You must be signed in to read the rest of this article.
Registration on CDEWorld is free. Sign up today!
Forgot your password? Click Here!
The terms ceramic and porcelain are often used interchangeably in dentistry. Ceramic comes from the Greek term keramos and means potter, referring to one’s ability to heat clay to form pottery.1 The word porcelain is said to have been invented by Marco Polo in the 13th century from the Italian word porcellana, or cowrie shell. Polo used the cowrie shell to describe Chinese porcelain because it was similarly strong and hard while remaining thin and translucent. A ceramic is a compound of metallic elements (eg, aluminum, calcium, lithium, magnesium, potassium, sodium, tin, titanium, zirconium) and nonmetallic elements (eg, silicon, fluorine, boron, oxygen), whereas porcelain is a ceramic consisting of a glass matrix phase and one or more crystalline phases (eg, leucite). All porcelains are ceramics, but not all ceramics are porcelains. For example, an all-zirconia crown is referred to as a high-strength ceramic but it does not have a glass matrix; therefore, it is not a porcelain. For the purposes of this article, the term ceramic is used to include all metal-free restorations.
The use of ceramics in dentistry dates back to Charles H. Land. In 1889, he patented the all-porcelain crown called the “jacket” crown.2 A tooth was restored by placing a porcelain covering or “jacket” around it. Improvements were made to the material and it was used quite extensively up to the 1950s. Internal microcracking during cooling was a major problem with this porcelain system. To reduce this microcracking risk, Dr. Abraham Weinstein developed the porcelain-fused-to-metal crown (PFM) in the late 1950s.3 In 1965, there was a resurgence of all-ceramic restorations with the addition of industrial aluminous porcelain (> 50%) to the feldspathic porcelain. McLean and Hughes developed a new version of the porcelain jacket crown with an inner core of aluminous porcelain that contained 40% to 50% alumina crystals.4 Despite having twice the strength of a PFM, this bilayered all-ceramic restoration was only used in the anterior region because of its lower strength. Its higher opacity was also a major drawback.5
Some dental ceramic systems have come full circle with the elimination of bilayered, veneered copings and the “rebirth” of the monolithic all-ceramic restoration. Others have vastly improved the esthetics and strength. The ceramics used in dentistry today have evolved into complicated systems that can be difficult to grasp. Classifying ceramic materials may help dentists and dental technicians better understand the different compositions, how they are processed, and where they should be used.
Classifications of Dental Ceramics
Dental ceramics can be classified in a number of different ways, including by their composition, processing method, fusing temperature, microstructure, translucency, fracture resistance,
and abrasiveness.1
Generally, all-ceramic restorations have been confined to the anterior region until recently with the introduction of monolithic lithium dioxide and zirconia restorations. These types of restorations have no limit in terms of where they can be used in the dental arch. All other ceramic systems (when used in a monolithic form) should be limited from the canine forward because of the lower flexural strength. These same ceramics can be used in the posterior regions only when supported by a high-strength core (metal or ceramic).
Classification by Composition
Ceramics can be divided into three categories by composition2: ceramics that are predominantly composed of glass, those made of particle-filled glass,1 and those consisting of polycrystalline.6
Ceramics that are composed mostly of glass have the highest esthetics. Manufacturers some-times add small amounts of filler particles to control the optical effects that mimic natural enamel and dentin. Generally, the more filler particles that are added to a ceramic, the greater the increase in the mechanical properties but the greater the decrease in its esthetic properties. Polycrystalline ceramics contain no glass at all. As noted earlier, these are not porcelains. The crystalline arrangement lends these ceramic materials the highest strength, but they are generally less esthetic. The principle is similar to tooth-colored filling material (composite resins). By definition, any material that comprises different materials is a composite; therefore, a ceramic is also a composite. With composite resins, filler particles are added to a resin matrix; greater filler content means greater mechanical properties, but results in lower translucency. With ceramics, the glass is the matrix and the fillers are crystalline particles that melt at high temperatures. Nonglass-containing polycrystalline ceramics comprise an aluminum oxide or zirconium oxide matrix and fillers that are not particles but elements that alter optical properties. These added elements are referred to as dopants.6
Conventional dental ceramics are based on a silica (SiO2) network and potash feldspar (K2O-Al2O3-6SiO2), soda feldspar (Na2O-Al2O3-6SiO2), or both.7 To control the coefficient of thermal expansion, solubility, and fusing and sintering temperatures, different elements are added, such as pigments (to produce the different hues), opacifiers (white-colored oxide to decrease translucency), and glasses.
