You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Introduction
An operculum is a flap of gingival tissue typically distal to a molar that remains as a sequela of the eruption.1 In some cases, an operculum can lead either to pain caused by the occlusion of the opposing maxillary molar (in this instance, the pericoronal gingiva becomes inflamed and often ulcerated because of trauma) or by local inflammation/infection because of food debris and plaque accumulation between the tooth surface and the gingival flap.1 The inflammation in the soft tissues surrounding the crown of the partially erupted tooth is called pericoronitis.2,3 It typically occurs in teeth that erupt very slowly or become impacted. When the follicle of the tooth communicates with the oral cavity, bacterial penetration into the follicular space instigates the infection.2 It is generally agreed that this process is potentiated by food debris trapped in proximity to the operculum and occlusal trauma of the pericoronal tissues by the opposing tooth.2,4 Pericoronitis may be acute or chronic. Acute pericoronitis is characterized by severe radiating pain, localized swelling of the pericoronal tissues, purulence or drainage (hence an unpleasant taste), trismus, regional lymphadenopathy, painful swallowing, pyrexia, and sometimes spreading of the infection to adjacent tissue spaces.2-4 The chronic form may be characterized by dull pain or discomfort, often accompanied by bad taste, lasting a couple of days with remission lasting several weeks or months.2,4 Stress, pregnancy, and/or fatigue are often associated with an increased occurrence of pericoronitis. Diagnosis is done on the basis of the symptoms and clinical presentation.
Treatment
The painful and/or inflamed/infected operculum can be removed by operculectomy—the surgical excision or ablation of the operculum via a wide variety of techniques: scalpel, caustic agents, radiofrequency surgery, electrosurgery, cautery, CO2 laser, or hot-tip diode surgery.1,2,4-6 Operculectomy, however, is also advocated as a precaution to prevent pericoronitis. In orthodontics, operculectomy is performed to aid in molar eruption (even in the absence of pain) and/or to gain access to the tooth for banding or bonding purposes.1,7,8 Convissar et al9 and Chmura7 both state that removing gingiva with a laser allows for immediate banding or bonding because of its hemostatic effect.
One of the potential disadvantages of operculectomy is that it may prove to be a temporary measure, because an operculum can regrow, which may lead to new trauma and inflammation.3 In such cases the removal of the offending molar may be indicated.2,3
Soft-Tissue Laser Surgery
The most valuable advantage of soft-tissue lasers over other surgical modalities,10-29 is their ability to precisely cut and efficiently provide hemostasis.
However, not all lasers are equally efficient at both cutting and coagulating. In order to understand how exactly lasers cut and coagulate, one should consider the wavelength-dependent nature of laser light’s interaction with soft tissue; namely, light absorption and light scattering by the soft tissue.
Laser Light Absorption and Scattering by the Soft Tissue
Figure 1 presents the modern understanding30 of how various laser wavelengths interact with the main chromophores, or absorption centers, in the oral soft tissue. Shown are the following three main wavelength groups of practical dental lasers:
• circa 1,000 nm (diodes and Nd:YAG laser);
• circa 3,000 nm (Erbium lasers); and
• circa 10,000 nm (CO2 lasers).
At Erbium and CO2 laser wavelengths, light scattering by the soft tissue is insignificant.31-33 At near-infrared diode and Nd:YAG laser wavelengths, soft-tissue light scattering dominates over absorption,30-33 which makes these wavelengths poorly suited for precise ablation, incision, and excision.31-34
Soft-Tissue Laser Ablation
Soft-tissue laser ablation (and incision and excision) is a process of vaporization of intra- and extracellular water heated by the laser light within the irradiated soft tissue.31-33 Water vapors, rapidly steaming out of the intensely laser-heated soft tissue, carry with them cellular ashes and other byproducts of this fast boiling and vaporization process.
Because the absorption of the near-infrared diode and Nd:YAG laser wavelengths (circa 1,000 nm) is weak and scattering by the soft tissue is strong, these wavelengths are inefficient and spatially inaccurate laser ablation devices.31-33 Due to very strong absorption by the soft tissue, mid-infrared Erbium (circa 3,000 nm) and infrared CO2 laser (circa 10,000 nm) wavelengths are efficient and spatially accurate laser ablation tools.31-33
Soft-Tissue Laser Coagulation and Hemostasis
Coagulation takes place as soft-tissue proteins denaturate in the 60°C to 100°C temperature range, leading to a significant reduction in bleeding and oozing of lymphatic liquids on the margins of ablated tissue during laser ablation, excision, and incision procedures. Due to the fact that blood is carried through a system of blood vessels, the blood vessel diameter (ranging between 21 µm and 40 µm, with an average value of 31 µm, based on measurements in human cadaver gingival connective tissue35) is a significant spatial parameter that influences the efficiency of the coagulation process. Collagen shrinks at increased temperatures, which in turn shrinks blood and lymphatic vessel walls, causing hemostasis during laser coagulation.
