You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
The regeneration of periodontal defects aims to improve the short- and long-term clinical outcomes of teeth with periodontal bone loss. True regeneration includes the formation of cementum, periodontal ligament, and alveolar bone.1 Significant periodontal pockets associated with deep intrabony defects are considered a clinical challenge and a chief cause of tooth loss for patients undergoing periodontal maintenance.2 Periodontal intrabony defects (also known as vertical or angular defects) are defined as anatomical sequelae of periodontitis progression, observed when the base of the defect is positioned apical to the interdental alveolar crest and surrounded by one, two, or three bony walls.3,4 The complex interaction of deep intrabony defects, associated loss of periodontal attachment, deep pocketing, and possible accompanying tooth mobility significantly decreases tooth prognosis.5 If the affected tooth is a molar, the treatment increases in complexity, with multiple intricacies added, such as furcation involvement, root trunk height, and accessibility for hygiene procedures. Hence, the regeneration of such defects can change tooth prognosis from uncertain to fair or favorable.6
Regenerative therapies can be used to gain clinical improvement beyond that which is achieved via flap surgery. Generally, the intent of regeneration procedures is twofold: regain a considerable portion of the lost attachment apparatus and reduce pocket depth. Ideally, this should be achieved without much increase in gingival recession.7 It is notable that different studies on regenerative approaches have shown great variability in clinical outcomes, typically attributed to systemic and local factors. Interestingly, a certain degree of variability of clinical outcomes, called the "center effect," may result from differences in the enrolled patient pool and/or the clinician's surgical ability, the efficacy of previous cause-related therapy, and supportive periodontal care programs.7-10
Many factors can affect the predictability of a regeneration procedure. For instance, smoking, the number of walls comprising the defect, tooth mobility, patient compliance, and defect width, depth, and morphology were all correlated with the outcomes of various regenerative approaches.7,8 Though technique-sensitive, periodontal regeneration seems to be efficient in treating intrabony defects, whether deep or shallow, wide or narrow.4 Achieving complete success in periodontal regeneration is challenging, which the authors believe may be attributed mainly to incomplete risk assessment, use of inadequate surgical approaches and materials, and insufficient clinical experience.10
The use of checklists in clinical practice can help clinicians avoid unexpected complications.11 For example, the authors have proposed a 10-key checklist for immediate implant placement at esthetic sites to achieve predictable outcomes.12 Checklist usage has been shown to help avoid complications in medicine, dentistry, and surgery.13 The purpose of this article is to describe seven essential keys to performing periodontal regeneration for intrabony defects with successful long-term outcomes (Table 1). This sequential, scientifically sound clinical approach can be used to help build and perform a strategy to optimize clinical outcomes in different patients with different defect anatomies.
Key No. 1: Periodontal Regeneration Risk Assessment for Intrabony Defects
The success of regenerative procedures in intrabony defects begins with a comprehensive approach to case selection. Based on previous decision trees,7,8,14 the authors propose a periodontal regeneration risk assessment to help clinicians identify patient-, tooth-, defect-, and operator-related factors that could influence the likelihood of achieving successful outcomes when performing regenerative procedures (Table 2). Based on the specific aspects observed in the risk assessment, the clinician should select the optimum treatment approach, including incision design, flap extension, and biomaterials, respecting the limitations of each case. This phase is critical to align the patient's expectations with treatment predictability. High-risk treatment sites should be thoroughly discussed with the patient.
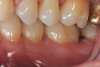
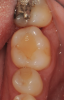
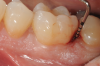
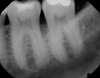
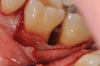
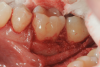
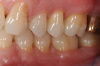
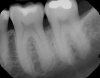
In the case presented, a 56-year-old healthy nonsmoking male patient, diagnosed with periodontitis stage III, localized, grade B, had been under care in a private practice periodontal office (RAL) for 25 years. He had a history of good compliance with his treatment (full-mouth plaque score <20%). During the COVID-19 pandemic, the patient missed three supportive periodontal therapy (SPT) visits over a year, resulting in periodontal breakdown interproximal between teeth Nos. 18 and 19 (mandibular left second and first molars, respectively). At his most recent periodontal maintenance visit, significantly increased periodontal probing depths of up to 10 mm with bleeding on probing were noted (Figure 1 through Figure 3). The periapical radiograph revealed a deep, narrow three-wall intrabony defect at the distal aspect of tooth No. 19, with class I buccal furcation involvement (Figure 4). According to the periodontal risk score (PRS),15 formerly known as the Miller-McEntire periodontal prognosis index, the tooth prognosis at the patient's initial examination was "good" (score = 5), taking into account that he was unaware of his hemoglobin A1C (HbA1c) levels. This scoring motivated the patient to be tested, and his follow-up HbA1c was <6%, thus reducing his PRS to 3, which was considered "excellent," as the PRS target goal for regenerative procedures is a score of <5.15
Tooth No. 19 presented with no mobility and tested vital endodontically. Soft-tissue anatomy was intact, with a keratinized tissue width of >2 mm. The operator was experienced, with no environmental stress and used checklists. Thus, based on the presurgical risk assessment, the patient presented a low-medium risk for periodontal regenerative therapy (Table 2).
