You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
Over the last two decades, countless discoveries have been made in medical science demonstrating dental healthcare providers' ability to reduce noncommunicable health risks, enhance patients' long-term systemic health outcomes, and lessen the 154-billion-dollar financial burden of periodontal disease in the United States.1However, there remains a crucial need for advances in preventive dental care, as approximately 42% of American adults present with some form of periodontal disease,2 while 22% have untreated dental caries.3 In restorative dentistry, new techniques and treatment workflows are continually emerging to foster minimally invasive care and improve clinical outcomes based on scientific evidence, while also enhancing the patient experience and increasing the longevity, function, and esthetics of the restorations. For dental prophylaxis, however, conventional ultrasonic cleaning, hand-scaling, and paste polishing remain the industry gold standard. Like restorative dentistry, dental hygiene needs to adopt new evidence-based treatment protocols and technologies to improve patients' dental preventive care.
Transforming the Focus from Calculus to Biofilm
Evidence has demonstrated that patient oral healthcare should be individualized.4 The advent of the 16S rRNA identification tool and a repository of oral bacterial genome sequences have advanced our knowledge of microbial science and evolved to enhance our understanding of the oral-systemic connection.4 It is important that dental practitioners understand the complexity of the oral microbiome and the potential shift from oral microbial symbiosis to dysbiosis.
Dental plaque is an archetypical biofilm composed of a complex microbial community. The microbial ecology, when balanced, maintains a harmonious relationship between the biofilm and the adjacent tissues.5 A mature biofilm is polymicrobial, consisting mainly of bacteria, but can also contain protozoa, viruses, and fungi.6 The polymicrobial communities can present with healthy, cariogenic, or periodontopathogenic profiles, and disease activation is multifactorial, including the host response and modifiable risk factors such as nutrition, salivary pH, the airway, and self-care product selection.7 The periodontal pathogens in the biofilm are responsible for the inflammatory cascade that breaks down the gingival epithelial barrier and provides entry into the bloodstream.8Periodontal pathogens invade the circulatory system and, dependent upon genetic factors, can increase risk factors to activate more than 50 non-communicable systemic diseases.
Therefore, a paradigm shift is needed in the dental hygiene process to include comprehensive biofilm management before calculus removal. Ensuring that biofilm management is prioritized at professional dental hygiene treatments will aid in restoring a symbiotic oral microbiome, while dental hygienists should also reinforce proper home care techniques and recommend self-care products and nutrition to enhance commensal bacterial species that promote oral health.
This paradigm shift will not only improve and maintain patients' oral health but can also benefit their systemic health. In a recent multi-year study by the Cigna Dental Team that evaluated the impact of dental care on medical costs, it was found that patients who receive routine comprehensive preventive and periodontal care have fewer emergency room visits and reduced overall medical expenditures.9
What's Trending in Dental Hygiene
The conventional approach to dental hygiene care in most US dental practices focuses primarily on treatment rather than on assessments that aid in the development of an individualized treatment plan. In oral medicine practice, comprehensive medical assessments, blood pressure screenings, and extraoral/intraoral screenings should be routinely done to provide individualized patient care. Comprehensive oral health assessments and data collection must also be performed. A complete mouth series of radiographs or panoramic radiographs should be updated every 3 to 5 years depending on the patient's dentition and disease risk factors. Comprehensive periodontal screening and salivary and bacterial biofilm assessments should be completed and the patient's periodontal stage and grade determined. Documentation with 3D scanning and intraoral photography also need to be obtained to assist in patient education about his or her dental condition. However, because of the amount of time that dental hygienists must devote to providing treatment with ultrasonic scalers, hand instrumentation, and paste polishing, the time available within the appointment to provide these essential assessments is limited; dental hygiene professionals who attempt to achieve this higher standard of oral health care for their patients while providing conventional therapies tend to run behind schedule, and many also experience work-related musculoskeletal disorders, quickly leading to burnout. In addition, the dental hygiene shortage precipitated by the COVID-19 pandemic has negatively impacted patient access to dental care. The higher wages due to high demand have reduced the margins on hygiene services.10 Furthermore, in the author's conversations, the mass exodus of dental hygienists during the COVID pandemic resulted in part from career-long frustration due to a lack of autonomy, overbooking, and lack of employee benefits.