For short laser pulses, the photothermal coagulation depth during laser ablation of the soft tissue is proportional to absorption depth (or inversely proportional to the absorption coefficient from Figure 1).
For Erbium laser wavelengths of circa 3,000 nm, optical absorption and coagulation depths are significantly smaller than gingival blood vessel diameters. Coagulation takes place on a relatively small spatial scale and cannot prevent bleeding from the blood vessels severed during tissue ablation. Coagulation depth can be increased by pulse width/rate increase, and by pulse power/fluence decrease.
For diode and Nd:YAG laser wavelengths of circa 1,000 nm, optical absorption and coagulation depths are significantly greater than blood vessel diameters. Coagulation takes place over extended volumes—far away from the intended ablation site where no coagulation is required.
For CO2 laser wavelengths of circa 10,000 nm, optical absorption and coagulation depths are of the same order as gingival blood vessel diameters. Coagulation extends just deep enough into a severed blood vessel to stop the bleeding. Coagulation depth can be increased by an increase in pulse width/rate, and by reducing pulse power/fluence.
Optimal Wavelength for Soft-Tissue Laser Surgery
Wavelengths circa 10,000 nm are >1,000 times superior to wavelengths circa 1,000 nm for soft-tissue ablation, and >10 times superior to wavelengths circa 3,000 nm for soft-tissue coagulation and hemostasis. The wavelengths circa 10,000 nm (eg, the CO2 laser) deliver both soft-tissue ablation and simultaneous coagulation, which is unobtainable with either diodes (circa 1,000 nm) or Erbium (circa 3,000 nm) wavelengths.
CO2 Laser Soft-Tissue Surgery
The CO2 laser photo-thermally cuts, ablates, and coagulates the oral soft tissues. The key to the success of the soft-tissue CO2 laser is its ability to cut and coagulate tissue simultaneously. The CO2 laser treatment of inflamed and/or hyperplastic oral lesions, including pericoronitis, offers several advantages over conventional scalpel surgery, such as non-contact incision (which ensures no mechanical trauma to the tissue), reduced-to-no-bleeding, a lack of need for sutures, precise tissue removal, and minimized postoperative pain and edema. The use of a CO2 laser on the oral soft tissue has no known contraindications or side effects.18 The excellent hemostatic capacity of the CO2 laser is described as a useful instrument for oral surgery in patients with hemorrhagic disorders or undergoing antithrombotic therapy.16-18
Laser Pulsing and Thermal Relaxation Time
The application of laser energy over an extended period of time may result in tissue necrosis due to thermal diffusion of heat from the irradiated tissue and charring on the margins of the laser incision. Proper pulsing is crucial for the appropriate application of laser energy during soft-tissue laser ablation and coagulation.31 The laser-irradiated tissue heats most efficiently when the duration of the laser pulse is much shorter than the thermal relaxation time (which is laser-wavelength and tissue-type specific). Optimal cooling is achieved when the duration between laser pulses is much greater than the thermal relaxation time.
The so-called “SuperPulse” design32 for CO2 laser pulsing parameters is optimized around the thermal relaxation time concept discussed above. SuperPulse ensures char-free soft-tissue ablation, incision, and excision with variable depths of coagulation/hemostasis on the margins of the cut.
The SuperPulse mode (Figure 2) is made with bursts of short laser pulses with very high peak power that are spaced far enough apart for efficient tissue cooling between the pulses. The SuperPulse minimizes the amount of heat diffusing from the cutting/ablation zone to the adjacent healthy tissue.
Laser Handpieces
Since the early days of surgical CO2 lasers in the 1970s and 1980s, the wide adoption of the technology had been inhibited by the articulated beam delivery system. The invention of the flexible hollow-fiber CO2 laser beam delivery system in the late 1980s changed the paradigm for CO2 laser surgery. The present-day flexible-fiber CO2 laser handpiece is light, pen-sized, disposable-free, autoclavable, and easily switches between (1) incision with photo-coagulation; (2) superficial ablation with photo-coagulation; and (3) photo-coagulation. There are two types of flexible-fiber CO2 laser handpieces; namely, tip-retainer and tipless.