Key No. 2: Surgical Preparation
Prior to a site-specific intrabony regenerative surgical procedure, sites with bleeding on probing should be managed with plaque control education and full-mouth periodontal scaling and root debridement.7 Scaling and root planing should only be performed supragingivally in the sites to be treated regeneratively, avoiding subgingival instrumentation in the planned surgical sites. Biofilm control evaluation and education should be reviewed with the patient. Teeth with Miller grade II or grade III mobility should be splinted and/or occlusally adjusted in all working, protrusive, and nonworking movements prior to surgical treatment.7 Vital teeth should be kept vital if the periodontal defect does not involve the apex. Existing root canal therapies should be carefully evaluated. Nonvital teeth must be successfully treated, and inadequate root canal treatments should be retreated with the aid of an endodontic evaluation and management.7,16
Any remaining bacterial deposits around the tooth in question should be removed on the day of the surgery. The adjunctive use of laser-assisted regeneration therapy can effectively sterilize the subgingival pocket area to the depth of the intrabony defect.17,18 Although controversial in the literature,19-21 antibiotics can be used by prescribing a loading dose (2 g of amoxicillin) to prevent postoperative infections and, thus, reduce postsurgical complications.
Key No. 3: Surgical Access
When planning for a regenerative procedure, several general requirements must be met for successful outcomes,22 including the creation of a space of sufficient volume to support a blood clot as well as stable, immobile soft-tissue protection of the treated area to avoid bacterial contamination.23,24 Optimal flap design to access intrabony defects must achieve passive primary closure. Wound stability is attained through proper soft-tissue management and adjacent teeth being stable (immobile).
Papilla preservation flaps are wide, mobile flaps to allow clear visibility of the defect area, easy application of biomaterials/barrier membranes, and passive primary closure of the flap while preserving interdental papilla.25-27 Several minimally invasive techniques utilizing the aid of operative microscopes and microsurgical instruments have been suggested. These techniques minimally elevate the tissues to reduce surgical trauma and increase flap stability. Therefore, the surgical approach should be chosen based on the defect morphology and extension. While minimally invasive techniques might provide better tissue stability, in some cases a large flap is necessary (eg, deep defects involving three or more sides of the root).7
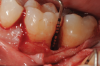
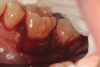
In the present case, intrasulcular incisions were performed, followed by a releasing incision in the mesial aspect of tooth No. 19. A full-thickness flap was elevated in the buccal and lingual aspects (Figure 5 and Figure 6) using a minimally invasive periosteal elevator. The site was carefully managed to avoid salivary contamination.
Key No. 4: Root Preparation and Conditioning
To increase the success rate of regenerative procedures, root preparation and modification are suggested.28 After flap elevation, the first step is the removal of the granulation tissues using hand, ultrasonic, and/or rotatory instruments. A high-speed handpiece with a surgical-length scaler bur or fine diamond tooth preparation bur under copious irrigation is used, and efficient debridement of the lesion is performed. The careful removal of all hard and soft deposits on the root surface and any clinically diseased cemental surface is essential for success. Although controversial, removing the smear layer and exposing collagen fibers through use of root conditioners, such as 24% ethylenediaminetetraacetic acid (EDTA) gel, might increase cell adhesion and present clinical benefits, including increased visualization and better hemostasis.29,30
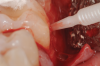
In the present case, the root of tooth No. 19 was prepared using piezoelectric scalers and nonfluted surgical-length burs to remove granulation tissues and any plaque/calculus on the root surface. Root conditioning was performed by applying 24% EDTA-containing gel for 2 minutes, followed by copious irrigation with sterile saline (Figure 7).