Classification by Processing Method
Another approach to classifying ceramics is by the method by which they are processed. This includes powder/liquid building, slip casting, hot-ceramic pressing, and additive and subtractive computer-aided design/computer-aided manufacturing (CAD/CAM).
Powder/Liquid Building
Mixing ceramic powder and liquid (ie, deionized water or the manufacturer’s modeling liquid) is a conventional processing method. This condensation method incorporates building on a ceramic or metal core with a powder/liquid ceramic slurry with a brush or spatula by hand. The slurry is condensed by vibration to remove excess liquid, which rises to the surface and is blotted away by an absorbent tissue. It is important to remove any voids during the application, but this does not always occur. Depending on the skill of the technician, some voids may remain, decreasing the overall strength of the restoration. At certain steps in the fabrication, the ceramic buildup is vacuum fired at a selected temperature, which removes the moisture and further condenses the ceramic through a process called “sintering.” During the sintering process, fusion occurs at the particles’ points of contact, which results in densification by viscous flow when the ceramic or glass particles reach their firing temperature.8 Typically, a restoration is overcontoured by 25% to allow for densification or shrinkage during the firing cycle.
Slip-Casting
The slip-casting fabrication method was introduced in the 1990s. This processing technique involves the creation of a porous core by slip casting, which is sintered and then infiltrated with a lanthanum-based glass, producing two interpenetrating continuous networks: a glassy phase and a crystalline infrastructure. The crystalline infrastructure could be alumina (Al2O3), spinel (MgAl2O4), or zirconia-alumina (12 Ce-TZP-Al2O3).9 Restorations produced through this method tend to have fewer defects from processing and have greater strength than conventional feldspathic porcelain.10
Hot-Pressed Ceramic
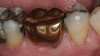
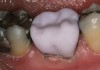
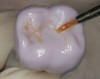
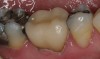
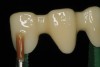
The hot-pressed ceramic fabrication technique was introduced in the late 1980s and allowed the dental technician to create the restoration in wax. Then, using the lost-wax technique, the technician was able to press a plasticized ceramic ingot into a heated investment mold. Ceramics containing high amounts of leucite glass or optimal pressable ceramics were initially used for this process.10 In 2006, lithium disilicate became the second generation of materials to use this method.11 A commonly used technique involves waxing the restoration to full contour and then hot pressing to yield a restoration (Figure 1). The incisal area is then cut back to create mamelons (Figure 2). This is followed by the application of various incisal porcelains. To account for shrinkage (densification) during the firing cycle, the layering porcelain is overcontoured (Figure 3 through Figure 7).