Tip-retainer laser handpieces use disposable hollow focusing tips made of high-temperature resistant aluminum-oxide ceramic (Figure 3) with a 250-µm spot size to allow for intrasulcular periodontal applications.
Autoclavable “tipless” handpieces closely simulate the scalpel-like experience for the clinician, but with no mechanical contact with the tissue. Achieving the designated beam spot size for cutting requires a 1-mm to 3-mm distance between the distal end of the handpiece and the target tissue.
Laser Power Density
An important parameter to consider when performing a CO2 laser surgery is laser power density. An analogy can be drawn here: regardless of how sharp the blade is, there will be no interaction between the blade and the tissue unless mechanical pressure is applied to the blade, forcing it through the tissue. For the CO2 laser, the power density of the focused laser beam is equivalent to the mechanical pressure that is applied to a cold steel blade—the greater the laser power density, the greater the rate of soft-tissue removal.
Laser Beam Spot Sizes for Cutting and Coagulation
To continue the analogy with a scalpel, the size of the laser beam focal spot defines the quality of the laser cut the same way as the sharpness of the steel blade defines the quality and the ease of the incision. Just as a dull blade cannot produce a quality incision, an oversized laser beam spot cannot produce a good quality incision. The smaller (or sharper) the focal spot of the beam, the narrower and deeper the incision. For a rapid switch from cutting to just photo-coagulation, the laser beam can be defocused by simply moving the handpiece away from the tissue by approximately 6 mm to 9 mm, and “painting” the “bleeder” for enhanced hemostasis (Figure 4 and Figure 5).
Wound Healing
The excellent hemostatic ability of the CO2 laser provides the clinician with improved visibility of the surgical field and, therefore, allows for more precise and accurate tissue removal.19 Due to efficient hemostasis, intraoral surgical wounds often do not require suturing or surgical dressing and can be left to heal by secondary intention.20
Significantly reduced wound contraction and scarring are among the most important advantages of CO2 laser treatment.20,21-23 In CO2 laser-irradiated wounds, the healing process is characterized by a more prominent fibroblastic proliferation, with young fibroblasts actively producing collagen. In comparison with scalpel wounds, only a small number of myofibroblasts (the cells responsible for wound contraction) is found in CO2 laser-excised wounds.23-25 At 72 hours postoperatively, a fibroserous membrane appears over the CO2 laser-irradiated wounds to replace the superficial necrotic layer of the laser-treated area.26,27 An epithelial covering of the wound starts to form from the periphery toward the center after 2 weeks, and is thinner and parakeratotic in comparison with the epithelium that forms after scalpel resection. This could account for the excellent esthetic outcome of all CO2 laser treatment, with no fibrosis or scarring and soft pliable residual tissue, while a scalpel can leave some scarring.22
Another advantage of the CO2 laser is minimal postoperative swelling and edema due to the intraoperative closure of lymphatic vessels on the margins of the CO2 laser incision. Lymphatic vessels regenerate in approximately 8 to 10 days after capillary-vessel proliferation.28
Decreased wound contraction combined with minimal lateral tissue damage, less traumatic surgery, more precise control of the depth of tissue damage, and excellent hemostatic ability make the CO2 laser a safe and efficient alternative to the conventional scalpel. Strauss et al8 and Deppe et al15 compared the recovery process after CO2 laser surgery with that after cryosurgery or electrosurgery and reported that the CO2 laser healing was faster and less painful.
CO2 Laser for Operculectomy
The inherent advantages of the CO2 laser, such as excellent hemostasis, diminished postoperative edema, and uneventful healing combined with the reduced risk of scarring, make the CO2 laser the most efficient surgical treatment method for operculectomy.7,9,16-18,28,37 In order to ensure the safest and most dependable treatment, the clinician should always rely on the basic principles of laser surgery physics8 using correct laser parameters (ie, SuperPulse) and the sharpest focal laser spot (ie, 250-µm diameter) with the ability to rapidly defocus for enhanced coagulation and hemostasis.
