You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Characterized by progressive destruction of tooth-supporting apparatuses, periodontitis is a chronic, multifactorial inflammatory disease that can lead to tooth loss and disability, thus negatively affecting masticatory function, esthetics, and quality of life.1 Periodontitis can also be a contributing aggravating factor in systemic disease progression and has been reported to possibly have a bidirectional cause-effect relationship.2 Periodontitis produces a wide range of adverse signs and symptoms. Evaluation of the severity and complexity of periodontal disease involves assessment of clinical and radiographic parameters such as bleeding on probing (BOP), probing depth (PD), clinical attachment level, mobility, furcation involvement, and radiographic bone loss.
Various methods have been suggested for determining prognosis of periodontally involved teeth. For a prognosis index to be routinely used clinically, it needs to be not only accurate but also simple and quick to use chairside, requiring minimal time to complete with the patient. In addition, an effective prognosis index should be meaningful to patients, providing them with motivation to improve their periodontal health.
The periodontal risk score (PRS) employs a simple scoring system that can be used in daily clinical practice to determine periodontal prognosis. Rather than relying solely on clinical indicators for oral health status and treatment-needs evaluation, the PRS also includes systemic and lifestyle parameters as prognostic factors to help determine tooth prognosis (Figure 1).3,4 Recognized risk factors that were recently incorporated into the periodontitis classification, including smoking and diabetes, are taken into consideration in this prognosis system.1 The PRS is used for patients either with periodontitis or a healthy periodontium and yields a numerical score for a patient's periodontal prognosis at 15 and 30 years. The PRS, with the "target goal" of a <5 score, translates clinical outcomes of treatment into patient value, helping patients understand their long-term periodontal prognosis.4-6 This approach is similar to the way a physician might discuss goals with a patient for smoking cessation, managing hypertension, or improving glycemic control. In this manner, the PRS serves as a simple, effective, evidence-based, statistically validated, and accurate motivational tool.3,4,7-11 The aim of this article, therefore, is to present to clinicians effective ways to implement this tool into daily practice.
Objectives and Applications of the PRS
There are four main objectives of using the PRS.4 The first is to motivate the patient to accept and complete treatment while heightening the patient's awareness of the importance of complying with periodontal maintenance, the main aspects of which are defined in this system as the "keys to success" (described in the next section of this article).4,12-14 The second objective is to encourage patients to make lifestyle changes to improve their overall periodontal and systemic health. This may include smoking cessation, better controlling blood sugar levels,4,8,9,15,16 and complying with the ongoing periodontal maintenance phase of treatment (ie, supportive periodontal therapy [SPT]).17,18
The third objective, obtained through the PRS's simplified scoring system, is to empower all clinical team members to present the PRS to the patient to initiate a conversation on treatment and on reaching the target goal of <5. In this regard, the authors consider dental professionals as "the physicians of the mouth."19 (To help train staff on scoring patients, the authors recommend reviewing the Levine-Miller MMPPI webinar20 in a scheduled team meeting.)
The fourth objective of using the PRS is to encourage the patient to directly refer family and friends to the practice.
Keys to Success
The "keys to success" comprise crucial aspects of periodontal maintenance. Discussing these keys with the patient and emphasizing their importance in helping the patient achieve the target goal of a PRS of <5 is an essential part of the initial examination, as this discussion provides the patient with a specific goal to strive for and the means for maintaining it. Following these keys will enable patients to succeed in their periodontal maintenance; however, the keys themselves are not a promise of success. Rather, they serve as a guideline to facilitate the patient's success, and the patient needs to accept his or her role as, in essence, a "co-therapist" and assume the responsibility to diligently adhere to the keys.
The following are the five "keys to success": brush, floss, and clean teeth and tongue daily; complete recommended treatment; adhere to the recommended maintenance schedule; control blood sugar (if the patient has diabetes) with a goal of <6% hemoglobin A1c (HbA1c); quit smoking (if the patient is a smoker).