CAD/CAM
In the mid 1990s, Nobel Biocare introduced the first all-ceramic product with a CAD/CAM substructure. The core consisted of 99.9% alumina on which a feldspathic ceramic was layered.11
The use of CAD/CAM technology expanded machinable ceramic fabrication by allowing scanning, designing, and milling of either a full-contoured restoration or a single- or multiple-unit framework by a computer.12 Two different CAD/CAM methods are used. The first method is an additive version in which an electrodeposition of powdered material is applied layer by layer to a conductive die through an electrical current.13 This technique is also referred to as rapid prototyping. The other (and more common) method is a subtractive method in which a substructure or full-contour restoration is milled from a solid block of ceramic material. The available materials for the subtractive CAD/CAM processing include silica-based ceramics, infiltration ceramics, lithium-disilicate ceramics, and oxide high-performance ceramics.14 For example, lithium disilicate is actually milled as lithium metasilicate and then heated to 820°C in a two-stage oven. During this firing cycle, there is a controlled growth of the grain size (0.5 μm to 5 μm) and a conversion of metasilicate crystals to disilicate crystals. This crystallization process not only changes the physical composition and strength but also causes the restoration to reach the indicated ceramic shade (Figure 8 through Figure 11).15
Classification by Fusing Temperature
Dental porcelains are classified by their firing temperatures. The categories are described as high-fusing (1,300°C), medium-fusing (1,101°C to 1,300°C), low-fusing (850°C to 1,100°C), and ultra-low-fusing (< 850°C).7 Denture teeth are an example of high-fusing porcelain. Crown and bridge porcelains can be either medium- or low-fusing, depending on the system, and ultra-low-fusing porcelain would be used for porcelains and glazes. To make it less complicated, some dental professionals now refer to just two categories, high-fusing porcelains or low-fusing porcelains, with the separation designated at 800°C.16
Classification by Microstructure
As previously mentioned, porcelains have two different phases: the glass phase (responsible for the esthetics) and the crystalline phase (associated with mechanical strength). In the case of feldspathic porcelain, a crystalline mineral called leucite (potassium-aluminum-silicate) forms when feldspar is melted. Between 1,150°C and 1,530°C, feldspar undergoes incongruent melting to form leucite crystals. Incongruent melting is a process in which one material does not uniformly melt and forms a different material.7 The leucite crystalline phase has a diffraction index similar to the glassy matrix that, in this case, contributes to the overall esthetics of the porcelain.17 The leucite content of a porcelain is associated with the crack propagation strength. Greater leucite content means a greater decrease in the propagation of a crack.18 This type of porcelain is referred to as leucite-reinforced. During the sintering process of all-ceramic restorations, microporosities are formed on the surface that lead to crack initiation and propagation, ultimately resulting in failure.19-21
Hot-pressed ceramics have high amounts of leucite crystals and are considered leucite-reinforced glass ceramics. During the heated injection molding cycle, the sintering process is avoided22 and the leucite crystals act as barriers that counteract the increase in tensile stresses that can lead to the formation of microcracks.21,23 This type of ceramic can be used to press as an all-ceramic restoration or to a metal coping (Figure 12 through Figure 15).
As previously mentioned, McLean and Hughes developed an all-ceramic crown that had an inner core of aluminous porcelain that contained 40% to 50% alumina crystals.4 The principle behind this addition was the dispersion of a high-strength crystal with a high-elastic modulus within the glassy matrix to increase the strength and hardness of the ceramic.24 Alumina increases the strength of feldspathic porcelain more than leucite, which increases the fracture resistance.25 The particle size of the alumina may be responsible for the increase in the mechanical properties by decreasing agglomeration.26 When ceramics are sintered, the particle size is critical. Finer powder yields a greater reduction in surface area. Fine powders tend to form clusters of irregular shape and uncontrolled size and are referred to as “agglomerates,” which hinder flow properties.27
As previously stated, lithium disilicate was the second generation of hot-pressed ceramic materials. These ceramic restorations are referred to as lithium-disilicate—reinforced glass ceramics. This ceramic material contains 70% lithium-disilicate crystals, which results in an increased flexural strength of approximately 360 MPa (milled version) to 400 MPa (hot-pressed version).28
The increase in strength is found in the unique microstructure of lithium disilicate, which consists of any small interlocking platelike crystals that are randomly oriented. The lithium-disilicate crystals cause cracks to deflect, branch, or blunt, which arrests the propagation of cracks.29
Zirconia as a pure oxide does not occur in nature. It has been given the nickname “ceramic steel,” and the scientific term is zirconia dioxide. This biomaterial is widely used in medicine and dentistry because of its mechanical strength as well as its chemical and dimensional stability and elastic modulus similar to stainless steel.30 Zirconia has a normal density of 6 g/cm2. The theoretical density (ie, 100% dense) of zirconium oxide is 6.51 g/cm2. The closer these two density values are, the less space between the particles, resulting in greater strength and a smoother surface.31
A unique characteristic of zirconia is its ability to stop crack growth, which is termed “transformation toughening” (Figure 16). An ensuing crack generates tensile stresses that induce a change from a tetragonal configuration to a monoclinic configuration and a localized volume increase of 3% to 5% (Figure 17). This volume increase results in a change of tensile stresses to compressive stresses generated around the tip of the crack. The compressive forces counter the external tensile forces and stop the further advancement of the crack.32-34 This characteristic accounts for the material’s low susceptibility to stress fatigue and high flexural strength of 900 MPa to 1200 MPa.35,36 Zirconia dioxide can be used as a monolithic restoration or a substructure with a veneering porcelain (Figure 18).