Operculectomy by CO2 Laser Ablation: Case Study
Initial Findings
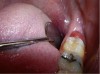
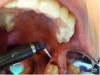
The patient presented with a raised, inflamed, painful operculum distal to the left mandibular second molar (Figure 6). The lesion was red, edematous, and bled easily. A periodontal pocket on the distal of the second molar was >6 mm deep. There was no evidence of abscess.
Diagnosis and Treatment Plan
The patient had been previously treated with a scalpel. He returned 24 hours later with severe pain and a granulomatous lesion present. The lesion was clinically diagnosed as chronic pericoronitis. It was decided to surgically remove the inflamed pericoronal tissue using a CO2 laser-ablation technique.
Surgical Laser Equipment and Settings
A flexible-fiber dental CO2 laser with two different autoclavable dental handpieces was used: (1) a dental angled tipless handpiece with a 0.25-mm spot diameter; and (2) a dental angled tip-retainer handpiece with a sterile, disposable, tapered ceramic with a 0.25-mm spot diameter periodontal tip.
For ablation of the inflamed tissue, the laser was set to 4 W in the SuperPulse mode with repeat pulsing at a 25 millisecond pulse-width and 29-Hz repetition rate. The tipless dental handpiece was used at a 3-mm to 5-mm nozzle-to-tissue distance.
For sulcus decontamination, the laser was set to 2 W in the SuperPulse mode with repeat pulsing at a 15-millisecond pulse-width and 20-Hz repetition rate. The tip-retainer dental handpiece was used with the periodontal tip’s distal end approximately 1 mm from the base of the pocket and parallel to the surface of the tooth; an air purge was constantly supplied through the tip and was set to the highest flow.
CO2 Laser Surgery
The procedure steps were as follows:
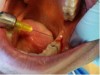
1. Anesthesia was administered by the local infiltration technique: 2% lidocaine with 1:100,000 epinephrine; a 30-gauge needle was used (Figure 7).
2. The angled handpiece of the CO2 laser was held perpendicular to the target lesion (Figure 8) and the ablation of the inflamed tissue began (Figure 9). The dental surgeon was careful to avoid contact between the laser beam and the tooth enamel. (Note: In this case, the inflamed tissue was located distally from the involved molar. There was no direct contact of the beam with the tooth, as the surgeon was careful not to hit the enamel. However, if an operculum covers part of the tooth, an adaptive tool (for example, a wax spatula) needs to be inserted between the tissue and the tooth to prevent possible damage. For an inexperienced CO2 laser surgeon, it is important and highly recommended to always shield the tooth during the procedure.6)
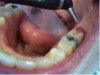
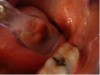
3. After the initial pass with the laser, the inflamed gingiva bled slightly and the surgeon defocused the beam by increasing the nozzle-to-tissue distance to quickly obtain hemostasis (Figure 10). Note the excellent visualization and the clear operatory field.
4. Additional tissue around the primary surgical site was superficially ablated to “feather out” the wound and make the treated area blend with the healthy gingiva better (Figure 11).
5. After the removal of lesion, a periodontal tip was placed approximately 1 mm from the base of the pocket to decontaminate the sulcus area. Laser energy was administered using horizontal movements along the entire distal sulcular aspect.
6. Finally, the laser was defocused by moving the handpiece farther away from the surgical area and additional hemostasis was created. No suturing or dressing was needed and the wound was left to heal by secondary intention.
Postoperative Instructions
The patient was dismissed with instructions to do warm salt water rinses and mild stimulation of the site with a rubber tip twice a day; he was also instructed to apply topical antibiotic and vitamin E gel twice daily directly to the area. The patient was advised to avoid spicy, acidic, or harsh foods or caustic mouthrinses. Although the patient was prescribed Vicodin 7.5, he did not need pain medications and was happy with the result. He reported to be completely pain-free in 24 hours.
Follow-up Examination
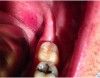
One week after the procedure, healing had progressed well (Figure 12). No swelling or inflammation was noted. There were no complaints from the patient during the postoperative period. The 5-week follow-up visit showed beautifully healed tissue with no scarring (Figure 13). The recovery was uneventful. The gingival overgrowth did not recur.