It should be noted that most of the bacteria left after brushing and interdental cleaning are on the posterior third of the tongue.21,22 These bacteria cannot be removed with a toothbrush without causing the patient to gag. To remove them, a metal tongue scraper is required. (The authors suggest visiting https://pdmillerswebtextbook.com/ to view online videos on tongue-cleaning technique and disinfecting the mouth.19)
Based on Miller et al's study, patients should never lose a tooth to periodontal disease if they follow all five "keys to success."3 However, additional cofactors may need to be discussed and possibly addressed with the patient. These include tooth-related issues associated with medication-induced xerostomia (MIX), which can result in increased susceptibility to caries, especially root caries. This is particularly important today (post-COVID), as patients are using more medications that are associated with MIX than in 2014 when the original study data was collected and published by Miller et al.3,23 In addition, parafunctional habits need to be controlled with the use of a nightguard appliance when indicated.
Even patients with a healthy periodontium should be encouraged to follow the "keys to success," as they are integral to not only achieving but also maintaining overall oral health. Also, if a previously periodontally treated patient is found to have a higher PRS (ie, a worsened prognosis) at a post-treatment periodontal maintenance visit, the five keys should be re-reviewed to ascertain which aspect(s) of periodontal maintenance the patient may need to better address. An elevated PRS might also prompt a discussion about possibly increasing the periodontal maintenance frequency, the possible need for site-specific retreatment, or treatment planning modifications.
PRS Scoring
Formerly known as the Miller-McEntire periodontal prognosis index (MMPPI), the PRS was initially intended to score the prognostic factors specifically for molar teeth. Today the authors use it more broadly and emphasize that even though the PRS scores a molar tooth, the test reflects on the periodontal prognosis of the whole mouth. The authors suggest scoring the most periodontally involved molar tooth (ie, the "worst" molar) that is planned to be maintained. In this system, scores range from 0 to 11, with the target goal being a PRS of <5, as mentioned earlier. Miller et al statistically showed that patients with a score of <5 should never lose a tooth to periodontal disease.3 Thus, with a score between 1 and 4 the tooth is considered to have an "excellent" prognosis; a score between 5 and 7 equals a "good" prognosis; and a score of 8 or higher equates to a "guarded" prognosis.7 If the patient follows the recommended treatment plan, the score post-treatment should markedly improve, which can be highly motivational to the patient.
The PRS assigns scores to seven prognostic factors, including recognized risk factors for periodontal disease, such as smoking and diabetes mellitus.2,4 A patient's smoking status, whether light, heavy, or a previous smoker, may play a role in tooth prognosis.24,25 For patients with previously diagnosed or suspected diabetes mellitus, HbA1c values need to be evaluated for scoring. Patients who have not been diagnosed with diabetes mellitus or are not aware of their HbA1c level, or have a history of diabetes in the family, should be referred to a physician for necessary blood work.4,26
To reiterate, the PRS is based on evaluation of the most periodontally involved, or "worst," molar that the clinician is planning to keep, but the score reflects on the whole mouth and not just the tooth that is scored. Use of the PRS is intended to reduce inflammatory periodontal-systemic interrelationships to improve not just the patient's periodontal health but also the patient's systemic health. The seven patient factors that are scored in the PRS (Figure 1) are as follows:
Age. Age has a minimal effect on long-term periodontal prognosis. Nonetheless, the patient's age is classified as follows: score 0 = age 1 to 39 years; score 1 = age ≥40 years.
Smoking. Smoking is the most significant negative factor in determining periodontal prognosis. Smoking increases the chances of losing teeth to periodontal disease by 246%.3 This factor is scored based on the patient's smoking status: score 0= nonsmoker or former smoker (≥3 months); score 4= smoker (determined statistically through the Cox hazard ratio3).
Note that the overall objective of this test is to reach a PRS below 5, at which point the patient should not lose a tooth to periodontal disease.3 For example, if a patient who is a smoker achieves a PRS of 5, the patient will have a 93% chance of maintaining his or her teeth for 15 years and an 80% chance of doing so for 30 years (Figure 1).4
HbA1c levels. The authors suggest that if patients do not know their HbA1c score, it may be scored as "2" until their blood work results are received. This is particularly important when patients have a history of diabetes in their family or have not seen a physician in more than 1 or 2 years. Using the PRS thus can prompt patients to obtain their HbA1c score and, if they are diabetic, motivate them to better control their diabetes, which, in turn, will have a positive influence on their periodontal status.27 HbA1c can be easily tested at a local pharmacy.
HbA1c levels are scored as follows: score 0 = <6%; score 1 = 6.1% to 7%; score 2 = 7.1% to 8%; score 3 = 8.1% to 9%; score 4 = >9%.