In these dire circumstances, advances in oral healthcare prevention and a paradigm shift in the culture of dental practices are needed to provide solutions for the trending issues that dental practices nationwide are experiencing.
Reimagining the Dental Hygienist as a Mid-Level Oral Medicine Practitioner
While it is true that some dental hygienists are content to provide the same treatment to all patients without practicing individualized care, the ranks of dental hygienists also include many high-functioning practitioners who properly assess and educate patients and recommend therapies based on the individual patient's oral condition.
Furthermore, dental hygienists have extensive periodontal training. It is therefore incumbent upon the dental healthcare community to foster a culture that supports dental hygienists' potential to serve as mid-level oral medicine practitioners who can provide patients with the comprehensive oral health assessments that are necessary for individualized care. Evidence-based protocols and technologies to enhance the productivity and scheduling autonomy of dental hygienists must be part of the solution for supporting their role as oral health practitioners.
Collecting the Data and Presenting the Evidence
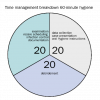
Using the 20-20-20 minute module, the 60-minute dental hygiene appointment can be broken down as follows: 20 minutes for data collection, case presentation, and oral hygiene instructions; 20 minutes for debridement; and 20 minutes for examination, appointment scheduling, and infection control (Figure 1). For the dental hygienist who is employing conventional instrumentation for cleaning, it is difficult to increase the time spent on data collection with this model, especially when faced with more complex cases.
Clinicians may neglect to update full mouth series (FMS), panoramic radiographs, or periodontal probe readings because these tasks are time-consuming. One of the most serious consequences of improper data collection is failure to diagnose periodontal disease. In documented instances of such supervised neglect involving failure to diagnose or treat periodontal disease promptly, in most cases radiographs were not taken routinely and periodontal assessment was rarely or never recorded.11
A simple way to assess the "health" of your hygiene department is to run the practice's periodontal percentage in the practice management program. Alternatively, software can be used to perform a data analysis of the dental practice with a focus directly on the hygiene department and each dental hygiene practitioner. These programs can highlight where production can be increased and alert the dentists, dental hygiene department, and team of any outstanding treatment plans and which patients are due for radiographs. In addition, these programs quickly provide each dental hygienist's key performance indicators (KPIs) and offer insight into where additional training and support may be needed. Some dental hygienists may decide to continue their education to understand the connection between the inflammatory cascade of both gingivitis and periodontitis, and some may also choose to learn communication techniques such as motivational interviewing, which can aid in communicating risk factors and proposed treatment to patients.
Artificial intelligence (AI) is one new technology that is being used to eliminate practitioner bias, as technology is thereby allowed to confirm diagnostics. AI can assist dental providers in standardizing the diagnosis of periodontitis, and it has been speculated that it may increase case acceptance for some patients.12
Another technology, voice-activated periodontal charting, allows patients to receive education about and play an active role in their periodontal assessment. This technology communicates to the patient what defines oral health, oral inflammation, and oral disease and the parameters for assessment before the periodontal screening, thus serving as an educational tool. Once the periodontal assessment is complete, the data on the percentages of pocket depths, clinical attachment loss (CAL), and bleeding points are used to assist in determining the stage and grade of the patient's periodontal disease.
After the comprehensive periodontal assessment, the clinician can determine if salivary diagnostics would benefit the case outcomes. Periodontal disease is both a disease and an infection, and salivary diagnostics provide biomarkers that can be used to direct precision periodontal individualized care. Combining salivary biomarkers and clinical parameters have been shown to result in better predicted scaling and root planing outcomes than either approach used alone.13 Additionally, salivary diagnostics will also help in determining the systemic risk factors associated with the pathogens identified in the periodontal test report.