Non-Laser Surgical Treatment Modalities
Soft-tissue intraoral lesions, including pericoronitis, have been treated with a number of non-laser surgical methods, such as scalpel surgery (used most frequently), cryosurgery, electrosurgery, electrocautery, radiofrequency surgery, and hot-tip diode excision.2,3,10
Scalpel Surgery
Surgical excision with a scalpel is the most widely used form of treatment. Among the drawbacks of conventional scalpel surgery is the need for intra- and postoperative bleeding management, suturing, and the risk of postoperative edema and infection. Furthermore, Haytac and Ozcelik37 evaluated and compared patient perceptions after frenectomy surgeries performed with the CO2 laser and traditional scalpel techniques. They concluded that CO2 laser treatment provides better patient perception in terms of postoperative pain and function than that obtained via scalpel surgery. They also inferred that “the CO2 laser offers a safe, effective, acceptable, and impressive alternative” to scalpel.37
Cryosurgery
Cryosurgery is a method of tissue destruction by freezing and has become an established surgical technique for the treatment of diverse benign and malignant cutaneous lesions12,13 and is used in dentistry.11,38 Although it is inexpensive, and has been used successfully for the treatment of various soft-tissue oral lesions,11 cryosurgery is time-consuming and has many contraindicated conditions, for example in patients with: cryoglobulinemia; cold urticarial vasculitis; agammaglobulinemia; dysfibrinogenemia; Raynaud’s disease and other collagen diseases; or platelet alterations, or are undergoing hemodialysis or immunosuppressive therapy.11,13 The possible complications include pain during freezing, edema and exudation, vesicles, and bullae. Finally, cryosurgery can result in pseudoepitheliomatous hyperplasia, postoperative infection, fever, and pyogenic granuloma.13 In addition, cryosurgery does not allow control of the depth of tissue removal with high precision to the extent that laser surgery does.
Electro- and Radiofrequency Surgery
The excessive heat with electrosurgery creates the potential for scarring. The study that compared thermal damages in different types of soft tissue with those created by electrosurgery14 concluded that the relatively narrow zone of thermal tissue necrosis makes CO2 laser excision superior for histologic examination of excised specimens than those created by electrosurgery. Abt et al10 point out that due to its non-contact cutting ability, CO2 laser surgery is faster than electrosurgery, which requires constant cleaning of the surgical instrument. In addition, the use of electrosurgery poses a significant risk or may even prove impossible in patients with metal orthodontic appliances.9
Radiofrequency surgery presents an advanced form of electrosurgery.39 It may also produce significant lateral heat to the surrounding tissues and should be avoided when in close proximity to bone40 and around pacemakers and other implantable or wearable medical devices. Accidental contact of a radiofrequency electrode to metal objects (such as implants, orthodontia, metal dental tools) may turn the objects into high-power radiofrequency antennae driven by the device’s own high-power radiofrequency energy sources (up to 100 W).
Electrocautery
The terms electrosurgery and electrocautery are often confused. Electrocautery, also known as thermal cautery, refers to a process in which a direct or alternating current is passed through a resistant metal wire electrode, generating heat. The heated electrode is then applied to living tissue to achieve hemostasis or varying degrees of tissue destruction.41 In electrocautery, unlike electrosurgery, the electric current does not pass through the patient; therefore, the technique can be safely applied with patients with pacemakers, ICDs, and deep-brain stimulators.42 Although electrocautery has been used to perform operculectomy, there is various literature6,43,44 advocating against it in dentistry because of the excessive thermal necrosis and scarring as its sequelae.
Soft-Tissue Diode/Non-Laser Thermal Ablation and Coagulation
Near-infrared laser light of circa 1,000 nm in diode devices does not ablate or cut the oral soft tissue optically. Instead, the laser light heats up the charred distal end of the fiberglass tip to 500°C to 900°C36 via heat conduction from the hot glass, and the tip then heats up the soft tissue. The contact with the hot charred glass tip causes the soft tissue to burn off (ablate), while coagulating the margins of the burn. Unlike non-contact surgical lasers (such as CO2 or Erbium), the ablative diodes are thermal contact, non-laser, wavelength-independent devices.
Conclusion
The 10,600 nm CO2 laser is a highly efficient and spatially accurate photo-thermal ablation device and an excellent coagulator due to the close match between coagulation depth and the diameters of oral soft-tissue capillaries.