Molar type. The most periodontally involved, or "worst," maxillary second molar (or maxillary first molar if the second molar is absent) that is planned to be maintained should be used for scoring, as this tooth scores the highest of the molar group (ie, score = 2) and has the poorest prognosis in the periodontal literature.13,28 If no molars are present, the next-"worst" single-rooted tooth or implant should be scored. For example, if a dental implant in a maxillary first molar site is being scored, it should be assigned a score of 1, the same score that would be assigned to a maxillary first molar tooth.
Molar types are scored as follows: score 0 = mandibular molar; score 1 = maxillary first molar; score 2 = maxillary second molar.
Probing depth. For this factor, the deepest PD in millimeters (mm) of the "worst" molar being maintained should be used. The scoring is as follows: score 0 = <5 mm; score 1 = 5 mm to 7 mm; score 2 = >7 mm to 10 mm; score 3 = >10 mm.
Furcation involvement. The following scores are assigned based on the number of furcations present in the molar that is being scored: score 0 = none; score 1 = one total furcation (regardless of whether class I or class II); score 2 = two total furcations; score 3 = three total furcations or T-T (through and through) furcation.
This index does not consider the class or severity of furcations, ie, class I, class II, or class III; it uses only the number of furcations associated with the molar tooth being scored. Based on the multivariable Cox proportional hazards model performed byMiller et al, furcation classification has a minimal effect on the final PRS, as it increases the chances of a patient losing teeth to periodontal disease by only 21%.3 Thus, treatment of a furcated molar tooth remains a viable option to retain the tooth in the long term.29
Mobility. Mobility of the molar to be scored is scored as follows: score 0 = none; score 1 = class I; score 2 = class II; score 3 = class III (tooth is depressible).
It is important to evaluate tooth mobility as part of the initial examination. Moreover, mobility values can be positively modified by occlusal adjustment.30
Final Score and Prognosis
The total PRS value is the sum of each of the individual scores of the seven prognostic factors. The post-treatment target goal is a PRS of <5. Scoring and prognoses are as follows: score 1 to 4 = "excellent" prognosis; score 5 to 7 = "good" prognosis; score 8 to ≥11 = "guarded" prognosis (Figure 1).
Case Report
In 2014, a male patient presented to the periodontal practice of one of the authors (RAL). He reported that he wanted to save his teeth while avoiding the suggested full-mouth resective osseous surgery performed by his general dentist 7 years prior, which resulted in the patient experiencing significant postoperative morbidity. The patient's dentist was recommending retreatment in the same manner, but the patient, extremely dissatisfied with the previous treatment, desired an alternative therapy to conventional periodontal surgery. After researching other periodontal treatments on the internet, he found the author's periodontal practice and inquired about laser-assisted new attachment procedure (LANAP®) therapy.31
The patient was 66 years old (age: ≥40 = score 1) and a former smoker who had quit 7 years prior (smoking: nonsmoker = score 0). He was generally healthy, with an American Society of Anesthesiologists (ASA) III status and a HbA1C of <6% (A1C levels: <6% = score 0). Medically the patient presented with medication-controlled hypertension and a history of a stroke and myocardium infarction. He was taking multiple oral medications and presented with MIX (ie, dry mouth).
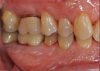
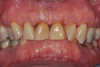
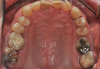
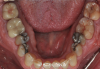
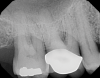
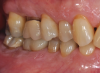
Periodontal examination revealed generalized BOP and PDs up to 9 mm in the maxillary and mandibular molars with multiple furcations in each molar (Figure 2 through Figure 7). He presented with class I mobility in many posterior teeth and class II mobility in teeth Nos. 3 and 9 (maxillary right first molar and maxillary left central incisor, respectively). The periodontist decided to score tooth No. 3 for the PRS, as this was the most periodontally involved molar that was planned to be treated and maintained (Figure 2 and Figure 8). This tooth (maxillary right first molar = score 1) presented with probing depths of 7 mm (score 1); three total furcation invasions (score 3) (mesial [degree II furcation], buccal [degree I furcation], and distal [degree II furcation]); and a class II mobility (score 2). The total PRS for tooth No. 3 was 8, representing a "guarded" prognosis. Based on this score, the likelihood of not losing any teeth to periodontal disease for 15 years was 81%, and for 30 years the likelihood was just 56% (Figure 9).7
The patient's PRS of 8 was discussed with him, and the recommended treatment plan involved oral hygiene (for which he received instructions), full-mouth single-visit LANAP surgical therapy under local anesthesia, with full-mouth occlusal adjustment in maximum intercuspation (MIP) and all working, non-working, and protrusive movements. After monthly postoperative visits with full-mouth polish, review of plaque control procedures, and review of the "keys to success" for periodontal maintenance, the patient was re-evaluated at 3 months and commenced the periodontal maintenance phase of his treatment (ie, SPT) under the periodontist's care every 3 months due to his advanced periodontal disease status with multiple molar furcation invasions, along with annual restorative examinations and vertical bitewing radiographs at his general dentist's practice.