Classification by Translucency
Translucency is the relative amount of light transmitted through a material.37 A natural tooth derives most of its color as a result of the light reflectance from dentin that is altered by absorption and scattering by the enamel.38 The shade of a human tooth is determined by the shade of the dentin because the enamel is more translucent. This translucency becomes more apparent in the interproximal and incisal portions of the tooth because of the lack of underlying dentin.
There are several factors that affect the translucency of dental ceramics. Thickness of the material has the greatest effect,39,40 but translucency can also be affected by the number of firings,41 the shade of the substrate,42 and the type of light source or illuminant.43 Because clinical settings can vary so widely, specimens should be compared at the recommended minimum thickness to be classified by translucency.40
Porcelain translucency is usually measured with the translucency parameter, which is defined as the color difference between a uniform thickness of ceramic material over a black and a white background44 or the contrast ratio (CR), which is the ratio of illuminance of a ceramic material when it is placed over a black background compared with a white background.45
The chemical nature, size, and number of crystals in a ceramic matrix will determine the amount of light that is absorbed, reflected, and transmitted compared with the wavelength of the source light.46 Therefore, the greater the number of crystals in the glassy matrix, the less translucent the ceramic. The greater the amount of unfilled glassy matrix (as in feldspathic porcelains), the more light that can travel through unobstructed, producing more translucency. Zirconia dioxide, which is lacking a glass matrix, has the highest opacity.
There are a variety of high-strength core materials, but the opacity of the core has an effect on the overall esthetics of the restoration. In their study, Heffernan and colleagues compared the translucency of six all-ceramic system core materials at clinically appropriate thicknesses using CRs and listed their findings in order of most translucent to most opaque.46 The researchers concluded that there was a range of ceramic core translucency at clinically relevant core thicknesses.46,47
Classification by Fracture Resistance
A quantitative way of expressing a ceramic’s resistance to brittle fracture when a crack is present is referred to as the “fracture toughness,” which is the ability to resist crack growth.1 If a material has a large value of fracture toughness, it will probably undergo ductile fracture. Brittle fracture is very characteristic of materials with a low fracture toughness value.48 Flexural strength (modulus of rupture or bend strength) is defined as a material’s ability to resist deformation under load. Flexural strength represents the highest stress experienced within the material at its moment of rupture and is measured in terms of stress.7 For example, zirconia’s reported flexural strength values range between 900 MPa and 1,100 MPa,49,50 and fracture has been reported between 8 MPa and 10 MPa.49
When comparing the flexural strength of ceramic material, in descending order the strongest is YZ® Zirconia (Vident™, www.vident.com), followed by In-Ceram® Zirconia (Vident), Procera® Alumina (Nobel Biocare, www.nobelbiocare.com), In-Ceram Alumina (Vident), lithium disilicate, In-Ceram Spinell (Vident), Empress® 1 (Ivoclar Vivadent, www.ivoclarvivadent.com), Vita Omega 900 (Vita Zahnfabrik, www.vita-zahnfabrik.com), Vita VM®9 (Vident), and conventional feldspathic porcelain.51,52
Classification by Abrasiveness
Ceramic restorations have been known to cause wear of opposing enamel.53 The abrasiveness of a dental ceramic is mainly determined by the smoothness of the material.54 For wear to occur, there must be friction developed by mechanical interlocking between the two wear bodies. Low-fusing porcelains were developed to incorporate finer-sized leucite particles in lower concentrations with the idea of lowering the abrasiveness of the ceramic surface.
In their study, Elmaria and colleagues53 compared the wear on opposing enamel by various restorative materials. These included gold, glazed, and polished or glazed-only Finesse® (Dentsply International, www.dentsply.com) (a low-leucite−containing ceramic), Procera AllCeram™ (Nobel Biocare), and IPS Empress (Ivoclar Vivadent). They found that gold, glazed-and-polished Finesse, and glazed-and-polished AllCeram were the least abrasive, whereas glazed-only IPS Empress was the most abrasive.