The ablation of hyperplastic oral soft tissue with the flexible-fiber waveguide 10,600-nm CO2 laser is a minimally invasive, dependable surgical modality that yields predictable results. It often does not require sutures and typically ensures scar-free healing. It is, in many respects, superior to most of the alternative surgical treatment options. Its extreme accuracy in cutting, minimal thermal damage to the adjacent healthy tissue, bloodless operating field with excellent visibility and, reportedly, relatively minimal postoperative pain28,37 make the CO2 laser an optimal surgical solution to the removal of pericoronal gingival tissue. The ability of the CO2 laser to cut in a non-contact mode helps to reduce the risks of complications typically associated with outpatient treatment.28
Acknowledgments
The authors would like to thank Anna Glazkova, PhD, for her assistance and contribution in preparing this material for publication.
References
1. Olivi G, Margolis F, Genovese MD (eds). Pediatric Laser Dentistry: A User’s Guide. Hanover Park, IL: Quintessence Publishing; 2011:145-156.
2. Moloney J, Stassen LF. Pericoronitis: treatment and a clinical dilemma. J Ir Dent Assoc. 2009;55(4):190-192.
3. McNutt M, Patrick M, Shugars DA, et al. Impact of symptomatic pericoronitis on health-related quality of life. J Oral Maxillofac Surg. 2008;66:2482-2487.
4. Doran J. Operculectomy. http://www.exodontia.info/Operculectomy.html. Accessed September 20, 2014.
5. Namour S. Atlas of Current Oral Laser Surgery. Boca Raton, FL: Universal Publishers. 2011;139-171.
6. Kotlow L. Lasers in pediatric dentistry. In: Convissar RA. Principles and Practices of Laser Dentistry. St. Louis, MO: Mosby; 2011:202-222.
7. Chmura LG. Soft tissue lasers in orthodontics. In: Convissar RA. Principles and Practices of Laser Dentistry. St. Louis, MO: Mosby; 2011:225-242.
8. Strauss RA, Fallon SD. Lasers in contemporary oral and maxillofacial surgery. Dent Clin North Am. 2004;48(4):861-888.
9. Convissar RA, Diamond LB, Fazekas CD. Laser treatment of orthodontically induced gingival hyperplasia. Gen Dent. 1996;44(1):47-51.
10. Abt E, Wigdor H, Lobraico R, et al. Removal of benign intraoral masses using the CO2 laser. J Am Dent Assoc. 1987;115(5):729-731.
11. Ishida CE, Ramos-e-Silva M. Cryosurgery in oral lesions. Int J Dermatol. 1998;37(4):283-285.
12. Graham GF. Cryosurgery for benign, premalignant and malignant lesions. In: Wheeland RG, ed. Cutaneous Surgery. Philadelphia, PA: WB Saunders; 1994:835-869.
13. Faber WR. Side effects and complications in cryosurgery. Dermatol Monatsschr. 1993;179:247-251.
14. Pogrel MA, McCracken KJ, Daniels TE. Histologic evaluation of the width of soft tissue necrosis adjacent to carbon dioxide laser incisions. Oral Surg Oral Med Oral Pathol. 1990;70(5):564-568.
15. Deppe H, Horch HH. Current status of laser applications in oral and cranio-maxillofacial surgery. Med Laser Appl. 2007;22(1):39-42.
16. Coleton S. Lasers in surgical periodontics and oral medicine. Dent Clin North Am. 2004;48(4):937-962.
17. Convissar RA, Goldstein EE. An overview of lasers in dentistry. Gen Dent. 2003;51(5):436-440.
18. Gama SK, De Araujo TM, Pinheiro AL. Benefits of the use of the CO2 laser in orthodontics. Lasers Med Sci. 2008;23:459-465.
19. Kotlow LA. Lasers in pediatric dentistry. Dent Clin North Am. 2004;48(4):889-922.
20. Zaffe D, Vitale MC, Martignone A, et al. Morphological histochemical and immunocytochemical study of CO2 and Er:YAG laser effect on oral soft tissues. Photomed Laser Surg. 2004;22(3):185-189.
21. Zeinoun T, Nammour S, Dourov N, et al. Myofibroblasts in healing laser excision wounds. Lasers Surg Med. 2001;28:74-79.
22. Wang X, Ishizaki NT, Matsumoto K. Healing process of skin after CO2 laser ablation at low radiance: a comparison of continuous-wave and pulsed mode. Photomed Laser Surg. 2005;23:20-26.
23. Grbavac RA, Veeck EB, Bernard JP, et al. Effects of laser therapy in CO2 laser wounds in rats. Photomed Laser Surg. 2006;24(3):389-396.