Key No. 5: Placement of Regenerative Biomaterials (Combined Therapy)
Tissue regeneration is based on three key components, often referred to as the "regeneration triad": cells, scaffolds (eg, bone grafts), and signaling molecules (eg, growth factors). Given sufficient vascularization, wound stability, and time, these components play an imperative role in tissue regeneration.31 Different systematic reviews have shown that combination therapies, ie, bone grafts, barrier membranes, and/or biological agents, should be employed for the treatment of residual pockets with deep (≥3 mm) intrabony defects.9,32,33
The use of biological agents, such as proteins or growth factors, is supported by a plethora of studies in the literature.4,7,9,34 The introduction of growth factors has marked a new era in periodontal regeneration.35 Enamel matrix derivative, for example, consists of a heterogeneous mixture of proteins containing amelogenins as a major component. Another well-studied, clinically and histologically proven cytokine for periodontal regeneration is recombinant human platelet-derived growth factor-BB (rhPDGF-BB).36-38 In combination with an osteoconductive scaffold, this growth factor was shown to significantly increase clinical attachment level gain, reduce gingival recession, and enhance bone fill and defect resolution compared to positive control. Human histologic studies showed evidence of the formation of new bone, cementum, and periodontal ligament when rhPDGF-BB was used with any type of bone graft, including mineralized freeze-dried bone allograft (FDBA), demineralized FDBA, tricalcium phosphate, or deproteinized bovine bone mineral in either intrabony or furcation defects.39,40 Finally, a recent systematic review showed that the efficacy of rhPDGF-BB in combination with bone grafts was not associated with any adverse effects.41
The addition of barrier membranes is fundamental in large and non-contained defects to keep the bone graft in place. Considering the higher postoperative morbidity and incidence of complications when using nonresorbable barrier membranes,7,9 resorbable membranes should be the first choice, as they will combine with a bone graft and act as a scaffold.42-45 Non-crosslinked collagen membranes are recommended.46,47
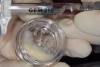
In the present case, rhPDGF-BB was used in combination with FBDA and firmly packed into the intrabony defect (Figure 8 and Figure 9). An amnion-chorion membrane was customized and adapted over the bone defect (Figure 10 and Figure 11).
Key No. 6: Primary Closure of the Flap
Successful regeneration requires tension-free primary flap closure over the entire surgical site.48,49 As previously mentioned, papilla preservation flaps and microsurgical techniques help improve esthetics while maintaining the primary closure of the flap and enhancing the ability to create space for regeneration in the interdental area.7,14,25,26 Exposure of barrier membranes is the most common complication in regenerative procedures; thus, flap management is pivotal in preventing unsuccessful outcomes.7,9,50
Another factor that may enable primary closure to be maintained is adherence to minimally invasive principles. In a recent systematic review, microsurgical techniques were found to significantly improve periodontal plastic surgical outcomes and postsurgical recovery.51 Therefore, periosteum-releasing incisions should be performed to achieve a tension-free flap closure. Additionally, due to a wide papilla dimension, a horizontal mattress combined with interrupted sutures can support better flap stability. Recommended types of sutures include nonresorbable, high-density polytetrafluoroethylene (d-PTFE), or slowly resorbable, which should be left in for at least 4 weeks unless they become loose, in which case they should be removed earlier.
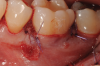
In the present case, stable and tension-free closure of the flap was achieved with the use of periosteum-releasing incisions and sutures. The 5-0 slowly resorbable sutures were left in for 4 weeks (Figure 12).
Key No. 7: Postoperative Protocol
Postsurgically, the effective control of supragingival and subgingival biofilm performed by mechanical means (eg, hand instruments and/or ultrasonic debridement), air-polishing devices, or other chemical means (eg, antiseptic) has been shown to be a chief component of successful regenerative treatment, especially in deeper periodontal pockets.52 Treatment results may be maintained long-term only with regular SPT.53 Highly comprehensive mechanical and antiseptic regimens were typically reported in regenerative therapy studies. Although recall duration and frequency varied widely throughout the regenerative studies, by and large they recommended a recall interval of two or more times every month in the first 3 months. In fact, some studies suggested bringing patients in for recall every week, at least during the first months after surgery.27,54
In the present case, the patient's postoperative protocol included the administration of analgesics and mouthrinse. Plaque control and polishing were performed every 2 to 3 weeks for the first 3 months. The patient was then placed on a 3-month recall system. At the 1-year follow-up, stability of the soft and hard tissues could be observed (Figure 13 and Figure 14). Following the seven-key checklist, this clinical case demonstrated that correct case selection, with consideration given to patient-, tooth-, defect-, and operator-related factors, combined with the use of an evidence-based surgical protocol, led to predictable outcomes.