The periodontist felt that teeth Nos. 17 and 18 (mandibular left third and second molars, respectively) had a questionable prognosis due to severe periodontal attachment loss, class III furcations, and probable furcation caries as detected on radiographs. These two teeth were treated the same as the others with the patient being advised that they may need either retreatment or extraction in the future with dental implants possibly being recommended.
During follow-up periodontal maintenance appointments over time, the patient's PRS for tooth No. 3 was reduced to a score of 5, a "good" prognosis with a 93% chance of periodontally preserving his teeth after 15 years and an 80% chance of doing so after 30 years (Figure 9). The furcation involvement score was reduced with the buccal furcation of tooth No. 3 non-probable (score = 1). The tooth mobility score of No. 3 was reduced by 1 (score = 1). The patient remained in periodontal maintenance every 3 months until 2018 with excellent compliance.17 Review of his PRS at each visit reinforced the positive results of treatment and the importance of his compliance to the "keys to success." His PRS for tooth No. 3 remained stable (score 5).
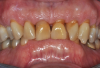
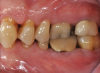
In 2018, however, the patient noticed significantly increased bleeding and tenderness associated with his non-scored mandibular left molars (Nos. 17 and 18), which he then elected to retreat with laser-assisted periodontal therapy. This retreatment was successful in eliminating his symptoms and resulted in periodontal stability (Figure 10 through Figure 13).
The patient was motivated by the continued discussions of his PRS and 15-year prognosis, and determined to maintain excellent compliance to the "keys to success." His maintenance plan included the use of a water flosser, flossing, and diluted oral sodium hypochlorite (NaOCl) bleach mouthrinse (dilution ratio of a half teaspoon of NaOCl to 8 ounces of warm water), which was used in his water irrigator device. After 4 years of therapy and maintenance, his PRS score was reduced from 8 to 5, thus lowering his periodontal risk and demonstrating the effectiveness of the PRS strategy.
Discussion
The PRS uses seven prognostic factors to formulate a final score. The test is simple to use, takes little time, and is evidence-based, statistically validated, and motivational.3,4,7-11 It has a superior predictive capability because of the high emphasis placed on systemic factors (age, smoking, diabetic status), with almost half of the total score based on the scoring of these factors (potentially 9 points).3,4 Moreover, compared with other indexes, the PRS is the only one that incorporates HbA1c levels.10 In a recent study published by Saleh et al, the authors compared 10 tooth-level prognostic systems, including the PRS, to predict periodontal-related tooth loss.10 The study examined 148 patients with periodontitis (3,787 teeth), with a follow-up period of 26.5 ± 7.4 years. All 10 systems accurately stratified teeth based on the risk of tooth loss due to periodontitis (TLP) at baseline when different classes of association were compared. Furthermore, most of the classes of all the prognostic systems showed statistically significant inter-class differences; the more severe the classes, the more risk of TLP. The authors concluded that the PRS may have shown the best discrimination and predictive capability for TLP.
Conclusion
The PRS is essentially a score sheet, or periodontal report card, that may be used in daily clinical practice for all patients. Its use is not limited to patients presenting with periodontitis, as it can and should be routinely used with periodontally healthy patients as well. The PRS can help the clinician determine an appropriate treatment plan, with the patient understanding that he or she needs to be a "co-therapist" in order to reach and maintain the target goal of a PRS of <5. The PRS can motivate patients to make positive lifestyle changes, which will improve their total score and, ultimately, the periodontal prognosis of their whole mouth. The PRS scoring takes only a few minutes chairside with the patient and enables the clinician to have a conversation that can "translate" recommended clinical procedures to "patient value." The PRS empowers patients to combat periodontitis and, ultimately, preserve their teeth.