24. de Freitas AC, Pinheiro AL, de Oliveira MG, Ramalho LM. Assessment of the behavior of myofibroblasts on scalpel and CO2 laser wounds: an immunohistochemical study in rats. J Clin Laser Med Surg. 2002;20(4):221-225.
25. Fisher SE, Frame JW, Browne RM, Tranter RMD. A comparative histological study of wound healing following CO2 laser and conventional surgical excision of canine buccal mucosa. Arch Oral Biol. 1983;28(4):287-291.
26. Basu MK, Frame JW, Rhys Evans PH. Wound healing following partial glossectomy using the CO2 laser, diathermy and scalpel: a histological study in rats. J Laryngol Otol. 1988;102(4):322-327.
27. Tambuwala A, Sangle A, Khan A, Sayed A. Excision of oral leukoplakia by CO2 lasers versus traditional scalpel: A comparative study. J Maxillofac Oral Surg. 2014;13(3):320-327.
28. Lambrecht JT, Stübinger S, Hodel Y. Treatment of intraoral hemangiomas with the CO2 laser. J Oral Laser Appl. 2004;4:89-96.
29. Wilder-Smith P, Arrastia AM, Liaw LH, Berns M. Incision properties and thermal effects of three CO2 lasers in soft tissue. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1995;79(6):685-691.
30. Jacques SL. Optical properties of biological tissues: a review. Phys Med Biol. 2013;58(11):R37-R61.
31. Fisher JC. Basic laser physics and interaction of laser light with soft tissue. In: Shapshay SM. ed. Endoscopic Laser Surgery Handbook. New York, NY: Marcel Dekker; 1987:96-125.
32. Fisher JC. Qualitative and quantitative tissue effects of light from important surgical lasers. In: Wright CV, Fisher JC, eds. Laser Surgery in Gynecology: A Clinical Guide. Philadelphia, PA: Saunders; 1993:58-81.
33. Vogel A, Venugopalan V. Mechanisms of pulsed laser ablation of biological tissues. Chem Rev. 2003;103(2):577-644.
34. Willems PWA, Vandertop WP, Verdaasdonk RM, et al. Contact laser-assisted neuroendoscopy can be performed safely by using pretreated ‘black’ fibre tips: Experimental data. Lasers in Surgery and Medicine. 2001;28(4):324-329.
35. Yoshida S, Noguchi K, Imura K, et al. A morphological study of the blood vessels associated with periodontal probing depth in human gingival tissue. Okajimas Folia Anat Jpn. 2011;88(3):103-109.
36. Romanos G. Diode laser soft-tissue surgery:advancements aimed at consistent cutting, improved clinical outcomes. Compend Cont Educ Dent. 2013;34(10):752-757.
37. Haytac MC, Ozcelik O. Evaluation of patient perceptions after frenectomy operations: a comparison of carbon dioxide laser and scalpel techniques. J Periodontol. 2006;77(11):1815-1819.
38. Tal H, Stahl S. Elimination of epithelium from healing postsurgical periodontal wounds by ultralow temperature: initial observations. J Periodontol. 1985;56(8):488-491.
39. Sumit S, Ramandeep SG, Simarpreet S, et al. Radiosurgery in dentistry: A brief review. Annals Dent Research. 2012;2(1):8-20.
40. Sherman JS. Advances in radiosurgery. Inside Dentistry. 2006;2(6):74-75.
41. Pollock SV. Electrosurgery. In: Bolognia JL, Jorizzo JL, Rapini RP (eds.). Dermatology. 2nd ed. Mosby Elsevier; 2008.
42. Weaver J, Kim SJ, Lee MH, Torres A. Cutaneous electrosurgery in a patient with a deep brain stimulator. Dermatol Surg. 1999;25(5):415-417.
43. Malone W. Electrosurgery in Dentistry. Springfield, IL; Charles C. Thomas Publisher: 1974.
44. No authors listed. Symposium in electrosurgery. Dent Clin North Am. 1982;26(4):681-921.
About the Authors
Robert Levine, DDS
Director of Laser Dentistry
Arizona School of Dentistry & Oral Health
Mesa, Arizona
Founder
Global Laser Oral Health LLC
Scottsdale, Arizona
Peter Vitruk, PhD, MInstP, CPhys
Founder
LightScalpel LLC
Woodinville, Washington
Faculty
California Implant Institute
San Diego, California
Faculty
Global Laser Oral Health LLC
Scottsdale, Arizona