Conclusion
Considering the increase in life expectancy and high incidence of peri-implant diseases, especially in periodontal patients, interventions to save teeth and increase their prognosis, such as regenerative procedures, must be performed with high predictability. The "7 keys" checklist for successful periodontal regeneration of intrabony defects is intended to be a guide for clinicians in the treatment planning and execution of regenerative procedures. Each key is essential in achieving a predictable long-term outcome.
Acknowledgment
The authors express their gratitude to Andrea Ravidà, DDS, MS, for his valuable help as a consultant.
About the Authors
Robert A. Levine, DDS
Clinical Professor, Periodontology and Implantology, Kornberg School of Dentistry, Temple University, Philadelphia, Pennsylvania; Private Practice, Pennsylvania Center for Dental Implants and Periodontics, Philadelphia, Pennsylvania
Debora R. Dias, DDS, MSc
PhD Candidate, State University of Maringá, Maringá, Paraná, Brazil
Muhammad H.A. Saleh, BDS, MS, MSD
Clinical Assistant Professor, Periodontics and Oral Medicine, University of Michigan School of Dentistry, Ann Arbor, Michigan
Jeffrey Ganeles, DMD
Adjunct Associate Professor, Nova Southeastern University College of Dental Medicine, Fort Lauderdale, Florida; Private Practice in Periodontics and Implant Dentistry, Boca Raton, Florida
Maurício G. Araújo, DDS, MSc, PhD
Associate Professor, State University of Maringá, Maringá, Paraná, Brazil; Private Practice in Dental Implants and Periodontics, Rio de Janeiro, Rio de Janeiro; Brazil
Queries to the author regarding this course may be submitted to authorqueries@broadcastmed.com.
References
1. Bosshardt DD, Stadlinger B, Terheyden H. Cell-to-cell communication-periodontal regeneration. Clin Oral Implants Res. 2015;26(3):229-239.
2. Matuliene G, Pjetursson BE, Salvi GE, et al. Influence of residual pockets on progression of periodontitis and tooth loss: results after 11 years of maintenance. J Clin Periodontol. 2008;35(8):685-695.
3. Lang NP. Focus on intrabony defects-conservative therapy. Periodontol 2000. 2000;22:51-58.
4. Nibali L, Sultan D, Arena C, et al. Periodontal infrabony defects: systematic review of healing by defect morphology following regenerative surgery. J Clin Periodontol. 2021;48(1):100-113.
5. Lang NP, Tonetti MS. Periodontal diagnosis in treated periodontitis. Why, when and how to use clinical parameters. J Clin Periodontol. 1996;23(3 Pt 2):240-250.
6. Saleh MHA, Dukka H, Troiano G, et al. External validation and comparison of the predictive performance of 10 different tooth-level prognostic systems. J Clin Periodontol. 2021;48(11):1421-1429.
7. Cortellini P, Tonetti MS. Clinical concepts for regenerative therapy in intrabony defects. Periodontol 2000. 2015;68(1):282-307.
8. Kao RT, Nares S, Reynolds MA. Periodontal regeneration - intrabony defects: a systematic review from the AAP Regeneration Workshop. J Periodontol. 2015;86(2 suppl):S77-S104.
9. Nibali L, Koidou VP, Nieri M, et al. Regenerative surgery versus access flap for the treatment of intra-bony periodontal defects: a systematic review and meta-analysis. J Clin Periodontol. 2020;47 suppl 22:320-351.
10. Cortellini P, Tonetti MS. Focus on intrabony defects: guided tissue regeneration. Periodontol 2000. 2000;22:104-132.
11. Renouard F, Amalberti R, Renouard E. Are "human factors" the primary cause of complications in the field of implant dentistry? Int J Oral Maxillofac Implants. 2017;32(2):e55-e61.
12. Levine RA, Ganeles J, Gonzaga L, et al. 10 keys for successful esthetic-zone single immediate implants. Compend Contin Educ Dent. 2017;38(4):248-260.
13. Renouard F, Renouard E, Hernanz M. Situational awareness: a method for analysis and prevention of complications in medicine and dentistry. Rev Periodoncia Clin. 2018;10:100-111.