About the Authors
Robert A. Levine, DDS
Clinical Professor, Periodontology and Implantology, Kornberg School of Dentistry, Temple University, Philadelphia, Pennsylvania; Private Practice, Pennsylvania Center for Dental Implants and Periodontics, Philadelphia, Pennsylvania
Preston D. Miller, DDS
Former Clinical Professor of Periodontics, University of South Carolina, Charleston, South Carolina
Debora Reis Dias, DDS, MSc
PhD Candidate, State University of Maringá, Maringá, Paraná, Brazil
Muhammad H.A. Saleh, BDS, MS, MSD
Adjunct Clinical Assistant Professor, Periodontics and Oral Medicine, University of Michigan School of Dentistry, Ann Arbor, Michigan
Dharmarajan Gopalakrishnan, BDS, MDS, PhD
Dean, Professor and Head (Chair), Department of Periodontology, Dr. D.Y. Patil Dental College and Hospital, Dr. D.Y. Patil Vidyapeeth, Pune, Maharashtra, India
Gustavo Fernandes, DDS, MSc, PhD
Adjunct Clinical Assistant Professor, Periodontics and Oral Medicine, University of Michigan School of Dentistry, Ann Arbor, Michigan
HimaBindu Dukka, BDS, MSD, MPH
Associate Professor, Department of Diagnosis and Oral Health; Adjunct Faculty, Department of Oral Immunology and Infectious Diseases; Director, Graduate Periodontics, University of Louisville School of Dentistry, Louisville, Kentucky
Kellyann Ehrmann, RDH
Private Practice, Pennsylvania Center for Dental Implants and Periodontics, Philadelphia, Pennsylvania
Philip L. Fava II, DMD, MDSc
Private Practice, Pennsylvania Center for Dental Implants and Periodontics, Philadelphia, Pennsylvania
Maurício G. Araújo, DDS, MSc, PhD
Associate Professor, State University of Maringá, Maringá, Paraná, Brazil; Private Practice in Dental Implants and Periodontics, Rio de Janeiro, Rio de Janeiro, Brazil
Queries to the author regarding this course may be submitted to authorqueries@broadcastmed.com.
References
1. Papapanou PN, Sanz M, Buduneli N, et al. Periodontitis: consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri-Implant Diseases and Conditions. J Periodontol. 2018;89 suppl 1:S173-S182.
2. Tonetti MS, Greenwell H, Kornman KS. Staging and grading of periodontitis: framework and proposal of a new classification and case definition. J Periodontol. 2018;89 suppl 1:S159-S172.
3. Miller PD Jr, McEntire ML, Marlow NM, Gellin RG. An evidenced-based scoring index to determine the periodontal prognosis on molars. J Periodontol. 2014;85(2):214-225.
4. Levine RA, Miller PD. The Miller McEntire Periodontal Prognostic Index (i.e., "The Perio Report Card") usage in practice. Adv Periodontal Surg. 2020;3-21. doi:10.1007/978-3-030-12310-9_1.
5. Levine RA, Shanaman RH. Translating clinical outcomes to patient value: an evidence-based treatment approach. Int J Periodontics Restorative Dent. 1995;15(2):186-200.
6. Sonick M. Treating People Not Patients. Harper Collins Publishers; 2022. https://www.peoplenotpatients.org/.
7. Gopalakrishnan D, Miller PD, Levine RA, et al. A 5-year prospective validation of the Miller-McEntire Periodontal Prognostic Index. J Indian Soc Periodontol. 2022;26(2):157-161.
8. Gopalakrishnan D, Miller PD, Mahuli AV, et al. Prospective evaluation of periodontally diseased molars in smokers using the Miller-McEntire Periodontal Prognostic Index. J Indian Soc Periodontol. 2018;22(4):304-309.
9. Gopalakrishnan D, Miller PD, Mahuli AV, et al. Evaluation of periodontally diseased molars in diabetics with Miller-McEntire Periodontal Prognostic Index. J Indian Soc Periodontol. 2018;22(5):401-405.