Because there are two different scenarios, strictly classifying ceramics by their abrasiveness can present a problem when measuring surface roughness. One scenario is the surface roughness after fabrication and the type of finishing process (glazed only or glazed and polished). The other scenario is measuring the surface roughness after any adjustments are made intraorally. Kou and colleagues55 evaluated the surface roughness of five different dental ceramic core materials after grinding and polishing. The samples included Vita In-Ceram® Alumina, Vita In-Ceram® Zirconia, IPS Empress 2 (Ivoclar Vivadent), Procera AllCeram, and Denzir (Denzir, www.denzir.com). A reference material was also included (Vita Mark II®, now Vitablocs® Mark II, Vident). Using a profilometer, the surface roughness (Ra value [μm]) was noted. The measurements were made before and after grinding with diamond rotary cutting instruments and after polishing with the Sof-Lex™ system (3M ESPE, www.3mespe.com). Before grinding, Procera AllCeram and Denzir had the smoothest surfaces, whereas IPS Empress 2 had the coarsest. After grinding, all materials except IPS Empress 2 became coarser. Polishing with Sof-Lex provided no significant differences between Denzir, Vita Mark II, and IPS Empress 2 or between Procera AllCeram and In-Ceram Zirconia. No significant difference was found between the ground and the polished Procera AllCeram or In-Ceram Alumina specimens. Smoother surfaces were found on Denzir, IPS Empress 2, and In-Ceram Zirconia after polishing the ground surface, whereas the polishing effect on Procera AllCeram and In-Ceram Alumina was ineffective.
Heintze and colleagues56 evaluated 20 in vitro studies in which a material and the antagonist wear of the same material were studied. They found that the results were inconsistent, mainly because of the fact that the test parameters differed widely. The test parameters differed in the amount of force, the number of cycles, the frequency of cycles, and the number of specimens. They concluded, as far as consistency and correlation with clinical studies is concerned, that the set-up of the unprepared enamel of molar cusps against glazed crowns seems to be the most appropriate method to evaluate a ceramic material with regard to antagonist wear. However, because of the high variability of the results, large sample sizes are necessary to differentiate between materials, which calls the whole in vitro approach into question.
Conclusion
There are a variety of ways to classify ceramic materials. As manufacturers continue to introduce new materials and formulations, classifications can change or new categories can be introduced. Laboratory technicians should be cognizant of changes in material selection and continue to base their choices on the clinical needs of the patient in terms of esthetics and strength.
References
1. Mclaren EA, Cao PT. Ceramics in dentistry—part I: classes of materials . Inside Dentistry. 2009;5(9):94-105.
2. Taylor JA. History of Dentistry: A Practical Treatise for the Use of Dental Students and Practitioners. New York, NY: Lea and Febiger; 1922:142-156.
3. Asgar K. Casting metals in dentistry: past-present-future . Adv Dent Res. 1988;2(1):33-43.
4. Kelly JR, Nishimura I, Campbell SD. Ceramics in dentistry: historical roots and current perspectives.J Prosth Dent. 1996;75(1):18-32.
5. Leinfelder KF, Kurdziolek SM. Contemporary CAD/CAM technologies: the evolution of restorative systems . Pract Proced Aesthet Dent. 2004;16(3):224-231.
6. Kelly JR . Dental ceramics: what is this stuff anyway? J Am Dent Assoc. 2008;139(suppl):S4-S7.
7. Anusavice KJ . Phillips’ Science of Dental Materials. 10th ed. Philadelphia, PA: WB Saunders; 1996.
8. Powers JM, Sakaguichi RL . Craig’s Restorative Dental Materials. 12th ed. St. Louis, MO: Mosby Elsevier: 2006:445.
9. Denry I, Holloway JA. Ceramics for dental applications: a review . Materials. 2010;3(1):351-368.
10. Powers JM, Sakaguichi RL . Craig’s Restorative Dental Materials. 12th ed. St. Louis, MO: Mosby Elsevier; 2006:454.
11. Helvey GA. A history of dental ceramics . Compend Contin Educ Dent. 2010;31(4):1-3.
12. Luthardt RG, Sandkuhl O, Herold V, Walter MH. Accuracy of mechanical digitizing with a CAD/CAM system for fixed restorations . Int J Prosthodont. 2001;14(2):146-151.
