261-266.