Advances in Dental Hygiene Therapy
The conventional approach to dental prophylaxis can be perceived as uncomfortable, laborious, and antiquated for both the practitioner and the patient. New workflows have been developed for biofilm management that have greatly improved the dental hygiene experience for both patients and practitioners.
With such a workflow, the first 20 minutes of the hygiene appointment is used to assess patients with comprehensive data collection, to use disclosing solutions to record the patient's plaque scores, and to provide oral hygiene instructions. Using disclosing solutions with each patient allows the patient education and treatment time to be tailored to fit the individual patient's needs; simply applying biofilm disclosing solution on the hard and soft tissues, rinsing, and reviewing the results will tremendously impact the dental hygiene visit. Biofilm disclosing allows the patient and practitioner to visualize the volume, acidity, and maturity of bacteria in their mouth (ie, the etiology of their oral disease), thus improving the patient's dental literacy. This in turn increases patient motivation to improve their oral hygiene through oral self-care and regular dental prophylaxis appointments.14 Furthermore, biofilm disclosing provides visual confirmation of comprehensive biofilm removal and a streamlined approach to managing biofilm and immature calculus.15Oral hygiene instructions are reviewed and prescribed while biofilm is visible, aiding in patient comprehension of the specific products, techniques, and frequency customized to improve their oral health condition. Restating the connection that pathogenic biofilm has on systemic health during the motivation phase of the workflow for biofilm management can potentially improve patient compliance.
The next 20 minutes of the hygiene appointment are dedicated to debridement. Conventional hygiene requires three passes around every surface of every tooth, first with an ultrasonic cleaner to remove calculus and biofilm, followed by hand instrumentation of every surface of every tooth, and lastly followed by a final pass circumferentially around every surface of every tooth with past polishing. Conventional treatment is limited to biofilm disruption and calculus removal on the non-shedding surfaces of the oral cavity. In contrast, the new workflows utilize erythritol powder airpolishing (EPAP), which has low abrasiveness to preserve biological tissues, making it safe to apply both supragingivally and subgingivally, yet highly efficient in disrupting biofilm on both hard and soft tissues. 16
Shifting the paradigm in the dental hygiene process to manage biofilm before calculus removal utilizing warm water, air, and minimally invasive erythritol powder improves efficiency and significantly reduces subgingival bacterial counts.17 Efficiency is also achieved by removing both biofilm and stain simultaneously and disrupting biofilms on both hard and soft tissues while maintaining the natural integrity of tooth structure.18
After a comprehensive assessment of the pocket depth, clinical attachment level, tissue biotype, and radiographs, the subgingival application of EPAP can be determined. Research shows that with the adjunctive application of subgingival EPAP, significantly more initially deep pockets (PPD ≥ 5.5 mm) were reduced to shallow (PPD ≤ 3.4 mm), and a considerable attachment gain was observed.19
Once a biofilm has been removed from both a supragingival and subgingival aspect and it has been confirmed by the provider by visual inspection that there is no disclosed biofilm, the remaining calculus deposits are removed by instrumentation. This approach preserves hard and soft tissue by instrumenting only where calculus remains.
With such a workflow, the final 20 minutes of the hygiene appointment are dedicated to a quality control pass through the mouth with a universal instrument, and an exploratory stroke can be applied to ensure that all hard and soft deposits have been removed. The hygienist should observe if any potential carious lesions may not have been detected before treatment, and should apply a fluoride or biocompatible hydroxyapatite paste to act as a pellicle layer until the natural pellicle reforms. If indicated, adjunctive therapies such as laser bacterial reduction, sealant placement, probiotic treatment, ozone application, or whitening treatment can be performed during this step. These adjunctive therapies offer increased production potential. The dentist will perform the examination and review any potential restorative needs. Lastly, the next recall appointment is scheduled based on the patient's biofilm load, oral hygiene self-care ability, and the medical risk factors evaluated in step one.