14. Reynolds MA, Kao RT, Nares S, et al. Periodontal regeneration - intrabony defects: practical applications from the AAP Regeneration Workshop. Clin Adv Periodontics. 2015;5(1):21-29.
15. Levine RA, Miller PD, Dias DR, et al. Translating clinical outcomes to patient value through use of the periodontal risk score: an evidence-based treatment approach. Compend Contin Educ Dent. 2023;44(1):18-24.
16. Cortellini P, Stalpers G, Mollo A, Tonetti MS. Periodontal regeneration versus extraction and prosthetic replacement of teeth severely compromised by attachment loss to the apex: 5-year results of an ongoing randomized clinical trial. J Clin Periodontol. 2011;38(10):915-924.
17. McCawley TK, McCawley MN, Rams TE. Immediate effects of laser-assisted new attachment procedure (LANAP) on human periodontitis microbiota. J Int Acad Periodontol. 2018;20(4):163-171.
18. Aoki A, Mizutani K, Schwarz F, et al. Periodontal and peri-implant wound healing following laser therapy. Periodontol 2000. 2015;68(1):217-269.
19. Nibali L, Buti J, Barbato L, et al. Adjunctive effect of systemic antibiotics in regenerative/reconstructive periodontal surgery-a systematic review with meta-analysis. Antibiotics (Basel). 2021;11(1):8.
20. Hai JH, Lee C, Kapila YL, et al. Antibiotic prescribing practices in periodontal surgeries with and without bone grafting. J Periodontol. 2020;91(4):508-515.
21. Pietruska M, Dolińska E, Milewski R, Sculean A. Effect of systemic antibiotics on the outcomes of regenerative periodontal surgery in intrabony defects: a randomized, controlled, clinical study. Clin Oral Investig. 2021;25(5):2959-2968.
22. Lynch SE, Marx RE, Nevins M, Wisner-Lynch LA, eds. Tissue Engineering: Applications in Oral and Maxillofacial Surgery and Periodontics. 2nd ed. Chicago, IL: Quintessence Publishing; 2008.
23. Sanz M, Tonetti MS, Zabalegui I, et al. Treatment of intrabony defects with enamel matrix proteins or barrier membranes: results from a multicenter practice-based clinical trial. J Periodontol. 2004;75(5):726-733.
24. De Sanctis M, Zucchelli G, Clauser C. Bacterial colonization of bioabsorbable barrier material and periodontal regeneration. J Periodontol. 1996;67(11):1193-1200.
25. Pagni G, Tavelli L, Rasperini G. The evolution of surgical techniques and biomaterials for periodontal regeneration. Dent Clin North Am. 2022;
66(1):75-85.
26. Cortellini P, Prato GP, Tonetti MS. The modified papilla preservation technique. A new surgical approach for interproximal regenerative procedures. J Periodontol. 1995;66(4):261-266.
27. Cortellini P, Bowers GM. Periodontal regeneration of intrabony defects: an evidence-based treatment approach. Int J Periodontics Restorative Dent. 1995;15(2):128-145.
28. Rosen PS, Reynolds MA, Bowers GM. The treatment of intrabony defects with bone grafts. Periodontol 2000. 2000;22:88-103.
29. Belal MH, Watanabe H, Ichinose S, Ishikawa I. Effect of PDGF-BB combined with EDTA gel on adhesion and proliferation to the root surface. Odontology. 2012;100(2):206-214.
30. Blomlöf J, Blomlöf L, Lindskog S. Effect of different concentrations of EDTA on smear removal and collagen exposure in periodontitis-affected root surfaces. J Clin Periodontol. 1997;24(8):534-537.
31. Langer R. Tissue engineering: perspectives, challenges, and future directions. Tissue Eng. 2007;13(1):1-2.
32. Stavropoulos A, Bertl K, Spineli LM, et al. Medium- and long-term clinical benefits of periodontal regenerative/reconstructive procedures in intrabony defects: systematic review and network meta-analysis of randomized controlled clinical studies. J Clin Periodontol. 2021;48(3):410-430.
33. Sculean A, Nikolidakis D, Schwarz F. Regeneration of periodontal tissues: combinations of barrier membranes and grafting materials - biological foundation and preclinical evidence: a systematic review. J Clin Periodontol. 2008;35(8 suppl):106-116.
34. Sculean A, Nikolidakis D, Nikou G, et al. Biomaterials for promoting periodontal regeneration in human intrabony defects: a systematic review. Periodontol 2000. 2015;68(1):182-216.