10. Saleh MHA, Dukka H, Troiano G, et al. External validation and comparison of the predictive performance of 10 different tooth-level prognostic systems. J Clin Periodontol. 2021;48(11):1421-1429.
11. Saleh MHA, Tattan M, Troiano G, et al. The periodontal risk score (PRS): initiation and model validation for 6762 teeth. J Periodontol. 2022. doi: 10.1002/JPER.22-0273.
12. Goldman MJ, Ross IF, Goteiner D. Effect of periodontal therapy on patients maintained for 15 years or longer. A retrospective study. J Periodontol. 1986;57(6):347-353.
13. Hirschfeld L, Wasserman B. A long-term survey of tooth loss in 600 treated periodontal patients. J Periodontol. 1978;49(5):225-237.
14. McFall WT Jr. Tooth loss in 100 treated patients with periodontal disease. A long-term study. J Periodontol. 1982;53(9):539-549.
15. Johnson GK, Guthmiller JM. The impact of cigarette smoking on periodontal disease and treatment. Periodontol 2000. 2007;44:178-194.
16. Patel RA, Wilson RF, Palmer RM. The effect of smoking on periodontal bone regeneration: a systematic review and meta-analysis. J Periodontol. 2012;83(2):143-155.
17. Levine RA, Wilson TG. Compliance as a major risk factor in periodontal disease progression. Compendium. 1992;13(12):1072-1076.
18. Echeverría JJ, Echeverría A, Caffesse RG. Adherence to supportive periodontal treatment. Periodontol 2000. 2019;79(1):200-209.
19. Miller PD. PD Miller's web textbook. https://pdmillerswebtextbook.com/. The Importance of Disinfecting the Mouth; The Miller McEntire Periodontal Prognostic Index. Accessed November 14, 2022.
20. Levine RA, Miller PD. The Use of the MMPPI in Clinical Practice: Benefits in Achieving Long-Term Periodontal and Systemic Health for Our Patients webinar. August 2018. https://register.gotowebinar.com/register/4584978205109305103. Accessed November 14, 2022.
21. Van der Sleen MI, Slot DE, Van Trijffel E, et al. Effectiveness of mechanical tongue cleaning on breath odour and tongue coating: a systematic review. Int J Dent Hyg. 2010;8(4):258-268.
22. Quirynen M, Avontroodt P, Soers C, et al. Impact of tongue cleansers on microbial load and taste. J Clin Periodontol. 2004;31(7):506-510.
23. Thomson WM, Ferguson CA, Janssens BE, et al. Xerostomia and polypharmacy among dependent older New Zealanders: a national survey. Age Ageing. 2021;50(1):248-251.
24. Fiorini T, Musskopf ML, Oppermann RV, Susin C. Is there a positive effect of smoking cessation on periodontal health? A systematic review. J Periodontol. 2014;85(1):83-91.
25. Ravidà A, Troiano G, Qazi M, et al. Dose-dependent effect of smoking and smoking cessation on periodontitis-related tooth loss during 10-47 years periodontal maintenance - a retrospective study in compliant cohort. J Clin Periodontol. 2020;47(9):1132-1143.
26. Weinspach K, Staufenbiel I, Memenga-Nicksch S, et al. Level of information about the relationship between diabetes mellitus and periodontitis - results from a nationwide diabetes information program. Eur J Med Res. 2013;18(1):6.
27. Polak D, Shapira L. An update on the evidence for pathogenic mechanisms that may link periodontitis and diabetes. J Clin Periodontol. 2018;45(2):150-166.
28. Helal O, Göstemeyer G, Krois J, et al. Predictors for tooth loss in periodontitis patients: systematic review and meta-analysis. J Clin Periodontol. 2019;46(7):699-712.
29. Nibali L, Shemie M, Li G, et al. Periodontal furcation lesions: a survey of diagnosis and management by general dental practitioners. J Clin Periodontol. 2021;48(11):1441-1448.
30. Hallmon WW, Harrel SK. Occlusal analysis, diagnosis and management in the practice of periodontics. Periodontol 2000. 2004;34:151-164.
31. Richman C, Levine RA, Sullivan A Jr, et al. Perceptions of the LANAP protocol. Decisions in Dentistry. 2020;6(10):18-23.