Conclusion
The paradigm shift to remove biofilm with minimally invasive powders such as EPAP before removing calculus can produce time savings for dental hygienists to devote to oral health assessments and to apply many other technologies to enhance the patient's oral health experience. These advances also impact the quality of care the dental hygienist provides and encourage hygienists to practice at the top of their scope as mid-level health practitioners. It is the opinion of this author that empowering your dental hygienists with autonomy and technology can potentially resolve the national dental hygienist shortage.
References
1. Botelho J, Machado V, Leira Y, Proença L, Chambrone L, Mendes JJ. Economic burden of periodontitis in the United States and Europe: an updated estimation. J Periodontol. 2022;93(3):373-379.
2. Eke PI, Dye BA, Wei L, et al. Prevalence of periodontitis in adults in the United States: 2009 and 2010. J Dent Res. 2012;91(10):914-920.
3. Bashir NZ. Update on the prevalence of untreated caries in the US adult population, 2017-2020. J Am Dent Assoc. 2022;153(4):300-308.
4. Wade WG. The oral microbiome in health and disease. Pharmacol Res. 2013;69(1):137-143.
5. Seneviratne CJ, Zhang CF, Samaranayake LP. Dental plaque biofilm in oral health and disease. Chin J Dent Res. 2011;14(2):87-94.
6. Larsen T, Fiehn N-E. Dental biofilm infections - an update. APMIS. 2017;125(4):376-384.
7. Zarco MF, Vess TJ, Ginsburg GS. The oral microbiome in health and disease and the potential impact on personalized dental medicine. Oral Dis. 2012;18(2):109-120.
8. Shrivastava D, Natoli V, Srivastava KC, et al. Novel approach to dental biofilm management through guided biofilm therapy (GBT): a review. Microorganisms. 2021;9(9):1966.
9. Cigna. Preventive dental treatment associated with lower medical utilization and costs. National Cigna study presented at the International Association for Dental Research Meeting; March 2015; Boston, MA.
10. Economic outlook and emerging issues in dentistry. https://www.ada.org/resources/research/health-policy-institute/economic-outlook-and-emerging-issues
11. Kierce EA, Kolts RJ. Improving periodontal disease management with artificial intelligence. Compend Contin Educ Dent.2023;44(6):e1-e4.
12. Baxter C. A review of dental negligence. Dentistry IQ website. https://www.dentistryiq.com/dentistry/endodontics/article/16351341/a-review-of-dental-negligence. Published September 1, 2004. Accessed July 31, 2023.
13. Y Liu, Duan D, Ma R, et al. The combined use of salivary biomarkers and clinical parameters to predict the outcome of scaling and root planing: a cohort study. J Clin Periodontol. 2020;47(11):1379-1390.
14. Huang J, Yao Y, Jiang J, Li C. Effects of motivational methods on oral hygiene of orthodontic patients: a systematic review and meta-analysis. Medicine (Baltimore). 2018;97(47):e13182.
15. Mensi M, Scotti E, Sordillo A, Agosti R, Calza S. Plaque disclosing agent as a guide for professional biofilm removal: a randomized controlled clinical trial. Int J Dent Hyg. 2020;18(3):285-294.
16. de Cock P. Erythritol functional roles in oral-systemic health. Adv Dent Res. 2018;29(1):104-109.
17. Petersilka G, Tunkel J, Barakos K, et al. Subgingival plaque removal at interdental sites using a low-abrasive air polishing powder. J Periodontol. 2003;74(3):307-311
18. Camboni S, Donnet M. Tooth surface comparison after air polishing and rubber cup: a scanning electron microscopy study. J Clin Dent. 2016;27(1):13-18.
19. Divnic-Resnik T, Pradhan H, Spahr A. The efficacy of the adjunct use of subgingival air-polishing therapy with erythritol powder compared to conventional debridement alone during initial non-surgical periodontal therapy. J Clin Periodontol. 2022;49(6):547-555.