35. Lynch SE, Colvin RB, Antoniades HN. Growth factors in wound healing. Single and synergistic effects on partial thickness porcine skin wounds. J Clin Invest. 1989;84(2):640-646.
36. Lynch SE, Nixon JC, Colvin RB, Antoniades HN. Role of platelet-derived growth factor in wound healing: synergistic effects with other growth factors. Proc Natl Acad Sci U S A. 1987;84(21):7696-7700.
37. Nevins M, Kao RT, McGuire MK, et al. Platelet-derived growth factor promotes periodontal regeneration in localized osseous defects: 36-month extension results from a randomized, controlled, double-masked clinical trial. J Periodontol. 2013;84(4):456-464.
38. Nevins M, Giannobile WV, McGuire MK, et al. Platelet-derived growth factor stimulates bone fill and rate of attachment level gain: results of a large multicenter randomized controlled trial. J Periodontol. 2005;76(12):2205-2215.
39. Camelo M, Nevins ML, Schenk RK, et al. Periodontal regeneration in human Class II furcations using purified recombinant human platelet-derived growth factor-BB (rhPDGF-BB) with bone allograft. Int J Periodontics Restorative Dent. 2003;23(3):213-225.
40. Thakare K, Deo V. Randomized controlled clinical study of rhPDGF-BB + β-TCP versus HA + β-TCP for the treatment of infrabony periodontal defects: clinical and radiographic results. Int J Periodontics Restorative Dent. 2012;32(6):689-696.
41. Tavelli L, Ravidà A, Barootchi S, et al. Recombinant human platelet-derived growth factor: a systematic review of clinical findings in oral regenerative procedures. JDR Clin Trans Res. 2021;6(2):161-173.
42. Stavropoulos A, Karring T. Guided tissue regeneration combined with a deproteinized bovine bone mineral (Bio-Oss) in the treatment of intrabony periodontal defects: 6-year results from a randomized-controlled clinical trial. J Clin Periodontol. 2010;37(2):200-210.
43. Gulameabasse S, Gindraux F, Catros S, et al. Chorion and amnion/chorion membranes in oral and periodontal surgery: a systematic review. J Biomed Mater Res B Appl Biomater. 2021;109(8):1216-1229.
44. Rosen PS, Froum SJ, Cohen DW. Consecutive case series using a composite allograft containing mesenchymal cells with an amnion-chorion barrier to treat mandibular class III/IV furcations. Int J Periodontics Restorative Dent. 2015;35(4):453-460.
45. Sbricoli L, Guazzo R, Annunziata M, et al. Selection of collagen membranes for bone regeneration: a literature review. Materials (Basel). 2020;13(3):786.
46. Jiménez Garcia J, Berghezan S, Caramês JMM, et al. Effect of cross-linked vs non-cross-linked collagen membranes on bone: a systematic review. J Periodontal Res. 2017;52(6):955-964.
47. Tal H, Kozlovsky A, Artzi Z, et al. Long-term bio-degradation of cross-linked and non-cross-linked collagen barriers in human guided bone regeneration. Clin Oral Implants Res. 2008;19(3):295-302.
48. Greenstein G, Greenstein B, Cavallaro J, et al. Flap advancement: practical techniques to attain tension-free primary closure. J Periodontol. 2009;80(1):4-15.
49. Ronda M, Stacchi C. Management of a coronally advanced lingual flap in regenerative osseous surgery: a case series introducing a novel technique. Int J Periodontics Restorative Dent. 2011;31(5):505-513.
50. Mayfield L, Söderholm G, Hallström H, et al. Guided tissue regeneration for the treatment of intraosseous defects using a biabsorbable membrane. A controlled clinical study. J Clin Periodontol. 1998;25(7):585-595.
51. Di Gianfilippo R, Wang IC, Steigmann L, et al. Efficacy of microsurgery and comparison to macrosurgery for gingival recession treatment: a systematic review with meta-analysis. Clin Oral Investig. 2021;25(7):4269-4280.
52. Gartenmann SJ, Dörig I, Sahrmann P, et al. Influence of different post-interventional maintenance concepts on periodontal outcomes: an evaluation of three systematic reviews. BMC Oral Health. 2016;17(1):19.
54. Zucchelli G, Bernardi F, Montebugnoli L, De SM. Enamel matrix proteins and guided tissue regeneration with titanium-reinforced expanded polytetrafluoroethylene membranes in the treatment of infrabony defects: a comparative controlled clinical trial. J Periodontol. 2002;73(1):3-12.