13. Beuer F, Schweiger J, Edelhoff D. Digital dentistry: an overview of recent developments for CAD/CAM generated restorations . Br Dent J. 2008;204(9):505-511.
14. Silva NR, Witek L, Coelho PG, et al. Additive CAD/CAM process for dental prostheses . J Prosthodont. 2011;20(2):93-96.
15. Helvey GA. Chairside CAD/CAM: lithium disilicate restoration for anterior teeth made simple . Inside Dentistry. 2009;5(10);58-67.
16. Leinfelder KL. Porcelain esthetics for the 21st century . J Am Dent Assoc. 2000;131(suppl 1):S47-S51.
17. Martinez Rus F, Pradies Ramiro G, Suarez Garcia MaJ, Rivera Gomez B. Dental ceramics: classification and selection criteria . RCOE. 2007;12(4):253-263.
18. Cesar PF, Gonzaga CC, Miranda Júnior WG, Okada CY. Correlation between fracture toughness and leucite content in dental porcelains . J Dent. 2005;33(9):721-729.
19. Probster L, Geis-Gerstorfer J, Kirchner E, Kanjantra P. In vitro evaluation of a glass-ceramic restorative material . J Oral Rehabil. 1997;24(9):636-645.
20. McLean J. The Science and Art of Dental Ceramics. Chicago, IL: Quintessence Publishing Co Inc; 1979.
21. Ohyama T, Yoshinari M, Oda Y. Effects of cyclic loading on the strength of all-ceramic materials . Int J Prosthodont. 1999;12(1):28-37.
22. Sorensen JA, Choi C, Fanuscu MI, Mito WT. IPS Empress crown system: three-year clinical trial results . J Calif Dent Assoc. 1998;26(2):130-136.
23. Dong JK, Luthy H, Wohlwend A, Scharer P. Heat-pressed ceramics: technology and strength . Int J Prosthodont. 1992;5(1):9-16.
24. McLean JW. Evolution of dental ceramics in the twentieth century . J Prosthet Dent. 2001;85(1):61-66.
25. Sherrill CA, O’Brien WJ. Transverse strength of aluminous and feldspathic porcelain . J Dent Res. 1974;53:683-690.
26. Chaiyabutr Y, Giordano R, Pober R. The effect of different powder particle size on mechanical properties of sintered alumina, resin- and glass-infused alumina . J Biomed Mater Res B Appl Biomater. 2009;88(2):502-508.
27. Balakrishna P, Murty BN, Anuradha M. A new process based agglomeration parameter to characterize ceramic powders . Journal of Nuclear Materials. 2009;384:190-193.
28. Della Bona A, Mecholsky JJ Jr, Anusavice KJ. Fracture behavior of Lithia disilicate and leucite based ceramics . Dent Mater. 2004;20(10):956-962.
29. Shenoy A, Shenoy N. Dental ceramics: an update . J Conserv Dent. 2010;13(4):195-203.
30. Piconi C, Maccauro G. Zirconia as a ceramic biomaterial . Biomaterials. 1999;20(1):1-25.
31. Duran P, Moure C. Sintering at near theoretical density and properties of PZT ceramics chemically prepared . J Mater Sci. 1985;20(3):827-833.
32. Helvey GA. Zirconia and computer-aided design/computer-aided manufacturing (CAD/CAM) dentistry . Inside Dentistry. 2008;4(4):72-79.
33. Christel P, Meunier A, Heller M, et al. Mechanical properties and short-term in-vivo evaluation of yttrium-oxide-partially-stabilized zirconia . J Biomed Mater Res. 1989;23(1):45-61.
34. Raigrodski AJ. Contemporary all-ceramic fixed partial dentures: a review . Dent Clin North Am. 2004;48(2):531-544.
35. Hauptmann H, Suttor D, Frank S, Hoescheler H. Material properties of all-ceramic zirconia prosthesis [abstract] . J Dent Res. 2000;79(suppl 1):S507.
36. Roundtree P, Nothdurft F, Pospiech P. In-vitro investigations on the fracture strength of all-ceramic bridges of ZrO2-ceramic [abstract] . J Dent Res. 2001;80:57.
37. Brodbelt RH, O’Brien WJ, Fan PL, et al. Translucency of human dental enamel . J Dent Res. 1981;60(10):1749-1753.
38. Seghi R R, Johnston WM, O’Brien WJ. Spectrophotometric analysis of color differences between porcelain systems . J Prosthet Dent. 1986;56(1):35-40.
39. Brodbelt RH, O’Brien WJ, Fan PL. Translucency of dental porcelains . J Dent Res. 1980;59(1):70-75.
40. Chu F, Chow TW, Chai J. Contrast ratios and masking ability of three types of ceramic veneers . J Prosthet Dent. 2007;98(5):359-364.
41. Ozturk O, Uludag B, Usumez A, et al. The effect of ceramic thickness and number of firings on the color of two all-ceramic systems . J Prosthet Dent. 2008;100(2):99-106.
42. Barath VS, Faber FJ, Westland S, Niedermeier W. Spectrophotometric analysis of all-ceramic materials and their interaction with luting agents and different backgrounds . Adv Dent Res. 2003;17:55-60.
43. Yu B, Lee YK. Color difference of all-ceramic materials by the change of illuminants . Am J Dent. 2009;22(2):73-78.
44. Johnston WM, Ma T, Kienle BH. Translucency parameter of colorants for maxillofacial prostheses . Int J Prosthodont. 1995;8(1):79-86.
45. Liu M C, Aquilino SA, Lund PS, et al. Human perception of dental porcelain translucency correlated to spectrophotometric measurements . J Prosthodont. 2010;19(3):187-193.
46. Heffernan MJ, Aquilino SA, Diaz-Arnold AM, et al. Relative translucency of six all-ceramic systems. Part I: core materials . J Prosthet Dent. 2002;88(1):4-9.
47. Barizon KTL. Relative Translucency of Ceramic Systems for Porcelain Veneers. [master’s thesis]. Iowa City, IA: University of Iowa; 2011.
48. Hertzberg RW . Deformation and Fracture Mechanics of Engineering Materials. 4th ed. Hoboken, NJ: Wiley; 1995.
49. Piwowarczyk A, Ottl P, Lauer HC, Kuretzky T. A clinical report and overview of scientific studies and clinical procedures conducted on 3M ESPE Lava All-Ceramic System . J Prosthodont. 2005;14(1):39-45.
50. Papanagiotou HP, Morgano SM, Giordano RA, Pober R. In vitro evaluation of low-temperature aging effects and finishing procedures on the flexural strength and structural stability of Y-TZP dental ceramics . J Prosthet Dent. 2006;96(3):383-388.
51. Rekow ED, Silva NR, Coelbo PG, et al. Performance of dental ceramics . J Dent Res. 2011;90(8):937-952.
52. McLaren EA, Giordano RA II. Zirconia-based ceramics: material properties, esthetics, and layering techniques of a new veneering porcelain, VM9 . Quintessence Dent Technol. 2005;28:99-111.
53. Elmaria A, Goldstein G, Vijayaraghavan T, et al. An evaluation of wear when enamel is opposed by various ceramic materials and gold . J Prosthet Dent. 2006;96(5):345-353.
54. Oh WS, DeLong R, Anusavice K. Factors affecting enamel and ceramic wear: a literature review . J Prosthet Dent. 2002;87(4):451-459.
55. Kou W, Molin M, Sjögren G. Surface roughness of five different dental ceramic core materials after grinding and polishing . J Oral Rehab. 2006;33(2):117-124.
56. Heintze SD, Cavalleri A, Forjanic M, et al. Wear of ceramic and antagonist-a systematic evaluation of influencing factors in vitro . Dent Mater. 2008;24(4):433-449.
About the author
Gregg A. Helvey, DD
Virginia Commonwealth University School of Dentistry
Richmond, Virginia
Private Practice
Middleburg, Virginia